Two studies at Durban
studies reported an evaluation of the impact of fat redistribution on quality of
life using a standardized physician and patient questionnaire and visual scales.
In the German study (n=250) patients were mostly (80%) male with a mean age of
39 years. Physicians diagnosed lipodystrophy in 36% of the study group. Patients
with lipodystrophy all reported loss of quality of life in at least one of the
measured parameters: 63% for social contact, 68% on daily performance and
sexuality and 83% on self-esteem. Abdominal enlargement was significantly
associated with diminished sexual and self-esteem ratings, leg and arm changes
were associated with daily performance and social functioning. In a survey of 74
attendees at a workshop at GMHC in NYC on body changes where 78% of respondents
reported body shape or metabolic changes thought related to ART, 30% of these
individuals had changed therapy and 7% interrupted ART specifically due to these
problems. In general (83% of cases), amongst those changing therapy, only the
drug perceived as causing the problem was changed.
In general, the studies
switching to PI-sparing are not comparative. It's difficult to compare to
another arm, because patients either want to switch or they don't, therefore
randomization is difficult. The ACTG is planning a study comparing a switch to
either an abacavir, EFV, or NVP regimen. In general, switches to NVP or EFV arms
do not appear to improve body changes so far in follow-up of ongoing studies
which extends to about 1 year. A Spanish NVP switch study (Lydia Ruiz reported
at Lisbon Conference in Nov. 1999, details on NATAP web site) suggested
improvements in lipoatrophy at 1 year. It's possible that longer follow-up is
required to see improvements. Preliminary data on a switch to an abacavir
regimen at the February Retrovirus Conference suggested improvements in body
changes, and follow-up data is expected to be presented at the end of September
at ICAAC. One study below switches from PI to EFV+abacavir regimen and patient
reports indicate improved body changes. Switching to NVP or abacavir does seem
to generally improve lipid abnormalities but not complete resolution, while
improvements to lipids when switching to EFV is inconsistent (2 studies below
reported reduction in triglycerides) and in general does not appear to improve
lipids, although HDL cholesterol appears to improve. The ACTG is planning an
interesting study combining Metformin and Rosiglytazone, two anti-diabetic
drugs. Metformin may decrease fat accumulation, but it also may decrease fat in
the periphery. Rosiglytazone may increase periphery fat. So, it's hoped the two
together may improve body changes.
In a Spanish study, 110
patients (80% male) were switched to nevirapine after at least 3 months
undetectable (<50) on PI therapy. 72% were naive to antiretrovirals prior to
starting the PI regimen they were receiving at the beginning of the study. 62%
had viral load of 100,000 copies/mL or more when their PI-containing regimen was
started. The reasons for changing PI included treatment simplification (41
patients), renal complications (24), lipodystrophy (20), hypertriglyceridemia
(3), and gastro-intestinal disturbances (3). The mean CD4 count at change was
601 cells (range 80 to 1500). After 1 year, 21% discontinued nevirapine. Three
were lost to follow-up, 3 were receiving methadone and elected to discontinue
NVP, 2 had hepatitis, and 3 had an increase in hepatic enzymes. 7 patients had a
severe rash and 5 had viral rebound, of which 4 were naive to therapy prior to
their PI-containing regimen and 2 had HIV RNA higher than 100,000 copies/mL at
that time. Mean CD4 rose at weeks 12, 24 and 36 by 1, 65 and 85 cells/_L,
respectively. Whereas mean triglyceride at 12, 24 and 36 weeks fell by 125, 16
and 574 mg/dL, respectively.
A large 100 patient
French cohort also evaluated switching to NVP in PI treated patients with plasma
VL <20 copies/ml for >1 year. Data at month 3 on 72 patients indicated a
significant rise in CD4 percentage (28.4% to 30.6%) and a rise in CD4 cells of
39/ mm3. Seven patients (10%) had detectable viral load levels at month 3;
available resistance data suggesting resistance only to NNRTI was present. There
were no significant changes in cholesterol or triglycerides. Twenty-one patients
reported side effects following switch with 5 patients discontinuing nevirapine.
Another cohort of
patients who mostly switched to NVP yielded somewhat disappointing results. In
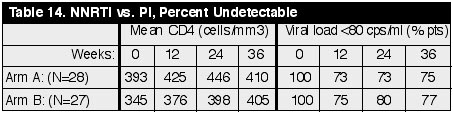
this single centre Italian study, a total of 28 NNRTI-naive, PI-treated patients
with a VL< 80 copies/ml for mean 21 months (range 7-33), and with metabolic
toxicities were switched to NVP (n= 24) or EFV (n=4). These patients (Arm A)
were compared to a cohort who continued the PI-containing regimens (Arm B). The
groups were well matched for age, sex, HIV risk factors, CD4 count when
initiating PI therapy and switching therapy, and for the duration with an
undetectable viral load. Results after 36
weeks are reported below: (See Table 14)
After 36 weeks of
switching therapy mean values of cholesterol, triglycerides, and glucose were
reported to have returned to within normal ranges. While there were no
statistical difference between the percent of patients with undetectable viral
load in the two treatment arms, it is concerning that as many as 25% of patients
in this cohort lost virological control, the majority in the first 12 weeks
after switch (5).
Concern that switching
therapy may be more risky in persons with prior therapy experience led one group
to investigate this approach only in persons on their first regimen. In this
study, 40 patients on PI regimens with undetectable VL for at least 6 months,
switched the PI for NVP 200-mg bid. Nucleosides were maintained. 90% of the
patients were men, with a median CD4 of 511 cells/mm3. The median follow up was
for 30 weeks. Six patients (15%) developed severe rash and were switched to EFV.
One patient discontinued due to hepatitis. Only 1 patient (2.5%) lost viral
control. Metabolic benefits were observed with 24 week results showing
triglycerides had declined 44% (p > 0.05), HDL-cholesterol increased 26% (p
> 0.001), and glucagon decreased 23% (p = 0.01). LDL cholesterol, glucose,
insulin and proinsulin did not alter significantly. Hip and spine bone mineral
density and truncal adiposity did not change.
A German study evaluated
42 patients with median time on PI-regimen of 24 months (range: 6-41) (mean age:
43�9.4) with VL <50 copies/ml and CD4+ >200 cells at baseline. Baseline
therapy switch included one PI in 23 patients (IDV: n = 13; NFV: n = 9; RTV: n =
1), and two PIs in 19 patients (RTV/IDV, n = 15; RTV/SQV, n = 1, NFV/SQV, n =
3). NRTIs were D4T/3TC(n = 29), or AZT/3TC (n = 13). VL was undetectable for a
median time of 12.5 months before substitution. Therapy was changed due to
adverse events to PI (n = 11), lipodystrophy (n = 20) and/or wish to simplify
regimen (n = 15). VL of all patients (39 with available data, 2 lost to follow
up) has remained <50 copies/ml and a significant reduction of CD8+/CD38+
cells from 66.9�17.1% to 60.5�18.1% at week 24. Non-fasting triglycerides
showed a significant decrease from 322�212 mg/dl at baseline to 252�206 mg/dl
at week 24, while values of HDL showed a significant improvement from 37�8
mg/dl to 43�9 mg/dl at week 24. Consistent with clinical trial data, 45.2% of
patients reported nervous system symptoms during the first weeks of therapy with
EFV.
An Italian study
specifically investigated the effect on switching in persons with elevated
triglycerides and viral load <500 copies/ml from PI to efavirenz. Patients
continued with the same NRTIs. Thirty-one patients receiving either IDV (14),
RTV (4), SQV (2), NFV (3) SQV+RTV (7) or SQV+NFV (1) for 19.8 +/- 8.2 months
were enrolled. Before PI treatment, their mean plasma triglyceride was 219 +/-
132 mg/dl. At switch this value was 718 +/- 453 mg/dl and was associated with
lipodystrophy in12 cases. Most patients showed reduction in triglycerides after
switch. After 1 month, tryglicerides had fallen to 362 +/- 233 mg/dl (P<
0.004), at 4 months 325 +/- 157 (P<0.008) and at month 8 359 +/- 149 mg/dl
(P> 0.03). Total cholesterol blood levels were unchanged. Mean HIV-RNA
dropped from 90 copies/ml to 49.3 copies/ml after one month. At 4 months all
patients were <50 copies/ml and at 8 months all but one patient was <50
copies/ml. CD4 T-cell levels increased from 525 cells/mm3 to 625 cells/mm3.
Treatment with efavirenz was generally well tolerated; only one patient
interrupted therapy because of dizziness.
A retrospective analysis
of 40 patients who had been switched for any reason from PI-containing
antiretrovital treatment (ART) to a regimen of two NRTI plus EFV reported data
suggesting lipid levels may not improve after switch to efavirenz but that viral
control is generally maintained after switching. All 11 patients in this cohort
who had a viral load <50 copies/ml prior to switch maintained control over an
observation period of 6 months. Median non-fasting cholesterol rose from 201 mg/dL
(range: 71-426) at baseline level to 218mg/dl (range of change �49 to +156)(p
= 0.01); and triglycerides from 221 mg/dL (range: 66-1957) to 238mg/dl (range of
change -433 to+853)(p = ns).
C. Katlama reported on
this French study of 165 randomized to continue their PI regimen or switch to
EFV (600 mg once daily). These were individuals who were likely to have been on
their first-line regimen upon entering study, although this was not mentioned in
the poster. They were on their PI regimen for about 2 years, were NNRTI na�ve,
and had 3 consecutive <50 copies/ml viral load measures before switching. So
this group was well suppressed virologically and was likely adherent.
The NRTIs were
maintained. This was a multi-center open-label study whose objective was to see
the effect of switching on the durability of viral suppression in individuals
who had viral suppression <50 copies/ml on a PI regimen. The mean duration of
PI treatment was 23.4 (�7.8) months in the EFV arm and 24.1 (�7.0) months in
the PI arm. There was a third arm in the study consisting of a substitution
regimen of EFV + PI(s) which was discontinued because of poor acceptance by
patients and investigators. Virologic failure was defined as confirmed on two
consecutive assays plasma viral load levels ≥50 copies/ml.
At baseline mean CD4s
were about 500 in both arms (range 113-1319). Over 90% of participants were
white. Over 80% were men. The PI(s) individuals were on in the EFV arm (n=69):
IDV 64%, NFV 26%, RTV 4%, RTV/IDV 2%, and RTV/SQV 2%. In the PI arm (n=65): IDV
60%, NFV 26%, RTV 12%, RTV/SQV (2%), and RTV/IDV (0).
At week 24 in the
ITT-analysis, 67/69 (97.1%) (n=69) in the efavirenz group had <50 copies/ml;
54/65 (83.1%) in the PI(s) arm had <50 copies/ml (statistically significant
difference between groups, p=0.0076). Using the Observed Data analysis, 67/68
(98.5%) in the EFV group had <50 copies/ml; in the PI(s) arm, 54/58 (93.1%)
had <50 copies/ml (no statistical significance, p>0.05). CD4 counts were
maintained in both groups --495 in EFV arm at week 24 and 554 in PI arm at week
24.
Regular cholesterol did
not improve after switching to EFV. The investigators compared cholesterol
changes from baseline between the 2 groups. In the EFV arm, mean baseline
cholesterol was 228 mg/dL (standard deviation 43), 244 (SD 41) at week 12, and
239 ( SD 45) at week 24. In the PI arm, mean baseline cholesterol was 210 (SD
45), 222 (SD 56) at week 12, and 220 (SD 51) at week 24. Increases in mean HDL
concentration were significantly greater at week 24 in the EFV arm: in the EFV
arm HDL (good cholesterol) was 45.7 mg/dL (SD 15.1) at baseline, 49.7 (SD 15.5)
at week 12, and 49.0 (SD 13.8) at week 24; in the PI arm, HDL was 42.1 at
baseline (SD 14), 43.2 (SD 15.6) at week 12, and 42.1 (SD 12.5) at week 24.
CNS
related adverse experiences reported in the EFV arm (there was statistical
significance between treatment groups for these adverse experiences): dizziness
(29%); insomnia (18.8%), 7.7% reported in PI arm; dreaming abnormally (23.2%);
vertigo (20.3%); depression (10.1%), 3.1% in PI arm and the difference in
depression was not statistically significant; concentration impairment (10.1%).
In a prospective,
multicentre, non-randomized study of 103 Spanish patients with a viral load
<200 copies/ml (86% <50) (mean 12 months, range 1-34) on a PI regimen
(mean 18 months range 4-35) switched to either efavirenz or nevirapine. Reasons
for NNRTI choice were not stated. Twenty three percent of patients were on their
first regimen. After 24 weeks, 93% of 33 efavirenz patients and 87% of 70
nevirapine patients remained <50 copies/ml in the as-treated analysis, and
78% and 75% in the intent-to-treat, respectively. Thirteen percent of patients
withdrew due to adverse events. Median CD4 count increased from 502 to 591
cells/mm3. Patients with a longer previous time on NRTI therapy (42 vs, 33
months), and a shorter time with undetectable HIV load (8 vs 12 months), had a
higher rate of viral failure (efavienz and nevirapine groups analysed together).
Adverse events secondary to NNRTI were observed in 27 patients (31%), mainly
hepatitis (9%) or rash (6%). Triglycerides and cholesterol levels significantly
improved after switching to the NNRTI. For triglycerides from 321 to 246mg/dl (p
= 0.01) and for cholesterol from 239 to 219mg/dl, p = 0.0001). Clinical
manifestations of lipoatrophy were not noted to improve.
Use of two of these agents intensifies the regimen. The safety, efficacy and impact on metabolic parameters of Abacavir (ABC) + Efavirenz (EFV) when substituted for a PI in persons with viral loads <50 c/ml was evaluated in an open label, single center study. To establish tolerability, ABC was added at baseline, then at week 6, EFV replaced the PI. Twenty-six patients have been followed-up for a mean 24 weeks. Four discontinued at week 2 (3 for ABC-hypersensitivity, 1 for virological failure due to non-compliance). No grade 3 or 4 laboratory toxicities were reported. After stopping PI, all patients have remained <50 with stable CD4 counts. Mean fasting-triglycerides, HDL and LDL cholesterol from the first 16 patients who reached week 24, improved significantly compared to baseline. Two of 4 diabetic patients saw resolution of diabetes. Five of 8 patients with lipodystrophy self reported improvements, 2 stabilization, and one worsening of signs.