 |
 |
 |
| |
Predictive Value of Early Virologic Response
Reported by Jules Levin
|
| |
| |
The published results of Pegasys plus ribavirin phase III study were published in the New England Journal of Medicine (2002; 347:975-982). This article was published around Sept 27, 2002 and a summary of the data in this study and the published article can be read on the NATAP website at http://www.natap.org/2002/september/092702_1.htm.
In this study Fried reported on the "Predictive Value of Early Virologic Response". Here are the results from this study. By week 12, 86 percent of patients (390 of 453) treated with peginterferon alfa-2a plus ribavirin had a virologic response, defined as a 2-log decrease from baseline HCV RNA levels (97 patients) or no detectable serum HCV RNA (293 patients). The absence of an early virologic response was not associated with early treatment discontinuation (before week 12) or dose modification (data not shown). Of those with early virologic responses, 65 percent subsequently had a sustained virologic response. Those with no detectable HCV RNA by week 12 were more likely to have a sustained virologic response than those who had only a 2-log decrease in HCV RNA (221 of 293 vs. 32 of 97). In contrast, among the 63 patients who did not have an early virologic response, 61 (97 percent) did not have a sustained virologic response.
At AASLD researchers reported interim data from a study confirming the previously reported marked association between early viral response and pegylated interferon a-2a (Pegasys) plus ribavirin (reported above), even in genotype 1 patients. (Brandao et al including Roche, abstract 805). There are 193 patients in this study. Patients received Pegasys 180 mcg per week plus 800 mg ribavirin for either 24 or 48 weeks. Viral load tests used in study: Cobas Roche Amplicor Monitor HCV test v 2.0 with sensitivity of 50 IU/ml and Amplicor Monitor test 2.0 with sensitivity of 600 IU/ml, at weeks 4, 12, 24, 36, and 48 of the treatment period and at weeks 12 and 24 of the treatment free period.
|
|
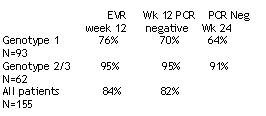 |
| |
The predictability of these early response data on SVR will be evaluated as additional data accumulates.
Similar study results have been reported for the use of PegIntron plus ribavirin. At the Schering Plough symposium at AASLD (Nov 1-5, 2002) in Boston, researchers reviewed some of this data. Gary Davis reviewed data has prepared for publication (Hepatology 2002; 36, Suppl 2, In press).
Here is Davis' verbatim report which I feel is worth reading. Interferon based regimens are highly effective for treatment of chronic hepatitis C (HCV), with the current standard regimen of pegylated interferon and oral ribavirin eradicating virus in more than half of cases. (edit note: Response rates vary depending on genotype and viral load before starting therapy). There are challenges associated with this regimen, however, in that the regimen is complicated, uses two drugs administered by two routes, lasts 6-12 months, requires frequent monitoring, usually causes side effects, and is expensive. Thus, determination of response early during the treatment course is desirable, thereby providing the option of discontinuing treatment in patients who would not respond.
HCV Kinetics Following Interferon
HCV replication generates 1010 to 1012 viral particles per day that have a half-life of only a few hours. Pegylated interferon inhibits HCV replication by about 90%, resulting in a rapid decline in HCV RNA. This first phase of viral decline, however, is not associated with treatment response. In contrast, the slow, prolonged, and more variable second phase of viral decay correlates very closely with sustained virologic response to interferon based treatment regimens. This close association suggests that measurement of HCV RNA during the first several weeks of therapy might predict eradication of HCV.
Edit note: there is viral kinetics data suggesting you can predict SVR by week viral load response. I think it is not advisable to discontinue thyerapy if no response is seen by week 4. Discontinuing therapy before week 12 may prevent a number of patients from the opportunity to achieve sustained virologic response (SVR) or histologic benefit. Some patients are late responders. Preliminary anecdotal clinical reports suggest HIV/HCV coinfected patients may respond later. The data on EVR predicting SVR are from studies in HCV monoinfected patients. Although there is preliminary data suggesting the 12 week cutoff concept can be applied for coinfected patients, two such small studies were presented at ICAAC 2002 by Soriani, I think it way premature to use this approach for HCV/HIV coinfected patients. There are anecdotal reports and data from studies showing that coinfected patients may be late responders. Around a 2 log drop in viral load by week is a good indication of whether a patient will reach undetectable viral load 6 months after stopping therapy in all patients including coinfection, but unless there is very little response in viral load by week 12, I suggest continuing to week 24. In fact, despite the data, I think that unless there is no viral response at all, the doctor and patient should consider continuing past 12 weeks. Some patients may respond later. Continue to evaluate and consider continuing until 6 months if response shows after 12 weeks.
Davis reported findings from Zeuzem (Gastrenterology, 2001; 128: 1438). Phase 1 lasts 2-3 days. Phase 2 lasts from day 3 to 2 weeks. Following phase 2 decay using standrad interferon, patients with no viral response during phase 2, 0/5 had an end-of-treatment response (ETR) and 0/5 had an SVR. For patients who were slow responders, 3/5 had an ETR and 1/5 had an SVR. For patients who were rapid responders 4/5 had an ETR and 3/6 had an SVR. Responses improved using pegylated interferon. For patients with no response during phase 2 4/8 had an ETR and 0/8 had an SVR. For slow responders 6/6 had an ETR and 2/6 had an SVR. For rapid responders during phase 2, 3/3 had an ETR and 3/3 had an SVR.
Early Virologic Response
Early studies of standard interferon monotherapy showed that failure to lose HCV RNA after 12 weeks of treatment, as measured by qualitative PCR testing, was strongly associated with treatment failure in both 6 and 12 month regimens. Davis et al studied the association between early response and treatment failure in data from the 511 sublects treated with pegylated interferon alfa-2b (PegIntron) 1.5 ug/kg once weekly plus ribavirin 800 mg/day in the phase 3 clinical trial by Manns et al. The definition of early virologic response (EVR) that optimized capture of potential responders (highest sensitivity), while excluding the largest proportion of nonresponders (highest negative predictive value) was sought. Quantitative HCV RNA levels were measured at 4, 12, and 24 weeks of treatment and compared with the baseline level. The best definition of EVR was a drop of at least two logs in HCV RNA from baseline or undetectable HCV RNA after 12 weeks of treatment. 74% (379/511) of patients achieved this. If treatment had been discontinued in the remaining 26% (132/511) of patients, only 0.8% (1/132) of all sustained viral responders would have been lost by premature termination of treatment. Furthermore, more than 99% (131/132) of those who failed to reach EVR were nonresponders 24 weeks after completing a full course of treatment. Looked at another way, among those who did not achieve EVR after 12 weeks, the chance of reaching SVR following completion of the full course of treatment was less than 1%.
Response rates were lower among patients who remained PCR positive at 12 weeks despite a 2-log drop (21% vs 84%; p<0.001)) and those who were unable to remain adherent to the prescribed dose (61% to 63% vs 75% to 77%).
Early Virologic Response
(Davis, GL, Hepatology, 2002; 36, Suppl 2, In press)
|
|
 |
| |
Subgroup Analysis
A 2 log or greater decrease in HCV RNA level or undetectable HCV RNA at week 12 was the optimal definition of EVR regardless of viral genotype, although almost all patients with genotypes 2 and 3 achieved EVR (95% vs 66% for genotype 1). Patients who had genotype 1, high HCV RNA levels, low serum ALT levels, and poor adherence in the first 3 months were less likely to achieve EVR.
The 2 log or greater drop in HCV RNA or undetectable HCV RNA at week 12 of treatment was also the optimal definition of EVR for patients who received ribavirin doses >10.6 mg/kg/day (the equivalent of an 800 mg dose in a 75-kg individual) and patients who received standard interferon and ribavirin.
Recommendations
Based on these data, patients who do not achieve EVR are not likely to achieve SVR, and therefore should discontinue therapy. Patients woth EVR who are still virus positive should be retested at 24 weeks. If virus is still present at that time, treatment should be stopped. EVR is so common in patients infecte with genotype 2 and 3 that for these patients, HCV RNA testing during therapy may not be cost effective. Consideration of these recommendations, however, requires recognition of the limitation of viral quantification and the potential, yet unproven, benefits to histology and inflammation in nonresponders.
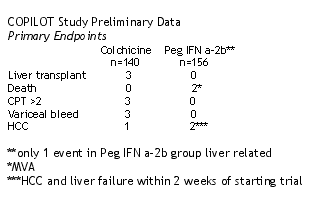
(edit note: preliminary data presented at AASLD from COLPILOT, which looks at whether maintenance therapy in nonresponders will slow progression, shows a hint that maintenance therapy can yield clinical benefit. The COLPIOT study and the NIH HALCT-C study are looking at this question and ongoing. I presume additional data will be presented at conferences in the next year. Here is the very early data reported at AASLS from COLPILOT.
|
|
 |
| |
Conclusions
Measuring EVR is helpful in the management of chronic hepatitis C in patients who receive treatment with standard or pegylated interferon plus ribavirin. It can provide patients and treating physicuans with an early goal and motivate them to adhere to treatment recommendations. Discontinuation of therapy in those who fail to achieve EVR reduces treatment costs and avoids the morbidity of therapy in those with no or little chance of clearance. Utilization of quantitative testing to access EVR has recently been recommended by the National Institutes of Heath Consensus Development Conference Statement, "Management of Hepatitis C: 2002", which can be read on the NATAP website.
|
|
| |
|
 |
 |
|
|