
Several studies on
combining ritonavir with amprenavir, indinavir or nelfinavir were presented at
Durban. Ritonavir is a potent inhibitor of cytochrome (CYP) p450 3A, the liver
and gut wall cytochrome responsible for metabolism of many drugs, including
other protease inhibitors. By inhibiting metabolism of other drugs through this
means, ritonavir increases exposure (increases amount of drug in body) and slows
the metabolism of other protease inhibitors. This can reduce the overall number
of pills taken and reduce dosing frequency. It can also remove the need for food
restrictions. It also offers adherence flexibility because blood levels of the
other PI are higher. Another benefit from combining RTV with another PI is
possible. At the First International Workshop on Clinical Pharmacology of HIV
Therapy in March 2000 in Amsterdam, R Garraffo from University Hospital in Nice,
France reported on in vitro (in the test tube) data showing that ritonavir
inhibited efflux (outflow) from cells of other protease inhibitors, with the
most affect on amprenavir and indinavir. Garraffo speculated that this effect
could reduce failure and resistance mutations. However, this needs to be
confirmed in human studies, and as the Garaffo stated, the dosing of RTV needed
for inhibition in humans remains to be identified.
On the other hand,
ritonavir can increase cholesterol and triglycerides. And inhibition of CYP3A
may cause interactions with legal and illegal drugs. Additionally, the long-term
consequences, if any, of chronically inhibiting CYP3A and P-glycoprotein is not
known.
RTV
Intensification of IDV.
Andy Zolopa followed up on his initial Retrovirus report of ritonavir
intensification of an indinavir regimen at Sitges & Durban. Zolopa set out
to test if increased trough of IDV, which results from giving 400/400 RTV/IDV
compared to 800 IDV every 8 hrs, produces greater virologic suppression in
patients with detectable virus while receiving an IDV based regimen; also to
study the effect of baseline genotype on response to IDV intensification with
RTV.
The median baseline
viral load was 2000 copies/ml (3.3 log) for the 37 individuals in the study, and
24 had baseline viral load between 50-400 copies/ml. Their median CD4 count was
390, and the median IDV experience was 32 months. Investigators used the Roche
Ultra-sensitive <50-copy viral load test. During the first 3 weeks of the
study, the only change in the regimen was the substitution of RTD/IDV 400/400 mg
bid for 800 mg every 8 hours using RTV dose escalation. Dose titration was
allowed during the first 2 weeks. A sub-study (n=16) evaluated IDV PK profiles
at baseline and RTV and IDV pk at week 3. Results confirmed what they had
previously seen in healthy volunteers: IDV Cmin increased 272%, Cmax decreased
51%, Ctrough (n=27) increased 386%, and AUC-24 increased 11%. 26/27 had
increased Ctrough.
At week 24, 52% of the
patients remaining on therapy had <400 copies/ml, 44% had <50 copies/ml,
and CD4s increased 23. Two patientsÌ dose reduced to 300/400 RTV/IDV bid.
The median IDV Ctrough
at week 3 was 0.792 ug/mL including all subjects with IDV Ctrough at week 3
(n=32), compared to the median baseline Ctrough of 0.13 (n=27). Interestingly,
the median decline in HIV viral load was greater in patients with IDV Ctrough
higher than the median. 75% of patients with Ctrough higher than the median at
week 24 had <400 copies/ml, while 37% with less than the median trough had
<400 copies/ml.
Genotype was available
for 30 subjects. Greater than 25% prevalence of PI mutations was found at
positions 10, 46, 54, 71, 82, and 90. By on-treatment analysis, at week 24,
virologic response (<400 copies/ml) was observed in 77% (10/13) of subjects
with 0-3 baseline mutations, and 22% (2/9) with 4-7 baseline mutations in HIV
protease (p=0.027). Zolopa also reported that the median decline in viral load
over the first 3 weeks of therapy was greater in those subjects with week 3 IDV
Ctrough higher than 0.792 ug/mL, regardless of baseline genotype. Zolopa
concluded that these data suggest that drug trough concentrations and HIV drug
susceptibility are the most important predictors of drug potency with protease
inhibitors.
Of the 37 subjects
enrolled 11 discontinued, 10 due to adverse events: GI (3), anxiety (1),
hypercholesterimia (1), Kidney calculus (1)--patient mistakenly took 800/400 IDV/RTV
bid, lactic acidosis (1), lipodystrophy (1), rash (1), somnolence (1). Adverse
events reported by 2 or more patients of at least moderate severity and possibly
or probably study drug related (n=37): diarrhea (15), hypercholesteremia
(9), hyperlipidemia (8), abdominal pain (6), weight loss (5), asthenia (2),
nausea (2), somnolence (2). Abnormal labs
(n=36) of grade 3 or 4: triglycerides (>750) 18 non-fasting and 11/13
fasting; cholesterol (>300) 14 non-fasting, and 11/12 fasting; Grade 2:
AST/ALT (>2.5 x ULN) 6, total bilirubin (>1.5 x ULN) 1I; there were no
reports of grade 3 or more liver function abnormalities.
Two studies, NICE and
BEST, reported on switching patients who were taking the standard IDV dose of
800 mg 3 times daily to twice daily RTV/IDV at the doses of 400/400 or 800/100
RTV/IDV. The 400/400mg of each drug bid dose may mean that therapeutic drug
blood levels of both ritonavir and indinavir are achieved, but is associated
with lower indinavir peak level (compared to 800/100), which may be associated
with risk to kidney stones (nephrolithiasis). However, it remains uncertain if
kidney stones are related to peak or AUC IDV levels. In addition, as you read
through the studies below bear in mind that weather conditions may be associated
with kidney stones. If the temperature is high, whether it be in Spain, Texas or
New York City, additional hydration may be required. Using 100mg of ritonavir
with 800mg indinavir bid uses fewer pills (3 bid versus 5 bid with 400/400), and
has a lower risk of ritonavir related side effects, such as cholesterol &
trygliceride elevations, nausea and diarrhea.
400/400
RTV/IDV. The NICE study, reported
by DeJesus, looked at people with HIV-RNA <400 copies/mL in an open-label
multi-center study comparing continuing indinavir 800mg tid to switching to
400/400mg bid of ritonavir/indinavir. The purpose of this study is to evaluate
adherence and convenience as reported by patient survey at weeks 4, 12 and 24
adherence and convenience. Safety and efficacy are being evaluated but were not
presented yet. The study has recruited 380 patients, 301 randomized to add
ritonavir, and 79 to remain on tid indinavir. Interim results after 4 weeks show
21% of those adding ritonavir and 15% remaining on indinavir alone have
discontinued. Adverse events, mainly gastrointestinal, were the cause of
discontinuation in 14% and 8% of patients, respectively. The adherence survey at
week 4 visit show that patients randomized to add ritonavir reported missing
fewer doses than they had reported missing prior to initiation of this regimen .
Additionally, they reported that it was easier to take the medication as
prescribed, easier to take at the same time each day, and easier to coordinate
with meals when compared to baseline. Similar effects were not seen for patients
who remained on indinavir alone. It seems strange to me that the discontinuation
rates were so high in both arms after only 4 weeks. So, by switching to 400/400
from IDV standard dosing, convenience and adherence improved while side effects
increased.
800/100
RTV/IDV. The BEST study (authors:
Gatell & Lange) reported a similar result. This study, however, switched
people from standard IDV dosing to 800/100 IDV/RTV twice daily (bid), and is a
randomized, open-label, multi-center study testing switching 326 patients with
viral loads <500 copies/ml. 161 added ritonavir, 165 remained on indinavir
alone.
At week 24, all 237
patients completed 3 months follow-up. Baseline CD4 counts were 407 and 440/mm3
for tid and bid arms, respectively. At 6 months the percent of patients with
viral load <500 copies/ml were: (See
Table 12)

Adverse events requiring
treatment interruption or discontinuation occurred in 7 (TDV TID) versus 19% of
patients (RTV/IDV 100/800 bid), mostly due to GI intolerance. The RTV liquid
form may have caused GI intolerance. Nephrolithiasis developed in 5% versus 12%
(IDV/RTV 800/100), leading to treatment discontinuation in 2% versus 3%,
respectively. The high rate of nephrolithiasis developing so quickly underscores
the need for continued high fluid intake on this dosing schedule. Again, bear in
mind varying temperature conditions in different areas. Nausea and/or vomiting
occurred in 2 versus 16% of patients, possible in part due to the use of liquid
ritonavir during the first months of the study. No differences in triglycerides
or cholesterol >3 times upper limit of normal were observed. In certain
salvage situations where higher blood levels may be desired using 800/200 may be
preferred, but after achieving <50 copies/ml it may be preferable to switch
to 400/400 to avoid kidney problems.
Amprenavir
+ Ritonavir. In evaluating
the PK interaction between Ritonavir and amprenavir three RTV-APV regimens were
tested in 12 healthy volunteers per group: Group I 400mg Ritonavir + 450mg
Amprenavir, group II 400/750 and group III 200/1200. Abbott reported 19
withdrawals due to amprenavir rash (despite excluding persons with known sulpha
drug allergy) which meant only seventeen subjects finished the 12-day study.
However, Glaxo Wellcome has not reported such incidence of rash in their studies
and they doubt the validity of such an occurrence and the Cmin finding of 0.63
in Group 2. Pharmacokinetic parameters were compared with historical controls
taking the approved 1200mg bid dose. PK
parameters are shown in table 13, below:
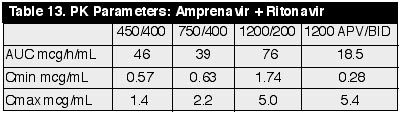
APV AUC, Cmax & Cmin
in groups 1 and 2 are not statistically different. Median RTV Cmin values were
2.1, 2.9, and 0.62 mcg/mL for groups 1, 2, and 3, respectively. RTV and APV IC50
in 50% human serum were reported by Ann Hsu of Abbott to be about 0.9 and 0.3
mcg/mL, respectively . Group. III had significantly higher mean amprenavir
exposure than those of Groups I and II, but an elimination half-life 50%
shorter. APV appeared to have no effect on RTV pk. These data suggest ritonavir
improves the PK profile of amprenavir with all three regimens yielding trough
values at 12 hours above current recommended amprenavir dosing. In question is
the degree of increase and benefit in APV blood levels (the increase is not
linear according to GW) comparing 600/100 APV/RTV bid to 1200/200 APV/RTV bid.
In a simulated model looking at 600/100 APV/RTV, APV Cmin was 1.99 mcg/mL (this
questions the Cmin reported above by Abbott of 0.63 for the 400750 arm) and 0-24
hr. AUC was 66.1 mcg*hr/mL. In a NIH salvage study in 10 individuals looking at
APV/RTV interaction, APV/RTV 1200/200 bid + abacavir yielded APV Cmin of 2.18
mcg/mL (AUC 43). APV/RTV 1200/500 bid + abacavir yielded APV Cmin of 2.30. So
the difference in Cmin was little comparing 500 to 200 RTV doses. Glaxo Wellcome
says 600/100 gives you higher levels than the IC50 of most resistant viruses,
and suggests 1200/100 bid rather than using 200 RTV is sufficient in covering
extremely resistant virus, but clinical studies have not yet been done to
confirm these dose combinations.
APV/RTV
1200/200 BID Retrospective Study.
R Hsu with Abbott and Paul Bellman, a practicing physician in NYC presented a
poster of Amprenavir usage in the highly experienced patient. This is a
retrospective look at 30 patients who had a median VL >110,000 copies/ml
prior to starting Amprenavir/Ritonavir. They had an average 5.2 NRTI mutations,
1.4 NNRTI mutations, and 4.8 PI mutations. Median starting CD4Ìs for the group
was 63 cells/mm3. What makes this Amprenavir salvage study different from
previous and less impressive Amprenavir salvage studies is the addition of RTV.
The doses were Amprenavir 1200mg bid, Ritonavir 200mg bid, along with various
other antivirals. Other physicians are using 1200/200 dosing to achieve high
blood levels of amprenavir when patients have extensive resistance. Side effects
were reported to be generally tolerated. The most common were diarrhea: 23% and
nausea: 7%. Only 2 patients discontinued due to side effects, one due to nausea
and one due to pill burden. The current formulation of amprenavir is 150 mg
pills so 1200 mg is 8 pills and is being combined in this regimen with 2 100 mg.
pills of ritonavir. 20 out of the 30 patients had a sustained virologic
suppression defined as significant decrease in viremia without rebound over 24
weeks. 10 out of the 30 patients experienced only transient viral response prior
to rebounding. And 10 of the 20 sustained virologic responders were suppressed
to <400 copies/ml at 24 weeks. CD4+Ìs increased a median of 149 cells/mm3
over the 24-week observation. Glaxo is exploring a range of Ritonavir enhanced
doses of Amprenavir including 600/100 bid, 900/100 bid, and 450/100 bid.
72-Week
RTV/IDV 400/400 bid in Treatment Na‘ve with High Viral Load.
JK Rockstroh and colleagues looked at a good sized group of ARV na‘ve patients
(n = 92) in a prospective, open-labeled, multi-center study. They gave them RTV
400mg bid and IDV 400mg bid with 2 background nucs. 49% had Combivir, 37% had
Zerit and Epivir, and 14% had Videx and Zerit. Median VL on entry was a high
220,400 copies/ml, and CD4 was 192. At 72 weeks, 56 patients remained on their
therapy and 100% of those were <500 copies/ml and 95% were <80 copies/ml.
36 patients in total dropped out, 10 due to AEÌs. 17 quit because they
didnÌt like oral liquid Ritonavir. The median increase in CD4Ìs over the 72
weeks was 275 cells/mm3. And the median viral load drop was 3.6 log. A side
question that was addressed by this group was whether folks who use RTV 400 with
IDV 400 bid fare better than IDV alone when looking at renal AEÌs from IDV.
This study did a sub-analysis of 27 patients looking at that question and found
that when compared to the 20 patients in the Boubaker Study, published in AIDS
1998 who were on IDV 800 tid, they did have a significant difference/decrease in
serum creatinine. When looking at all the patients in this study there were no
cases of kidney stones, flank pain, or elevations of creatinine reported.
Rockstroh reported that the quadruple therapy was well tolerated except for mild
diarrhea, initial nausea and increased triglyceride and cholesterol levels.
As with any study that
includes RTV there were increases in tryglycerides and cholesterol. The
triglycerides increased from a mean of 132 at baseline to 304 at 72 weeks, and
the cholesterol increased from 179 to 251. This study did have a greater % of
dropouts, which may be explained by the oral formulation of RTV. Longer term
effect of pk enhanced IDV on kidney has not been evaluated yet.
RTV/IDV
800/100 in Naive. A German
research group led by A. Wickesberg and colleagues tried IDV 800 bid and RTV 100
bid with 2 or 3 nucs as background HAART. In this study, 48 na‘ve patients were
enrolled. Baseline viral load was 320,000 or 5.5 log copies/ml. Median baseline
cd4+ was 50, which is lower then the group reported on above, who were also na‘ve,
but went on RTV 400 and IDV 400 with impressive results. At 24 weeks, in this
800/100mg study, 25% of patients had left the study. They reported 8 cases of
kidney stones. There was a significant decrease in viral load of 1.84 log
copies/ml at this 24-week juncture. T-cells increased over 220 to a median of
270.5 cells/mm3. For those who remained on therapy at 24 weeks, 95.8% had <
400 copies/ml HIV RNA and 75% had <50 copies/ml. The larger doses of RTV also
probably add to enhanced virologic potency of the regimen allowing greater
numbers of individuals to reach undetectability. However, the tolerability of
RTV 400mg bid is less than at 200mg bid and with more RTV one sees a greater
increase in lipids.
Increased plasma drug
levels using twice daily ritonavir/indinavir at 100/800 mg correlate with
virological response in a salvage therapy.
This study was reported
at the Resistance Workshop in June. A Moreno and a research group from Ramon y
Cajal Hospital in Madrid, Spain on this study which was intended to evaluate the
efficacy of indinavir in a twice daily dosing regimen with co-administration of
100 mg of ritonavir, and to explore the influence of plasma drug levels in the
rate of virologic response.
This is a prospective
study of 59 patients extensively pretreated and switching to a 4-drug salvage
regimen including two NRTIs plus the twice-daily combination of IDV 800 mg + RTV
100 mg, after meals. Pharmacokinetics of indinavir and ritonavir was assessed in
11 patients.
Previous ART exposure
was 44 months, and 78% and 39% of patients had previously failed regimens with
either IDV or RTV. Median CD4 was 248 and HIV RNA was 3.9 log (7900 copies/ml).
The median number of mutations in the protease gene was 9 (3-14), predominantly
at positions 82 (53%), 90 (42%), and 46 (32%). After 24 weeks, 61% of patients
had a viral load decrease greater than 1 log, and 38% were below 50 copies/ml.
Renal toxicity was observed in 33%, but again this study was conducted in a warm
climate and additional hydration may be needed. Drug discontinuation was
necessary in 6 cases (10%), but in 7 IDV/RTV dose adjustments to 400/400 avoided
treatment withdrawal. Indinavir trough levels were well above the IC95 (median
1.75 mg/l, IQR 1.07-2.57), but ritonavir trough levels were below the IC95 in
88% of patients. At month 3, patients with IDV levels above the median (>7.09
mg/l) had a mean viral load decrease of 2.86 log in comparison to 0.66 log in
those below the median IDV peak level (p=0.04), and at month 6 the decrease was
1.75 versus 0.5 log. After 24 weeks, the median decrease in viral load in
patients exhibiting or not exhibiting the V82A and L90M mutations were 1.33 log
and 1.95 log copies/ml, respectively. In addition, a close correlation was
observed between higher peak levels of IDV and nephrotoxicity.
800/200
Ritonavir+Indinavir. Jon Condra of
Merck suggested at the Salvage Workshop in April that 800mg indinavir+200 mg
ritonavir ought to be more effective for salvage because the 800/200 dose
produces a higher trough level of indinavir. But, renal or kidney concerns
appear heightened with higher IDV blood levels. Initial 800/200 can be dose
reduced after induction. In addition to Condra's talk, two private practitioners
reported their retrospective analyses of patients who had used 800/200. Howard
Grossman's report on 41 patients found no cases of kidney stones. The second
report by Rafael Campo from the University of Miami did not mention whether or
not they observed kidney stones. It's possible that Grossman and Campo
instructed their patients to use extensive fluid hydration, and thety practiced
it, while patients in other studies were not as attentive.
In Condra's report he
presented in vitro data from looking at a panel of 20 PI-resistant viral
isolates from patients who experienced long-term virologic failure of IDV
monotherapy, patients failing nelfinavir as their first PI, and from patients
who failed IDV following their initial nelfinavir failure. All viral isolates
except one contained multiple IDV mutations. The one exception had NFV related
mutations only (D30N). Condra reported that most of the viral isolates had
extensive phenotypic resistance to IDV, RTV, and NFV (corrected for human serum
protein binding). Protein binding reduces the amount of drug that the virus is
exposed to in the blood. Different protease inhibitors have different degrees of
protein binding. Using in vitro testing, Condra said that the 800/200 dose
resulted in IDV drug levels 70 times higher than that necessary to suppress 95%
of viral replication (IC95). He said the IDV drug levels achieved exceeded the
IC95 in at least 18 of the 20 viral isolates. The IDV trough levels achieved
with the 800/200 dose are higher than that achieved with the 400/400 or 800/100
dose. As mentioned in a study reported above, patients who started on 800/100
and developed nephrolithiasis were switched to 400/400.
From data initially reported
at the 6th Retrovirus Conference in 1999, Condra reported that the 12 hour trough
of IDV (using a high-fat meal) with the 400/400 dose was 1891 nM, with the 800/100
dose 2233 nM, and with 800/200 it was 5344 nM. The data reported at the 6th
Retrovirus Conference in 1999 can be seen on the NATAP web site at: www.natap.org/indinavirock.html.
Both studies were
retrospective analyses of patient's medical charts. Howard Grossman reported on
41 patients who were treatment experienced (30/41 -73%- had prior NNRTI
experience) and had a median viral load of 30,000 copies/ml. Their median CD4
was 258 (range 113-378). The mean observed time on study therapy was 7.2 months.
Using an as-treated analysis, the proportion of patients that had <400
copies/ml at 3, 6, and 9 months were 51% (21/41), 56% (17/30) and 62% (10/16),
respectively. The dose was 800/200 indinavir/ritonavir. The study design is a
multi-center (6 site) retrospective chart review study of individuals who had
failed at least 1 PI containing regimen. 95% of the patients had prior exposure
to indinavir or ritonavir. The patients had a mean number of 6 prior
antiretroviral (ART) regimens, and a mean number of 3 prior PI regimens. The
average number of NRTIs in the new RTV/IDV regimen was 2 (range 1-4). 7/29
patients were NNRTI na‘ve at baseline, and 29/41 patients had a NNRTI in their
IDV/RTV regimen. After 12 (n=41), 24 (n=30), and 36 (N=16)weeks, the median
viral load reduction from baseline was -1.65 log, -1.46 log, and -1.66 log,
respectively. The median CD4 increase was about 75 at week 24.
Grossman reported there
were no cases of kidney stones, and there were 2 discontinuations. 19 of 41
patients reported side effects. There were 7 cases of nausea & vomiting, 2
cases of diarrhea, 5 cases of rash & dry skin, and 1 case of paresthesia.
Rafael Campo, from the
University of Miami, reported on his retrospective analysis of 27 patient's
medical charts, who started salvage regimens consisting of 800 mg IDV twice
daily and 200 mg of RTV twice daily, with at least one NRTI and/or a NNRTI.
Patients had at least one PI containing regimen which they failed. Failure was
defined as a rebound in viral load to >1000 copies/ml after having a VL
<400 on at least one occasion or as failure to achieve VL <400 after >6
months on the original PI regimen. Responders were defined as patients with a
viral load <400 on at least one occasion after starting the RTV/IDV regimen.
Non-responders were defined as patients who never reached <400 regardless of
how much their viral load decreased.
Campo reported that 4/4
with baseline phenotypic resistance, and 10/13 with baseline genotypic
resistance achieved viral suppression. He associates failure to achieve viral
suppression to non-adherence. Grossman's report did not address adherence of his
patients.
Campo reported that by
reviewing patient's charts he found 15 responders and 12 non-responders followed
for a mean of 32 and 31 weeks, respectively. At initiation of IDV/RTV regimen
CD4 counts were 283 and 150, and viral load was 156,000 copies/ml and 228,000
copies/ml for responders and non-responders, respectively. Both groups had
previously used 6-7 HIV ART drugs. Both groups had 2.8 mean prior HAART
regimens. Their mean duration of prior therapy was 117-137 weeks. Both groups
had received a mean of about 2.5 protease inhibitors, and 2.5 protease inhibitor
regimens. The responder group had 101 weeks mean duration of PI therapy and the
non-responders 86 weeks.
Campo reported that 83%
(5/6) responders had at least 2 new drugs as part of their IDV/RTV regimen,
while only 17% (1/6) of the non-responders had 2 new drugs. He also reported
that 5/8 (63%) of the responders were NNRTI na‘ve and used a NNRTI in their new
regimen, while 3/8 (37%) of the non-responders were NNRTI na‘ve and used a
NNRTI in the new regimen.
8 of the 15 responders
(53%) have maintained VL <400 copies/ml and 3 (20%) have <50 copies/ml. 5
patients (33%) had transient VL rebounds to >400 but it was associated with
non-adherence. Campo reported they returned to <400 upon re-initiation of IDV/RTV
regimen. 2 patients have had viral load rebounds to 151,000 and 2696 copies/ml,
respectively, reportedly due to non-adherence. Their viral loads decreased to
451 and 873 copies/ml, respectively (6 and 14 weeks later, respectively) upon
re-starting the regimen.