| |
|
Selection and fading of resistance mutations in women and infants receiving nevirapine to prevent HIV-1 vertical transmission (HIVNET 012)
|
| |
| |
Susan H. Eshleman; Martin Mracna; Laura A. Guay; Martina Deseyvea; Shawn Cunningham; Mark Mirochnickb; Philippa Musokec; Thomas Flemingd; Mary Glenn Fowlere; Lynne M. Mofensonf; Francis Mmirog; J. Brooks Jackson
From the Department of Pathology, The Johns Hopkins Medical Institutions, Baltimore, Maryland, the aFred Hutchinson Cancer Research Center, Seattle, Washington, the Department of Pediatrics, Boston University, Boston, Massachusetts, USA, the cDepartment of Paediatrics, Makerere University, Kampala, Uganda, the dDepartment of Biostatistics, University of Washington, Seattle, Washington, the eDivision of AIDS, NIAID/NIH, Rockville, the fPediatric, Adolescent, and Maternal AIDS Branch, NICHD/NIH, Rockville, Maryland, USA, and the gDepartment of Obstetrics and Gynaecology, Makerere University, Kampala, Uganda.
AIDS 2001;15:1951-1957
Objective: To examine the emergence and fading of NVP resistance (NVPR) mutations in HIV-1-infected Ugandan women and infants who received single dose NVP to prevent HIV-1 vertical transmission.
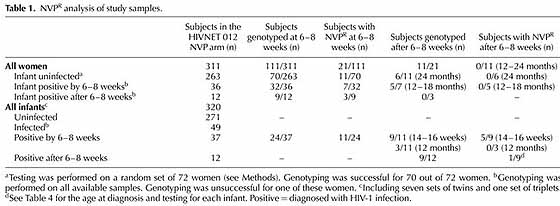
Design: We examined NVPR in women and infants who received NVP in the HIVNET 012 clinical trial, including 41 out of 48 women with infected infants, 70 randomly-selected women with uninfected infants, and 33 out of 49 infected infants.
Methods: Plasma HIV-1 was analyzed using the Applied Biosystems ViroSeq HIV-1 Genotyping System.
Results: NVPR mutations were detected in 21 out of 111 (19%) women tested 6-8 weeks after delivery. The rate of NVPR was similar among women whose infants were or were not HIV-1 infected. K103N was the most common mutation
detected. NVPR mutations faded from detection within 12-24 months in all 11 evaluable women. High baseline viral load and low baseline CD4 cell count were associated with development of NVPR. NVPR mutations were detected in 11 out of 24 (46%) evaluable infants who were infected by 6-8 weeks of age. The most common NVPR mutation detected in infants was Y181C. Those mutations faded from detection by 12 months of age in all seven evaluable infants. Of nine evaluable infants with late HIV-1 infection, only one had evidence of NVPR.
Conclusions: NVPR was detected more frequently in infants than women following NVP prophylaxis, and different patterns of NVPR mutations were detected in women versus infants. NVPR was detected infrequently in infants with late HIV-1 infection. NVP-resistant HIV-1 faded from detection in women and infants over time.
Introduction
Nevirapine (NVP) is a potent non-nucleoside inhibitor of HIV-1 reverse transcriptase (RT). In the HIVNET 012 clinical trial, administration of a single 200 mg dose of NVP to pregnant Ugandan women at the onset of labor and a single 2 mg/kg dose to their infants within 72 h of birth was shown to significantly reduce the rate of HIV-1 mother-to-child transmission (MTCT) [1,2]. That regimen was more effective than a short course of zidovudine prophylaxis starting in labor, and was simpler and much less expensive. The efficacy, simplicity and low cost of the HIVNET 012 NVP regimen make it attractive for use in developing countries. The World Health Organization recently recommended implementation of this regimen as one of several options for prevention of MTCT in resource-limited settings [3].
When NVP is administered as monotherapy for treatment of HIV-1 disease, rapid selection of HIV-1 variants with nevirapine resistance (NVPR) mutations occurs [4]. We recently reported that NVP-resistant HIV-1 can also be selected in women who receive single dose NVP prophylaxis to prevent HIV-1 MTCT [5]. In that report, we analyzed HIV-1 from 15 women who were enrolled in the Ugandan Phase I/II trial, HIVNET 006 [6]. Women in HIVNET 006 received the same NVP prophylaxis regimen as women in HIVNET 012. In that cohort, the K103N NVPR mutation was detected in HIV-1 from three out of 15 (20%) women 6-8 weeks after delivery. Our analysis of women in HIVNET 006 further suggested that a more prolonged exposure to NVP favors selection of NVP-resistant HIV-1 [5]. In this report, we extend our previous studies by examining the emergence and fading from detection of NVP-resistant HIV-1 in a large cohort of Ugandan women and infants who received NVP prophylaxis in the HIVNET 012 trial.
| |
| |
 |
|
| |
NVPR in late-infected infants and their mothers (breastfeeding)
In HIVNET 012, 98% of women breastfed their infants. Because of the potential for HIV-1 transmission by breastfeeding, infants who tested negative for HIV-1 infection at 6-8 weeks of age were followed for 18 months for evidence of late
HIV-1 infection. Nine infants were diagnosed with HIV-1 at later time points. In addition, three infants who were not tested at 6-8 weeks of age, but who tested negative at birth, were diagnosed with HIV-1 at later times . The median age of HIV-1 diagnosis in those 12 infants was 301 days (10 months; range, 77-550 days). Samples collected 6-8 weeks after delivery were available from nine out of 12 of the mothers of the late-infected infants. NVPR mutations were detected in three of those women (Table 4). Samples collected from those women at later time points were not available for analysis.
Logistic regression analysis revealed an association between baseline viral load and development of NVPR mutations and between baseline CD4 cell count and resistance. In a univariate model, women with high viral loads were more likely to
develop resistance (per increase of one log10 HIV-1 RNA: odds ratio, 3.97; 95% confidence interval, 1.54-10.20; P = 0.0042). Similarly, women with low CD4 cell counts were more likely to develop resistance (per decrease of 100 cells:
odds ratio, 1.63; 95% confidence interval, 1.20-2.21; P = 0.0016).
Discussion
We detected NVP-resistant HIV-1 6-8 weeks after delivery in 19% of 111 women tested who received single dose NVP in HIVNET 012. Several factors may have contributed to the selection of NVP-resistant HIV-1 in this setting. These include
the high potency and long half-life of NVP in pregnant women during labor (median t1/2 = 61.3 h) [6,8], the ability of a single mutation (K103N or Y181C) to cause high level NVPR, and the probability that minor HIV-1 variants with those
mutations are likely to be present at low background levels in most infected women prior to NVP administration [4,9]. We found that the K103N mutation was selected more frequently than Y181C in women following single dose NVP. This is consistent with early detection of K103N in patients receiving chronic NVP therapy [10]. Early emergence of variants with K103N may reflect a fitness advantage of those variants compared to variants with Y181C. With increased NVP exposure, Y181C variants with higher levels of phenotypic NVPR would be expected to emerge.
Frequent emergence of NVPR among women in HIVNET 012 may in part reflect their advanced stage of HIV-1 disease. At study entry, women analyzed in this report had relatively high viral loads (median, 40 067 copies/ml) and relatively low CD4 cell counts (median, 412 cells/l). Both of those factors were associated with development of NVPR in this cohort. Therefore, the rate of NVPR following single dose NVP prophylaxis may be lower in cohorts with less advanced HIV-1 disease. In the USA, NVP prophylaxis is one of the recommended options for prevention of HIV-1 MTCT in women in labor who have not received antiretroviral therapy during pregnancy [11]. Initiation of fully suppressive, highly active antiretroviral therapy in such women during the immediate postnatal period would probably reduce the risk that NVP-resistant HIV-1 would emerge.
In the HIVNET 012 cohort, NVP-resistant HIV-1 faded from detection in women over time. HIV-1 variants with NVPR mutations may continue to circulate in these women as minor variants, and be maintained as provirus in infected cells.
However, replacement of the major HIV-1 population with NVP-sensitive HIV-1 makes it less likely that NVP-resistant HIV-1 would be transmitted from women to other adults. Furthermore, our inability to detect NVP-resistant HIV-1 12-24
months after delivery suggests that the single dose NVP prophylaxis regimen would remain effective for interruption of intrapartum transmission in subsequent pregnancies. This is because most of the HIV-1 population would be sensitive to
NVP at the time of labor, when NVP is administered, and would be effectively inhibited during labor and delivery, when HIV-1 transmission is most likely to occur. This requires confirmation. In contrast, persistence of minor variants or
proviruses with NVPR mutations could potentially limit the use of NVP or other non-nucleoside RT inhibitors for subsequent treatment of HIV-1 infection. In developing countries, where the NVP prophylactic regimen is most likely to be
implemented, current treatment options for HIV-1 infection are extremely limited. If treatment options in those countries were widely expanded in the future, women with NVP-resistant HIV-1 could be offered alternative treatment regimens with other antiretroviral drugs.
We found no association between post-partum selection of NVP-resistant HIV-1 in women and the risk of MTCT with the first use of NVP prophylaxis. We also found little evidence for transmission of NVP-resistant HIV-1 variants from women to infants who were infected by 6-8 weeks of age. The risk of transmission of NVP-resistant HIV-1 variants through breastfeeding requires further evaluation. The kinetics of the emergence and fading of NVP-resistant HIV-1 in women receiving single dose NVP has not been defined, and the duration NVP-resistant HIV-1 persists in the mother will affect the
risk of late postnatal transmission of resistant virus. Of the nine late-infected infants in this study, only three had mothers with NVPR detected at 6-8 weeks postpartum. Of those, two infants were infected with NVP-sensitive HIV-1 and one was infected with NVP-resistant HIV-1. Additionally, 11 women with NVP-resistant HIV-1 had uninfected infants and did not transmit HIV-1 despite breastfeeding. However, because evaluation of NVPR in infants in HIVNET 012 was possible only using follow-up samples collected months after the diagnosis of HIV-1 infection for most infants, it is possible that additional NVP-resistant strains were transmitted to infants by breastfeeding, but that those variants faded from detection before follow-up samples were collected.
Our data suggest that NVP-resistant HIV-1 is selected independently in infants after NVP administration. The relatively high rate of NVPR observed among infants in HIVNET 012 could reflect the high viral loads typically present in HIV-1
infected infants. Increased exposure to NVP in infants compared to their mothers may also favor emergence of NVP-resistant HIV-1. Infants in HIVNET 012 were essentially dosed twice: once by the placental transfer of maternally
administered NVP, and once within 48-72 h of birth. The half-life of NVP in infants is long (median t1/2 = 46.5 h) [6,8]. When pregnant women and infants received the same regimen used in HIVNET 012, the NVP concentration in infants was > 100 ng/ml (> 10 times the 50% inhibitory concentration of the drug) for 7 days [6,8]. The greater NVP exposure of infants versus women in HIVNET 012 may also explain the more frequent detection of the Y181C mutation (rather than K103N) in infants. The fading of NVP-resistant HIV-1 that we observed in infants paralleled that observed in women post-partum, and was consistent with reduced fitness of HIV-1 with NVPR mutations in the absence of the drug.
The findings of this report emphasize the importance of evaluating the development of drug resistance among women receiving short-course antiretroviral prophylaxis regimens, particularly for prophylaxis regimens using antiretroviral drugs in which a single mutation can confer resistance, such as NVP or lamivudine. Such evaluations should include: (i) further
characterization of the kinetics of the emergence and fading of NVPR in women receiving single dose NVP prophylaxis; (ii) assessment of whether there is a risk for transmission of NVP-resistant virus to infants postnatally through breastfeeding or to sexual partners and the magnitude of that risk; and (iii) assessment of the association of NVPR with disease progression.
Such monitoring should be planned within future perinatal trials, as well as within the context of implementation efforts. Epidemiologic studies could also be considered to evaluate the effectiveness of the single dose NVP regimen in future pregnancies.
The potential for selection of NVP-resistant HIV-1 in women and infants receiving single dose NVP prophylaxis must be balanced against the documented efficacy, simplicity, and cost-effectiveness of the HIVNET 012 regimen. This regimen
can significantly reduce HIV-1 MTCT in settings where other prophylactic regimens are impractical and treatment options are extremely limited. If implemented rapidly, this regimen can prevent HIV-1 infection in millions of HIV-1 exposed infants over the next decade.
|
|
| |
|
|
|