 |
 |
 |
| |
NATAP REPORT OF HEPATITIS ABSTRACTS PRESENTED AT 52ND ANNUAL MEETING OF THE AMERICAN ASSOCIATION FOR THE STUDY OF LIVER DISEASES
Written for NATAP by Andrew Talal, MD, Cornell-Ny Hospital, NYC
|
| |
| |
I. Review of Hepatitis C Treatment Session 1
Sunday, November 11, 2001: 3:00-4:30 PM
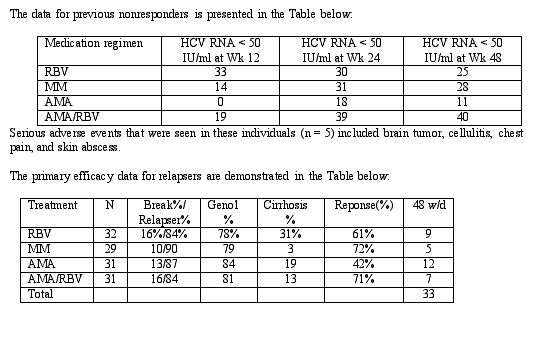
Abstract 277 Analysis of 40 kDA Peginterferon Alfa-2a (Pegasys) in combination with ribavirin, mycophenolate mofetil, amantidine, or amantidine plus ribavirin in patients that relapsed or did not respond to rebetron therapy: A report of two randomized, multicenter, efficacy and safety studies.
This study tested the hypothesis that the sustained virologic response to HCV may be increased if pegylated interferon is combined with other antiviral and immunomodulatory agents. This study evaluated the efficacy of treatment of pegylated interferon in combination with four different agents (ribavirin [1000-1200 mg/d], amantidine [100 mg BID], amantidine [100 mg BID]/ribavirin [1000-1200 mg/d], mycophenolate mofetil [1 gm PO BID]) in patients who had either relapsed or not responded to a previous course of at least 12 weeks of treatment with interferon and ribavirin. Nonresponders (NR) were defined as individuals who had received at least 12 weeks of combination therapy with cirrhosis excluded. Individuals who broke through were defined as those who had undetectable HCV RNA while on therapy who subsequently reverted to detectable HCV RNA while still on therapy. Relapsers (R) were defined as those with undetectable HCV RNA while on therapy who subsequently developed detectable HCV RNA after completing at least 6 months of therapy. Patients were treated for a total 48 wks. Main point of study was to evaluate the safety and efficacy of pegylated interferon alfa-2a combined with each of the four agents. This report shows ETR, 48-week preliminary response.
For this study, subjects were randomized to one of four treatment groups: 1) pegylated interferon alfa-2a combined with ribavirin 800-1000 mg/day (NR = 30, R = 32) 2) pegylated interferon alfa-2a combined with mycophenolate mofetil 1 g bid (MM) (NR = 29, R = 29) , 3) pegylated interferon alfa-2a combined with amantidine 100 mg bid (NR = 29, R = 31), 4) pegylated interferon alfa-2a combined with amantidine 100 mg bid and ribavirin 800-1000 mg/day (NR = 31, R = 31). The primary outcome was the number of individuals with 1) HCV RNA below assay detection (<50 IU/ml) after 48 weeks of therapy and 24 weeks of follow-up or 2) at least a 2 log decrease in HCV RNA from baseline).
|
|
| |
 |
|
| |
The implications of this study are that Peg-IFN alfa 2a combined with other antivirals the virologic response is significantly improved compared to standard interferon ~30% in all groups except with amantidine as a single agent. Additionally, the kinetics between prior nonresponders and individuals who have relapsed is different. In nonresponders, only in the group receiving PegIFN-2a/RBV was a response at Week 12 predictive of ETR, while in relapsers HCV RNA determinations at Wks 12, 24, 48 are predictive of a response.
Editorial note: it's anticipated that about 20% with ETR will relapse after stopping IFN/RBV. These data are ETR, and RBV prevents relapse and there appears to be no reason to assume AMA or MM will prevent relapse. It appears questionable these drugs (AMA, MM) will be useful.
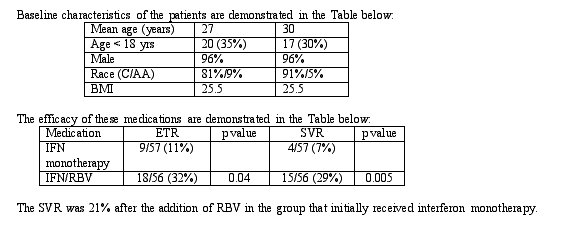
Abstract 278: Hepatitis C in adults and adolescents with inherited coagulation disorders in the United States: Results of a randomized, multicenter trial of combination therapy (interferon alfa-2B + ribavirin)
Prior to testing of the blood supply, transmission of hepatitis C to hemophiliacs and other individuals with coagulation disorders was very common. The goal of this study was to evaluate the effect of combination therapy with interferon and ribavirin in individuals with coagulation disorders. Between 1/97 and 1/00 113 HIV- individuals with detectable HCV RNA were randomized to receive 1) interferon alfa-2a (3 MU TIW) in combination with ribavirin (1000 mg/d) (n = 56) or 2) interferon alfa-2a (3 MU TIW) as a single agent (n =57). Individuals who received IFN monotherapy who had detectable HCV RNA after 12 weeks had ribavirin added to the regimen. Individuals who received combination therapy with HCV RNA below assay detection or > 1 log decrease were treated for a total of 48 weeks. Individuals were followed for an additional 24 weeks after treatment discontinuation. The primary goal of the study was the antiviral response to therapy in individuals with coagulation disorders. The secondary goal of this study was the safety and tolerability of anti-HCV therapy in this population.
|
|
| |
 |
|
| |
By multivariate analysis, the factors that were associated with a response were: treatment group (OR = 9.8, p = 0.002), non type1genotype (OR 9.0, p = 0.002), and adolescent (OR = 10.6, p < 0.001). One interesting finding from this study is that adolescents had a significantly increased response rate compared to that in adults, as shown in the Table below:
|
|
| |
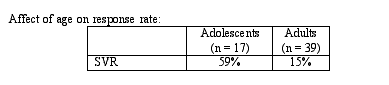 |
|
| |
In terms of the secondary goal of this study (safety and tolerability of therapy) 20% of the individuals withdrew.
The implications of this study are that the SVR among individuals with coagulation disorders is significantly lower than compared to historical controls in the general population. However, consistent with the findings of previous studies, the SVR in individuals who receive combination therapy is significant increased compared to those who receive interferon monotherapy. At least among individuals with coagulation disorders, the SVR among adolescents significantly increased compared to adults and adolescents had a significantly lower HCV RNA at baseline. Delayed administration of ribavirin change response rate to that similar to response in individuals treated with combination therapy from the initiation of the study. In this study, there was no association between SVR and BMI among adolescents.
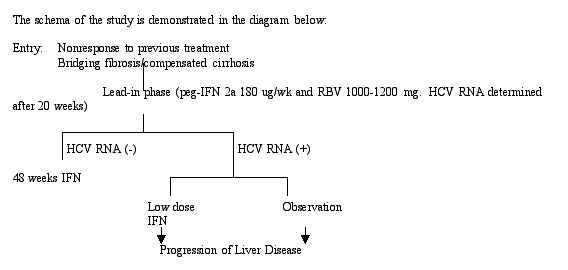
Abstact 279: Retreatment of interferon and interferon-ribavirin non-responders with peginterferon-alpha-2a and ribavirin: Initial results from the lead-in phase of the HALT-C trial.
Treatment options for individuals with cirrhosis or individuals who did not respond to anti-HCV therapy have been limited to IFN "maintenance therapy" with the goal to reduce the incidence of hepatic decompensation and hepatocellular carcinoma (HCC). The National Institutes of Health is conducting a four-year study (HALT-C) to evaluate the efficacy of pegylated interferon to decrease the incidence of decompensation and HCC in chronically HCV-infected individuals. This study reported on the results of the "lead in-phase" in which all participants (who were nonresponders to a previous course of treatment with combination therapy with interferon and ribavirin) were retreated with pegylated interferon and ribavirin. The goals of the lead in-phase were to evaluate whether the SVR would increase in previous nonresponders. Target enrollment is 1350 patients. This preliminary report is the week 20 response for the first 268 patients enrolled. There were 15 dropouts so 253 were assessed. 107 have HCV-RNA undetectable.
|
|
| |
 |
|
| |
The preliminary results reported at 20 weeks included an overall on-treatment response of 42%, a response rate of 53% in those individuals who were previously treated with interferon monotherapy, a response rate of 10% in African Americans, a response rate of 43% in other races, and a response rate by genotype 1/2/3of 28%/50%/80%. 53% of individuals who had received previous treatment with interferon monotherapy responded compared with 31% of those treated with IFN/RBV combination therapy.
In terms of safety and tolerability of the regimen, dose reduction was required in 59% and discontinuation was required in 5.5%.
Response by genotype: 1a, 30%; 1b, 39%; 2a, 35%; 2b, 88%; 3a, 93%.
5.5% dropped out of study before week 2 (mostly due to side effects), and 59% required dose reduction to IFN or RBV or both.
Factors associated with improved response included bridging fibrosis. Patients with cirrhosis did not respond as well (44% vs 31%, p=0.03). Factors not associated with virologic response included elevated baseline HCV RNA, BMI, and gender.
(Editorial note: 31% on treatment response appears encouraging; SVR has yet to be determined and study outcome will take several years to reach).
Abstract 280: Treatment with 40kDA peginterferon alfa-2A (Pegasys) in combination with ribavirin significantly enhances quality of life compared with interferon alfa-2b plus ribavirin.
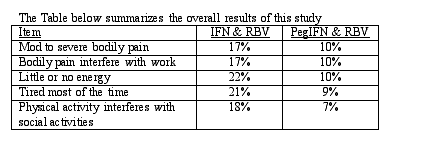
Quality of life (QOL) is a very important measure of how a person feels when they are being treated for hepatitis C. The objectives of this study were to evaluate the QOL in patients undergoing treatment with three different types of medications: 1) Peg IFN alfa 2a 180 ug and ribavirin 1000-1200 mg, 2) IFN alfa 2b 3 MIU/tiw and RBV 1000-1200 mg, 3) Peg IFN alfa 2a 180 ug and placebo. After 24 weeks of therapy, QOL and fatigue were determined using the SF36 Health Survey and the fatigue stability scale (FSS). (Both of these are questionnaires that are completed by patients in which they indicate how they feel, how the illness or the medication has affected their functioning, and how the illness has impacted on their daily lives). These were used to determine the impact of medication on daily living and the degree of fatigue was assessed by visual analogue scale (a line in which patients circle how they feel).
In this study, subjects were randomized to receive the medication combinations listed above and they were treated for 72 weeks. During this study, these patients were asked to assess their quality of life at baseline, and at weeks 2, 12, 24, 48, and 72. Subjects indicated that QOL components including body pain (p = 0.002), vitality (p = 0.02), and social functioning (p = 0.04) and burden of fatigue (p = 0.02) were significantly improved in those who received Pegasys intereron compared to those who received standard IFN. Overall, the physical components of the SF36 were significantly improved (p = 0.05) in those who were treated with Pegasys interferon while there was no difference in those items that constitute the mental components. Degree of fatigue (p = 0.01) and impact of fatigue (p = 0.05) were also significantly different in those who were treated with Pegasys interferon compared to those who received standard interferon.
|
|
| |
 |
|
| |
The implications of this study are that patients who are being treated with Pegasys interferon and ribavirin have significantly less fatigue and the impact on daily living is significantly lower in those individuals treated with Pegasys interferon. The difference between the regimens that contain Pegasys interferon and standard interferon was detectable two weeks after medication initiation. The patients who were treated with Pegasys interferon had less intense or disabling pain, decreased fatigue and less interaction with quality of life and daily functioning. Therefore in addition to increased therapeutic efficacy, Pegasys interferon appears to be better tolerated than standard interferon. These findings could affect medication compliance and adherence to therapy, thereby partially accounting for the improved efficacy observed with Pegasys interferon.
Abstract 281: Durability of sustained virologic response in patients with chronic hepatitis C after treatment with interferon-a 2B alone or in combination with ribavirin
SVR to standard interferon therapy 40-60%. The goal of this study is to assess the durability of SVR. For this study, data from four separate treatment protocols was aggregated. Subjects chronically infected with HCV were treated with interferon alfa-2b plus ribavirin or interferon alfa-2b monotherapy for either 24 or 48 weeks. Of 2089 patients from 3 trials, 455 receiving IFN/RBV and 103 receiving IFN had an SVR. 395 with an SVR agreed to be followed for a mean of 41/2 years., 322 treated with combination therapy and 73 treated with interferon. Two-thirds of the subjects are male, 70% with mild disease, 59% of the subjects were followed for 4 yrs, 26% followed for 3 yrs. 37% were genotype 1, 26% genotype 2 and 34% genotype 3.
Results: Out of 395 subjects, 10 individuals relapsed (7 of those treated with combination therapy and 3 treated with interferon monotherapy) after 24 weeks of follow-up. All relapses occurred within first 21/2 years after medication discontinuation. A total of five patients had detectable intrahepatic HCV RNA despite negative serum HCV RNA at the time of biopsy 6 months after medication discontinuation. Two out of five had subsequently relapsed suggesting that detectable hepatic HCV RNA six months after medication termination may be a risk factor for late relapse. 95% of the relapsers had normal ALT. One of the patients who relapsed developed hepatocellular carcinoma.
The conclusions of this study are that only 2.5% of patients relapse after 6 mos. after therapy discontinuation. Relapse occurs in the first two years, so two years after medication discontinuation if an individual does not have detectable HCV RNA in plasma, they are extremely unlikely to relapse. Presence of detectable HCV RNA in liver may be a risk factor for recurrence.
Abstract 282: Impact of pegylated interferon alfa-2B and ribavirin on progression of liver fibrosis.
Introduction: Anti-HCV treatment has two distinct aims: 1) to achieve a virologic response thereby permitting fibrosis regression and 2) in those individuals who do not achieve a virologic response, to prevent progression of fibrosis and development of hepatocellular carcinoma. The goal of this study is to assess the affect of different therapeutic combinations and their affect on fibrosis regression.
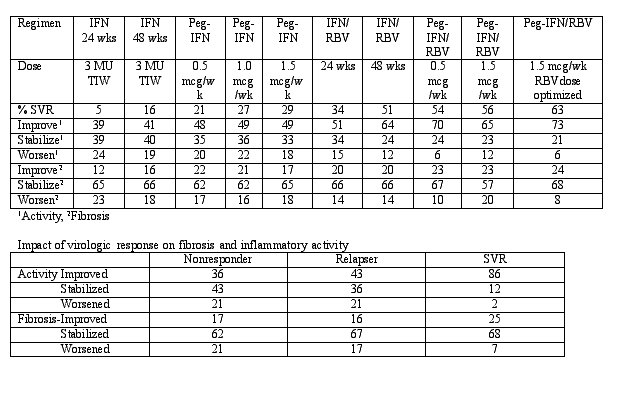
Methods: For this study, a database containing 4,493 patients who were enrolled in 4 trials studying 10 different regimens. A total of 3010 patients with paired biopsies were evaluated by one pathologist. 69% of the individuals were genotype 1, 28% genotype 2-3, 3% genotype (4,5,6). The distribution of fibrosis among these patients was F0= 2%, F1= 73%, F2= 13%, F3= 7%, F4= 5% and inflammation A0=2%, A1=21%, A2= 39%, A3= 38%. Sixty-six percent of the sample was male, the mean age was 43 years, mean wt was 80 kg, mean duration of infection was 18 years, mean duration between biopsies 20 months, and 16 mm was mean size of the biopsy.
Results: The percent SVR in individuals treated with different therapeutic combinations is demonstrated in the table below:
|
|
| |
 |
|
| |
These results demonstrate that there is a very strong correlation between virologic response and fibrosis stabilization and an improvement in inflammatory activity.
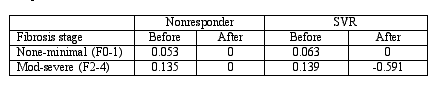
The Table below shows the fibrosis progression rate (units/yr) stratified by degree of fibrosis and histologic response:
|
|
| |
 |
|
| |
As is demonstrated in the Table, there is a very strong correlation between a virologic response and a histologic response. Even in those individuals who fail to achieve an SVR, therapy can slow the rate of fibrosis. In those individuals with moderate to severe fibrosis who achieve an SVR, fibrosis may revert at a very high rate.
|
|
| |
 |
|
| |
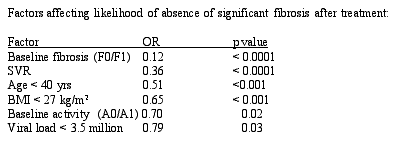
Reversal of cirrhosis in HCV infection: Among 153 subjects who had cirrhosis on the first biopsy, 75 subjects had reversal of cirrhosis on second biopsy. There was no significant difference between those patients who had reversal and those who did not in terms of body weight, size of the biopsy, the time at which the follow-up biopsy was performed, or the duration between the biopsies. However, factors that were significantly different between those who reversed and those who did not included: 1) whether the medication regimen was administered for 24 or 48 weeks (100% vs. 69%), 2) whether the individuals achieved an SVR (33% vs. 15%, p = 0.02), 3) the age of the individual (44.7 vs. 47.2 [with younger individuals more likely to have cirrhosis reversal]) and 4) whether there was an improvement in the inflammatory response as determined by HAI score.
Conclusions/Commentary: For many years, we have believed that cirrhosis was not reversible and the dogma has been to treat patients early to avoid the development of cirrhosis. Recently, in many different entities that can all result in cirrhosis, we have learned that cirrhosis may be reversible. Some of the most convincing data has been published in individuals who had biliary cirrhosis in which a blockage of the bile duct leads to cirrhosis. In a large number of individuals in whom the blockage is relieved, the cirrhosis can reverse. These investigators have now demonstrated the same phenomenon in HCV. This is a very important study because it challenges our thinking regarding the reversibility of cirrhosis in HCV-infected individuals and shows that it can regress in 75/153 patients.
|
|
| |
|
 |
 |
|
|