|
NATAP REPORTS |
Current Review & Update on Hepatitis C & HIV/HCV Cection |
SUMMER 2001 |
|
Treatment of HCV in Special Populations
|
|
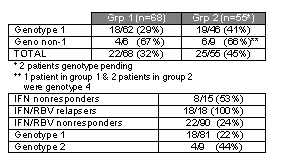
Hard To Treat Populations: African-Americans, genotype 1 & 4, cirrhotics, nonresponders & relapsers Hard to treat populations are also seeing improved response rates with pegylated interferon. In an analysis of African-American’s response to standard interferon alfa-2b plus ribavirin in a large study conducted by Schering-Plough, John McHutchison has reported that African-Americans responded as well as Caucasians with genotype 1 (25% in Caucasians vs 22% in African-Americans). Some experts have questioned this equivalence and some studies have shown less response by African-Americans, while anecdotal reports have reported a decreased response to therapy by African-Americans. A large, NIH-funded study will address the reasons why African-Americans manifest decreased responsiveness to treatment of HCV. Some experts feel that there may be genetic differences in the immune response. It appears that adherence is just as important in taking HCV medications as it is in HIV. Cirrhotics saw a very nice 30% response rate to Pegasys monotherapy in a study that was reported by Heathcote in 2000. Preliminary data reported by Ira Jacobson (Cornell) at DDW in 2001 showed that 30% of previous non-responders to standard interferon plus ribavirin with genotype 1 were PCR negative at week 24 with pegylated interferon plus ribavirin therapy. Study results using both pegylated interferons show African-Americans improve the response rate over use of standard interferon. Following PEG-Intron monotherapy treatment: 14% of African-Americans/Blacks had SVR and none who received standard interferon achieved any benefit. Analysis of data from the Pegasys monotherapy data base (55 African-American patients) noted that 15% of African-Americans/Blacks achieved SVR to this monotherapy as compared to 34% of Caucasians. None of the African-American patients who received standard interferon monotherapy had achieved SVR. In terms of ALT, 19% of African-Americans achieved normal ALT compared to 39% of Caucasians. Only 7% of African-Americans achieved normal ALT using standard IFN. In terms of a histologic response, 33% of African-Americans had a decrease of 2 points in HAI score compared to 61% for Caucasians, no doubt due to an improved SVR rate. Only 28% of African-Americans using standard IFN achieved a histologic response. In terms of hepatic fibrosis progression, several studies suggest that HCV disease may progress more slowly in HCV monoinfected patients compared to that in HIV/HCV coinfected patients. Study research also suggest HCV may progress more slowly in African-Americans with HCV alone, but this has not been studied in coinfected African-Americans, nor are we well acquainted with the clinical significance of this suggested slower progression. Cirrhotic patients are also considered to be difficult to treat. Thirty percent of cirrhotic patients achieved a virologic response to Pegasys 180 ug given once a week for 48 weeks, which was comparable to the 35% response rates in non-cirrhotics. These data are encouraging for difficult to treat African-American/Black patients, cirrhotic patients, and those with genotypes 1 and 4. Traditionally, genotype 4 patients also have been difficult to treat, and relatively few individuals achieve SVR. Although quite infrequent, occasionally genotype 4 is encountered in the United States. In a subset analysis from the Pegasys database, 45% of genotype 4 patients achieved SVR. It was discussed earlier in this report how the response to pegylated interferon+ribavirin appears to have increased substantially for genotype 1, from the 29% seen in the large registrational study of IFN/RBV to 45% in the pegylated + ribavirin studies. Interestingly, the SVR for individuals with genotype 2 have not improved as dramatically in response to pegylated interferon and ribavirin as those in individuals with genotype 1 infection. Pegylated Interferon + Ribavirin in Previous Non-responders Preliminary data from several studies presented at DDW and recently in other venues show promising results for patients who previously had viral failure from standard IFN+RBV, particularly for previous nonresponders. The first dataset comes from Ira Jacobson (Weill Medical College of Cornell University). Three groups of patients are being studied (n=330): IFN monotherapy nonresponders, combination relapsers, and combination nonresponders. Patients receive a PegIntron+RBV regimen for 48 weeks: (group 1, n=68) PegIntron 1.0 ug/kg + RBV 1000 mg/day (if <75 kg) or 1200 mg/day (if >75 kg), or (group 2, n=57) PegIntron 1.5 ug/kg + RBV 800 mg/day. Therapy is discontinued if patients have detectable HCV RNA as determined by PCR at 24 weeks. At baseline, the average fibrosis stage was 1.8-1.9, ALTs were 93-104 IU/L, HCV RNA ranged from 1.1 x 106 to 1.6 x 106 copies/ml, genotype 1 was present in 81-91%. One hundred and twenty-three patients were reported to have reached week 24 of treatment. Subjects who are treatment
week 24 PCR Negative PCR Negative at week 24:
treatment response to PEG IFN/RBV by Nonresponders (NR) and Relapsers. In summary, Jacobson found that in genotype 1 nonresponders the 24 week treatment response is higher with 1.5 ug/kg of peg interferon (30% vs 17%). Preliminary results suggest very high response rates in combination therapy relapsers with either dose of PEG. In this group of patients, PEG IFN a-2b and RBV is associated with a similar side effect profile as combination therapy. No definitive conclusions can be reached until a larger number of patients have been studied and data on SVR become available. Peg Intron/RBV for IFN or IFN/RBV Failures: 2 additional studies at DDW Dr Gaglio (DDW) from New Orleans reported on 254 patients receiving Peg IFN a-2b 1.5 ug/kg + RBV 800 mg for 48 weeks in study for IFN or IFN/RBV failures. One hundred thirty two patients were treated for 24 weeks and virologic response (PCR negative) was seen in: 16/75 (21%) of nonresponders 9/17 (53%) in partial responders 22/29 (76%) in relapsers 6/8 (75%) in breakthrough relapsers Dose reduction was required in about 20% of patients due to anemia (39%), leukopenia (35%), thrombocytopenia (19%), and dyspnea (6%). Medication discontinuation occurred in 3%. On the basis of these results, Dr. Gaglio concluded therapy was relatively well tolerated. Peg IFN/RBV in IFN/RBV Failures The second abstract included over 200 IFN/RBV nonresponders and relapsers from 11 midwest cities who were randomized to receive PEG IFN a-2b 0.5 ug/kg + RBV 800 mg/day or Peg IFN a-2b 1.5 ug/kg + RBV 800 mg/day. One hundred and two patients have completed 24 out of 48 weeks of therapy. Of 66 nonresponders: --3% (1/34) who received PegIntron 0.5 ug/kg+ RBV 800 mg are PCR negative. --28% (9/32) who received 1.5 ug/kg + RBV 800 mg are PCR negative Of 40 relapsers: --52% (11/21) receiving lower dose IFN regimen are PCR negative --68% (13/19) receiving PegIntron 1.5 ug/kg are PCR negative Pegasys + RBV in IFN and IFN/RBV Failures Preliminary data was reported (AASLD, June 2001) from the HALT-C study (which is a four year maintenance therapy study) by Adrian Di Bisceglie. At week 20, 40% (59/146) of patients who had previously not responded to interferon or IFN-ribavirin had negative HCV RNA. At DDW, Nezam Afdhal presented data from a small study that previous non-responders to combination therapy could respond well to Pegasys: HCV RNA is negative at week 24 in 9/30 (30%) and ALT is normal in 16/30 (53%) at week 24 for those receiving Pegasys + ribavirin. Nezam Afdahl (Beth Israel Deaconness Medical Center, Boston) reported at DDW on 30 non-cirrhotic patients who did not respond previously to IFN/RBV and who received Pegasys + RBV, as part of a study in which other patients received amantadine or mycophenolate acid. The patients were treated for 48 weeks, and preliminary results were reported. The mean age of the 30 patients who received Pegasys+RBV was 45 years, the mean weight was 90 kg; 70% were male and 30% were female; the mean ALT was 113 IU/L, 60% had high HCV RNA, 90% were genotype 1, and none of the patients had cirrhosis. In this study, patients who received at least one dose of study medication and who subsequently discontinued therapy are considered nonresponders. Afdahl reported that no unexpected adverse events were seen. The results of this study are:--at week 24, 14/30 (46%) HCV RNA declined by at least 2 log, --at week 12, 10/30 had negative PCR and --at week 24 9/30 (30%) had negative PCR; --14/30 had normal ALT at week 4, and 16/30 (53%) had normal ALT at week 24. Preliminary data from Steven Herrine (Thomas Jefferson University Hospital; abstract 1966) was reported at DDW. In this study patients also received Pegasys/RBV or amantadine and mycophenolate acid. The patients were viral breakthroughs or relapsers. A viral breakthrough is when HCV-RNA is reduced while on therapy but rebounds while still on therapy. A relapse is when HCV-RNA is undetectable at the end of treatment but rebounds after stopping therapy. Eighty-four percent were relapsers, 78% were infected with genotype 1; 56% had a high viral, 75% were male, the average weight was 92 kg, and the average age was 48 years. At week 24, 28/32 (87%) had a reduction in HCV RNA of at least 2 log, and 22/32 (69%) had HCV RNA below assay detection (21/32 had negative PCR by week 12). The average baseline ALT was 117 IU/L, and at week 4, 15/32 had normal ALT, and at week 24 17/32 had normal ALT. 18 months therapy for hard to treat patients: cirrhosis; high viral load, genotype 1 This study suggests that 18 months of therapy may improve the response rate for hard-to-treat populations. At the 2001 EASL Conference, an update was reported on the Benelux Study of treatment for 18 months compared to 6 months. 300 patients without previous treatment received standard IFN plus RBV 1000-1200 mg/day. Hard to treat populations received the greatest benefit: patients with genotype 1, cirrhosis, and high baseline viral load, in the as treated analysis. After 18 months treatment the SVR was 43% (ITT) compared to 34% treated (ITT) for 6 months. The relapse rate was 13% in 18 month-treatment group vs 38% in 6 month group. In the group receiving IFN alone for 18 months the SVR was 16% (ITT) and relapse rate was 39%. Using as-treated analysis for patients who were 80% adherent for 80% of the intended duration of therapy, patients with cirrhosis receiving 18 months IFN/RBV had a higher SVR of 57% (as-treated analysis) compared to 42% without cirrhosis. For patients receiving 6 months treatment the opposite occurred, as expected: those with cirrhosis had SVR of 29% compared to 37% without cirrhosis (as-treated analysis). Patients with genotype 1 receiving 18 months therapy had higher SVR than those taking 6 months treatment (36% vs 23%). But, those with genotype 2 or 3 had the same SVR (71-72%) regardless of the duration of therapy. Patients with high HCV RNA at baseline (> 3 million copies/ml) and 18 months of treament had better SVR than those who received 18 months of treatment with HCV RNA < 3 million copies/ml (42% vs 18%). Individuals with HCV RNA < 3 million copies/ml had the same SVR (47%-49%) whether they received 18 or 6 months of therapy (as-treated analysis). |
| < All newsletters |