 |
 |
 |
| |
EPOETIN ALFA (PROCRIT) ONCE WEEKLY MAINTAINS RIBAVIRIN DOSE IN
HEPATITIS C VIRUS (HCV)-INFECTED PATIENTS TREATED WITH COMBINATION THERAPY:
interim results
Reported by Jules Levin
|
| |
| |
After 8 weeks on EPO, patients were on average taking 940 mg/day of ribavirin vs patients not receiving EPO who were taking on average 857 mg/day of ribavirin. This data finds that patients were able to maintain ribavirin dose rather than decreasing dose due to anemia. Study investigators reported that patients taking EPO reported improved quality of life as measured by physical, mental, vitality, and mental health parameters. And hemoglobin increased from 11g/day to 13 in the patients taking EPO after 8 weeks, patients not receiving EPO had hemoglobin levels of 10.6 g/day. This addresses development of anemia. These benefits may improve adherence and sustained responses. Further studies are warranted to confirm these findings and to further explore effectiveness of EPO during interferon plus ribavirin therapy for HCV. 83% of patients receiving EPO reported at least 1 AE vs 72% in the patients not receiving EPO. Although no serious adverse events occurred in the 48 patients during the 8-week double blind phase, at the time of abstract submission to AASLD, 3 serious adverse events had occurred in 3 patients in the EPO group: death due to cirrhosis and pneumococcal sepsis in 1 patient (investigator said this was unrelated to study drug); autoimmune hemolytic anemia in 1 patient (investigator said unrelated to study drug) and syncopal episode in 1 patient (AE relationship to study drug considered doubtful), both adverse events occurred in the open-label phase following 8-week double-blind study.
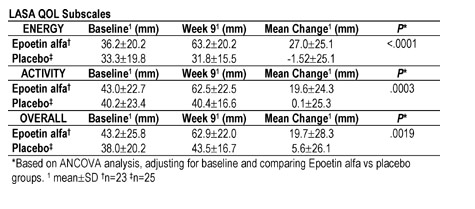
Background: Data from clinical studies in HCV-infected patients treated with ribavirin/interferon alfa (RBV/IFN) or RBV/pegylated IFN (PEG-IFN) suggest that 1000/1200 mg/day RBV enhances rates of sustained virologic response (Hadziyannis et al., EASL. 2002; Manns et al., Lancet. 2001; McHutchison et al., Hepatology. 2000). However, combination therapy is associated with several side effects that result in dose reduction or premature treatment discontinuation. 54% of RBV/IFN-treated patients experience hemoglobin (Hb) drops of 3 g/dL; 31% of treated patients experience Hb drops of 4 g/dL. Comparatively, RBV dose reduction to 600 mg/day effects a mean Hb increase of 1.1 g/dL (Sulkowski et al., AASLD. 2000). In a proof-of-principle, randomized, open-label, multicenter study, once-weekly (QW) 40,000 IU Epoetin alfa (PROCRIT®) was effective in maintaining RBV dosing, increasing Hb levels, and improving the quality of life (QOL) of anemic (Hb£12 g/dL) HCV-infected patients treated with RBV/IFN (Sulkowski et al., STC-AASLD. 2001). Interim results from the follow-up, placebo-controlled, double-blind phase of a larger trial (N=180) using Epoetin alfa in anemic HCV-infected patients treated with either RBV/IFN or RBV/PEG-IFN, are presented. Methods: This study has two treatment phases: an 8-week double-blind phase (DBP) followed by an open-label phase (8 weeks for efficacy parameters). Anemic HCV-infected patients (Hb£12 g/dL) anticipated to receive at least 16 wks of RBV/IFN or RBV/PEG-IFN therapy are randomized to treatment with 40,000-60,000 IU SC of Epoetin alfa or placebo QW during the DBP. After completion of that phase, randomization will be unblinded, and eligible patients will receive QW Epoetin alfa during the open-label phase. The primary efficacy endpoint, assessed at the end of the DBP, is maintenance of RBV dosing. Secondary efficacy endpoints are Hb levels, QOL self-assessments (Linear Analog Scale Assessment (LASA) and Short Form-36.v2), and transfusion utilization. Results: The interim analysis included 48 patients evaluable for the DBP. Epoetin alfa (n=23) and placebo (n=25) groups were similar at baseline (BL): mean age=49 yrs, 65% women, mean Hb=11.1 g/dL (Epoetin alfa), 10.8 g/dL (placebo). Prior to HCV therapy, mean Hb was 14.0 g/dL in both groups. At Wk 9, maintenance of RBV dose from BL was achieved in 22/23 (95.7%) of Epoetin alfa-treated patients, compared with 15/24 (62.5%) of placebo-treated patients (P<.010). The mean Hb at Wk 9 vs BL increased significantly (P<.0001) in the Epoetin alfa group (2.11±1.00 g/dL) but remained relatively unchanged in the placebo group (-0.20±1.00 g/dL). Similarly, LASA QOL subscores (Energy, Activity, Overall) at Wk 9 vs BL increased significantly in Epoetin alfa-treated patients but remained unchanged in the placebo group (Table). Adverse events (AEs) were reported in about 77% of patients in both groups and were similar to those associated with IFN or PEG-IFN RBV therapy. A total of 3 serious AEs occurred in 3 patients receiving Epoetin alfa: relationship to Epoetin alfa was deemed by respective Investigators to be doubtful (syncopal episode) and unrelated (death in patient with cirrhosis and pneumococcal sepsis, autoimmune hemolytic anemia). Conclusion: Based on these interim results, QW Epoetin alfa administration to HCV-infected patients treated with either RBV/PEG-IFN or RBV/IFN maintains RBV dose by correcting HCV therapy-induced anemia and significantly improving patient QOL.
|
|
 |
| |
|
 |
 |
|
|