Pharmacokinetics on Two Pegylated Interferons Reported at AASLD 1999
This report contains PK data reported by both Schering and Roche on their pegylated interferon products.
A BRANCHED METHOXY 40 KDA POLYETHYLENE GLYCOL (PEG) MOIETY OPTIMIZES THE PHARMACOKINETICS (PK) OF PEGINTERFERON a-2A (PEG-IFN) AND MAY EXPLAIN ITS ENHANCED EFFICACY IN CHRONIC HEPATITIS C (CHC)
N E Algranati, and others with Hoffmann-La Roche reported on the PK of their PEG-IFN drug in development now. They said standard TIW IFN dosing may allow viral replication during times that IFN concentrations are not measurable. PEG-IFN is a modified form of recombinant IFN by covalent attachment of a branched PEG moiety. Pegylation of interferon slows the metabolism significantly, allowing for a once weekly
injection instead of the 3-times weekly approved dosing for the non-pegylated form.
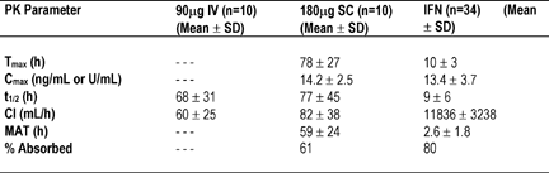
This study was intended to characterize the PK profile of PEG-IFN. The PK of PEG-IFN after a single 180mg SC dose or a 90mg IV dose were characterized in 20 healthy subjects. Serial blood samples were collected for up to 168 hours.
Data from PEG-IFN were compared to published results from IFN. The authors reported that after sub-cutaneous dosing, substantial PEG-IFN concentrations are seen 3 to 8 hours after dosing. Pegylation reduces clearance (Cl) 100-fold. Sustained absorption (MAT, mean absorption time; time required for 50% to be absorbed) and reduced clearance of PEG-IFN provide sustained serum concentrations after SC administration. >60% of the dose is absorbed from the SC route (Table).
As you can see in Table below the the half-life of Pegasys was found to be 77 hours (after 77 hrs. half of the amount of drug remained in blood), while a standard interferon injection had a half-life of nine hours. Glue concluded this PK profile provides antiviral exposure throughout 1 week and appears to be the key to enhanced efficacy of PEG-IFN in CHC.

PEG-INTERFERON-a2B: PHARMACOKINETICS, PHARMACODYNAMICS, SAFETY AND PRELIMINARY EFFICACY DATA
Paul Glue and others from Schering-Plough reported on this study at the Dallas Liver Meeting (Nov '99) which assessed the biologic activities, pharmacokinetics (PK), pharmacodynamics (PD) and safety of Schering's pegylated interferon-a2b (PEG-Intron).
Clinical PEG-Intron doses of 0.035-2mg/kg s.c. were administered once weekly for 24 weeks to treatment-naive HCV-infected patients (n=6/dose level). PK was assessed at weeks 1 and 4; safety, HCV-RNA 2'5'-OAS, and neopterin over 24 weeks treatment and follow-up. Intron A 3MIU TIW was the active control.
Glue reported PEG-Intron by itself is "biologically active" with antiviral activity (~25% of interferon-a2b). The cleaved product, interferon-a2b, is 100% biologically active. PEG-Intron showed antiviral activity in an HCV surrogate virus (BVDV) cell-based system.
Both PEG-Intron and Intron A were rapidly absorbed with maximal concentrations occuring ~8-12 hours post-dose. PEG-Intron had sustained maximal serum concentrations for 48-72 hours post-dose, while Intron A concentrations declined rapidly. Volume of distribution for both compounds was similar (~IL/kg). Upon multiple dosing there was an apparent increase in bioavailability for both Intron A (accumulation factor, R=2.1) and PEG-Intron (R=1.2-1.4). PEG-Intron had delayed elimination compared with Intron A (PEG-Intron t1/2=54 hr., vs Intron A =8.1 hours), and mean apparent clearance of PEG-Intron (0.013 L/hr/kg) was ~10% that of Intron A (0.11 L/hr/kg).
Both Intron A and PEG Intron had similar profiles for safety (reduced platelet and neutrophil counts) and tolerability (fever, flu-like symptoms). PEG-Intron-induced decreases in platelet and neutrophil counts were maximal after the first dose, and remained stable over 24 weeks of treatment. The magnitude of these decreases was dose-related, although the decline became asymptotic beyond 1-1.5m/kg.
PEG-Intron increased serum neopterin and 2'5-OAS concentrations over the entire treatment period. HCV-RNA titers were decreased by PEG-Intron. The magnitude of the initial fall in HCV-RNA was dose-related, and similar trends were noted for virologic response during follow-up.
Authors concluded-- (1) PEG-Intron is a "biologically active molecule" and demonstrated antiviral activity in vitro. (2) PEG-Intron has delayed clearance characteristics, consistent with once weekly dosing. (3) PEG-Intron's safety and pharmacodynamic profiles are similar to those of Intron A. (4) At doses above 0.5mg/kg once weekly, PEG-Intron appears to have at least equivalent, if not superior efficacy to Intron A 3MIU TIW.