A prospective study of discontinuing primary and secondary Pneumocystis carinii pneumonia prophylaxis after CD4 cell count increase to > 200
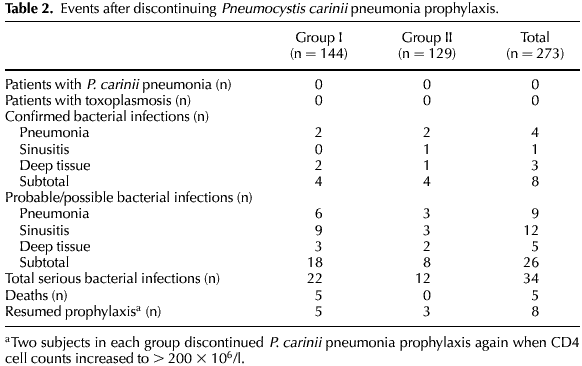
The study authors conclude what other previously reported studies have concluded‹that PCP prophylaxis appears to be safe to discontinue when CD4s increase to a certain level above 200 and remain there for a period of time. PCP becomes a risk when CD4s are below 200 and therefore PCP prophylaxis treatment is used. The investigators looked at both primary and secondary prophylaxis. Primary is when a patient never had PCP before, and secondary is when they had a previous case of PCP. No cases of PCP developed in either group. The authors reported a total of 34 serious bacterial infections were diagnosed, 22 in patients receiving primary prophylaxis and and 12 in group receiving secondary prophylaxis, the majority involving either the upper or lower respiratory tract (Table 2). The authors concluded, however, the number of confirmed serious bacterial infections and new AIDS-defining illnesses during the course of this study was low.
Susan L. Koletar; Alison E. Healda; Dianne Finkelsteinb; Richard Hafnerc; Judith S. Currierd; J. Allen McCutchane; Marc Valleeb; Francesca J. Torrianie; William G. Powderlyf; Robert J. Fass; Robert L. Murphyg; for the ACTG 888 Study Team*
From the The Ohio State University Hospitals, Columbus, Ohio, aDuke University Medical Center, Durham, North Carolina, bHarvard School of Public Health, Boston, Massachusetts, cNational Institutes of Health, Bethesda, Maryland, dUniversity of California, Los Angeles, eUniversity of California, San Diego, California, fWashington University School of Medicine, St. Louis, Missouri and gNorthwestern University Medical School, Chicago, Illinois, USA.
AIDS 2001;15:1509-1515
ABSTRACT:
Objective: To assess the incidence of Pneumocystis carinii pneumonia (PCP) after discontinuation of either primary or secondary prophylaxis.
Design: This was a prospective, non-randomized, non-blinded study.
Setting: Twenty-five University-based AIDS Clinical Trials Group units.
Participants: Participants either had a CD4 cell count 100 at any time in the past and no history of confirmed PCP (group I; n = 144), or had a confirmed episode of PCP 6 months prior to study entry (group II; n = 129). All subjects had sustained CD4 cell counts > 200 in response to antiretroviral therapy.
Interventions: Subjects discontinued PCP prophylaxis within 3 months or at the time of study entry. Evaluations for symptoms of PCP and CD4 cell counts were performed every 8 weeks. Prophylaxis was resumed if two consecutive CD4 cell counts were < 200.
Main outcome measure(s): The main outcome was development of PCP.
Results: No cases of PCP occurred in 144 subjects (median follow-up, 82 weeks) in group I or in the 129 subjects (median follow-up, 63 weeks) in group II (95% upper confidence limits on the rates of 1.3 per 100 person-years and 1.96 per 100 person-years for groups I and II, respectively). Eight subjects (five in group I and three in group II) resumed PCP prophylaxis after two consecutive CD4 cell counts < 200.
Conclusions: The risk of developing initial or recurrent PCP after discontinuing prophylaxis is low in HIV-infected individuals who have sustained CD4 cell count increases in response to antiretroviral therapy. Neither lifelong primary nor secondary PCP prophylaxis is necessary.
This study was designed to assess prospectively the impact of discontinuing either primary or secondary PCP prophylaxis in patients with sustained CD4 cell count increases in response to antiretroviral therapy.
The primary objective of this prospective, non-randomized trial was to assess the incidence of histologically confirmed PCP in HIV-infected individuals who dis- continued PCP prophylaxis following sustained in- creases in CD4 cell counts induced by antiretroviral therapy. The incidences of presumed PCP and other opportunistic and serious bacterial infections were also assessed. Subjects aged at least 13 years with confirmed HIV infection were recruited at 25 AIDS Clinical Trials Group (ACTG) units. The protocol was reviewed and approved by the Institutional Review Board at each site and all patients gave written informed consent prior to participation. Initial subjects enrolled had a history of a CD4 cell count < 100 any time in the past, no confirmed history of PCP and had been receiving primary PCP prophylaxis (group I). One year after the study had been opened to accrual, the protocol was amended to allow inclusion of subjects with a history of histologically or cytologically confirmed PCP more than 6 months prior to study entry who had been receiving secondary PCP prophylaxis (group II). All subjects had CD4 cell counts . 200 on two measurements at least 12 weeks apart before study entry. Subjects with signs or symptoms compatible with PCP, or who required chronic treatment with any drug known to have anti-PCP activity or had received immunomodulatory therapy within 60 days were not eligible for study entry.
Conventional PCP prophylaxis was to be resumed if subjects had a decline in CD4 cell counts to < 200 3 10 6 /l at two time-points within a 4 week period, or if they developed oral thrush or unexplained fever of more than 2 weeks duration. Subjects continued the same evaluation schedule after re-initiation of prophylaxis. Follow-up of both groups is ongoing. The median CD4 cell count at the time of study entry was 360 for both groups. There were 133 subjects (49% ) known to have had a CD4 cell count, 50 3 10 6 /l prior to study entry; the majority of these (68%) were in group I. The median time on antiretroviral therapy before study entry was 132 weeks for group I and 115 weeks for group II. Stored plasma for HIV RNA has not been analyzed, but viral load data was available for 72 subjects (26% of the total group) who were co-enrolled in other ACTG studies. At baseline, 67% of these subjects had plasma HIV-1 RNA levels , 500 copies/ml (68% in group I, and 64% in group II). Prophylactic regimens prior to were similar between the two groups with trimethoprim±sulfamethoxazole being the most common. At least one prior AIDS-defining illness was in 60 subjects (42%) in group I with cytomegalovirus (CMV) disease, esophageal candidiasis and Kaposi's sarcoma being most common. In addition to PCP, patients in group II had at least one AIDS-defining illness. CMV disease, Kaposi's sarcoma, and Candida esophagitis were also the most common other AIDS-related illnesses in this group.
The median duration of follow-up was 82 weeks (range, 64±135 weeks) for group I and 63 weeks (range, 24±87 weeks) for group II. Twelve subjects (8.3%) in group I and eight subjects (6.2%) in group II have prematurely discontinued the study; median follow-up for these subjects was 50.5 weeks and 31.4 weeks for group I and II, respectively. Reasons for early termination included patients' refusal of additional follow-up (seven subjects in group I and four in group II) or inability to continue follow-up for non-medical reasons (five subjects in group I and four in group II).
There have been no confirmed or presumed cases of PCP in the 144 subjects in group I (with a total of 231 person-years of follow-up) and the 129 subjects in group II (with 153 person-years of follow-up), yielding 95% upper confidence limits on the rates of 1.3 cases per 100 person-years and 1.96 cases per 100 person- years in groups I and II, respectively. A total of 34 serious bacterial infections were diagnosed, 22 in group I and 12 in group II, the majority involving either the upper or lower respiratory tract (Table 2). One case of disseminated Mycobacterium avium complex (MAC) disease was reported in group I; at the time of diagnosis (study week 56), the CD4 cell count of this subject was 654 3 10 6 /l. No other new AIDS-defining illnesses, including toxoplasmosis, have been reported. There have been five deaths, all occurring in group I; causes of death included pancreatitis, renal failure, acute myocardial infarction, lung cancer and intracranial hemorrhage.
This prospective study both corroborates and adds to growing body of literature supporting the safety of discontinuing primary PCP prophylaxis. A recent literature review by Kovacs and Masur [27] show that the incidence of primary PCP is exceedingly rare in responders to antiretroviral therapy. The majority of these studies included subjects with median nadir CD4 cell counts . 100 and shorter durations of follow-up. In contrast, subjects discontinuing primary PCP prophylaxis in our study all had nadir CD4 cell counts , 100 3 10 6 /l and the median duration of follow-up was comparable to that in the study published recently by Lopez Bernaldo de Quiros et al. Rigorous evaluation by our study team of any possible endpoints also adds to the assurance that none developed either confirmed or probable PCP.
Stopping secondary prophylaxis has not been studied as extensivel because of uncertainty about continued risk of PCP recurrence despite quantitative increases in CD4 cell counts. In addition, early concerns focused on higher rates of both PCP and death among patients who had a history of PCP noted in studies evaluating different prophylactic regimens. Such concerns have been at least partially alleviated by studies of immune reconstitution. Significant increases in naive (CD45RA) CD4 cells have been observed as soon as 12 weeks and more consistently at 6 months after initiating highly active antiretroviral therapy. Clinical studies reporting successful discontinuation of maintenance therapy for MAC and for CMV also support the phenomen- on of immune reconstitution. Our study design took account of the observations of time to naive cell recovery by requiring at least 12 weeks of CD4 cell counts consistently > 200 3 10 6 /l. This was felt to be particularly important considering the fact that all subjects enrolled in this group had histologically-proven PCP in the past. This inclusion criteria is unique to our study and further substantiates the safety of discontinuing secondary PCP prophylaxis demonstrated by two recent reports in which no cases of PCP were noted. As with group I, evaluations of possible study endpoints were rigorous and median duration of follow-up is approximately 3 months longer than other reported studies.
While the study results strongly support discontinuation of secondary prophylaxis, it is important to note that they can only be applied to patients who had PCP at least 6 months prior to stopping prophylaxis. Also, until more data is available, patients with declines in CD4 cell counts to , 200 3 10 6 /l should resume PCP prophylaxis.
One limitation of this report is the absence of complete viral load data for all subjects. This could be particularly important as Williams et al. demonstrated that HIV-1 RNA levels influenced the risk of opportunistic infections independently of the CD4 cell count.
A recognized secondary benefit of some PCP prophylaxis regimens has been the prevention of other infections, notably toxoplasmosis and bacterial respiratory tract infections. In a study by Currier et al. significantly fewer subjects (38%) receiving trimetho-prim-sulfamethoxazole as PCP prophylaxis had one or more reported bacterial infection compared with those (51%) not receiving this agent. Although this was one of the additional arguments against discontinuing PCP prophylaxis, such benefit was not demonstrated clearly in this study as the number of confirmed serious bacterial infections and new AIDS-defining illnesses during the course of this study was low, presumably due to immune reconstitution. Other advantages of stopping prophylaxis include decreased pill burden, less potential side-effects and drug interactions, cost, and decreased likelihood of developing resistance among typical bacteria as well as among P. carinii.
Current guidelines for the prevention of PCP address the issue of discontinuing PCP prophylaxis in the context of the limited information that was avail- able at the time of their publication. The cautious recommendations are directed primarily at discontinuing primary prophylaxis, acknowledging the limited data regarding secondary prophylaxis. Our study provides additional evidence supporting the low probabil ity of developing PCP in patients who have CD4 cell count increases sustained at > 200 for at least 12 weeks, regardless of prior history of PCP. This study, in conjunction with other recent studies, supports the concept that primary PCP prophylaxis is not necessary in patients responding to potent antiretroviral therapy. In addition, our results add to the growing body of evidence that lifelong secondary PCP prophylaxis is also unnecessary.
