| |
Once-Daily HAART: Toward a New Treatment Paradigm
Ian Frank
University of Pennsylvania, Philadelphia, Pennsylvania
|
| |
| |
JAIDS Journal of Acquired Immune Deficiency Syndromes 2002; 31:S10-S15
Highly active antiretroviral therapy (HAART) suppresses HIV replication to undetectable levels with concomitant increases in CD4+ T-cell counts and improvement in immune function. However, complex dosing, large pill burdens,
and side effects make long-term adherence difficult, with the result that patients may achieve only suboptimal exposure to antiretroviral drugs, increasing the risk of treatment failure and viral resistance. Treatment strategies in other chronic conditions show that simpler regimens with fewer daily doses and fewer pills per dose increase adherence and treatment
success. This article discusses adherence and its influence on treatment outcomes, reviewing evidence from recent studies that have evaluated the safety and efficacy of treatment regimens with reduced pill burdens and improved tolerability.
THE IMPORTANCE OF ADHERENCE
Findings from multiple studies utilizing a variety of methodologic approaches have indicated that in the treatment of HIV infection, suboptimal adherence is a strong predictor of treatment failure. A study that documented the link between treatment adherence and successful outcomes compared adherence rates in a group of prisoners receiving antiretroviral therapy as part of the AIDS Clinical Trials Group studies and a group of patients participating in the same studies at the University of Miami (1). The 50 prisoners in this study received directly observed therapy (DOT), whereas 50 patients cared for in an outpatient clinic received self-administered treatment (SAT). At baseline, the DOT patients had lower CD4+ T-cell counts (246 vs. 329 cells/mm3) and higher HIV RNA levels (5.6 vs. 5.3 log10 copies/mL), but more course of the study after 80 weeks of therapy (p < .01). All of the patients were receiving multidrug regimens involving three or four drugs, including two nucleoside reverse transcriptase inhibitors (NRTIs) and either efavirenz or a protease inhibitor (PI), or both. Within the SAT group, the response rate was lowest among the patients taking a four-drug regimen, suggesting that simpler
regimens are more likely to improve adherence and, thus, response to treatment.
In another study, in which adherence was measured by electronic monitoring, Paterson et al. compared the proportions of subjects achieving an undetectable viral load based upon the proportion of doses taken. Adherence rates
correlated with virologic success (p < .001) and higher CD4+ T-cell counts (p = .006). In this study of 81 patients receiving PI-containing three-drug combinations, only 5 of 23 patients (22%) with a greater than 95% adherence rate failed to maintain HIV RNA levels <400 copies/mL over a median of 6 months of follow-up. The risk of treatment failure increased with decreasing adherence. Treatment failed in 61% of patients with adherence rates of 80% to
95% and in 80% of patients with less than 80% adherence. Neither physicians nor nurses were able to consistently identify which patients would have difficulty adhering to treatment regimens, misjudging the rate of adherence in 41% and 30% of their patients, respectively.
Adherence tends to decline over time. This was shown in two multicenter, randomized trials in which patients completed 7-day recall adherence reports at 1 month, 4 months, and 8 months of follow-up. At 1 month, 70% of patients
reported 100% adherence, but this proportion declined to 63% at 4 months and to 58% at 8 months (p < .05). Elevations in HIV RNA and decreases in CD4+ T-cell counts correlated with the decline in adherence at each time point,
indicating that treatment fatigue can have a marked effect on long-term efficacy.
FACTORS AFFECTING ADHERENCE
Factors that are associated with poor adherence rates include active drug or alcohol abuse, active mental illness (particularly depression), medication side effects, and inconvenient treatment regimens. Race, gender, disease
stage, or a history of substance abuse do not seem to be strongly correlated with a decrease in adherence or with treatment fatigue. Predictors of good adherence include confidence in the effectiveness of HAART medications,
availability of social support, trust in caregivers, understanding of the importance of adherence and the consequences of the lack of it, and consistency in keeping clinic appointments. Strategies to improve adherence are listed in Table 2.
Table 2. Strategies to enhance adherence
| |
 |
| |
Establish readiness to begin therapy
Negotiate a treatment plan agreed to by doctor and patient
Reduce the number of pills and dosing frequency
Explain the goals of therapy and the importance of adherence
Inform patient of side effects and how they will be treated
Avoid adverse drug reactions
Provide a written schedule of medications and pictures of pills
Utilize support groups and community based organizations to stress the importance of adherence
Pill burden, defined as both pill count and regimen complexity (i.e., dosing frequency and food requirements), is an important contributor to adherence problems. The most significant regimen-related strategy to improve adherence may be reduction in the number and frequency of pills to be taken. A meta-analysis of 23 clinical trials evaluated the relationship between pill burden and the durability of virologic response. Each trial lasted at least 24 weeks and enrolled 30 or more patients who were treatment naive or who had previously received only limited antiretroviral therapy. All trials used three-drug regimens: two NRTIs combined with a PI, a nonnucleoside reverse transcriptase inhibitor (NNRTI), or a third NRTI. Multivariate analysis of the trial results indicated that the antiretroviral regimens did not differ in efficacy,
and neither baseline viral load nor CD4+ T-cell counts were significantly associated with outcomes at 48 weeks. However, increased pill count was negatively associated with maintenance of undetectable viral loads at week 48 and seemed to be the most significant predictor of response to treatment.
A similar negative correlation between regimen complexity and both adherence and treatment outcome was observed in an open-label study comparing Combivir®/abacavir with Combivir/indinavir therapy in 342 treatment-naive patients. (Combivir is a combination tablet containing zidovudine and lamivudine.) After 24 weeks, a greater percentage of patients receiving the simpler Combivir/abacavir regimen than those receiving Combivir/indinavir had achieved HIV RNA levels <400 and <50 copies/mL. In addition, fewer patients receiving Combivir/abacavir than those receiving Combivir/indinavir reported difficulty taking their medication as scheduled (7% vs. 31%) or reported missing more than one dose per week or any dose within the last 4 weeks (22% vs. 52%).
RATIONALE FOR ONCE-DAILY DOSING
The evidence that patients adhere better to once-daily regimens than to twice-daily or more frequent dosing schedules has come from therapeutic areas other than HIV, for which true once-daily regimens are still in development. Dezii et al. studied adherence retrospectively in patients receiving glipizide for diabetes and in those receiving venlafaxine for the treatment of depression/anxiety. Short- and longer-term adherence was evaluated. As determined by filled prescriptions, patients receiving glipizide once daily were more adherent than those receiving the same medication twice daily at both 6
and 12 months of follow-up (13% and 20% greater adherence, respectively). Similarly, patients receiving venlafaxine once daily were also more adherent than those receiving it twice daily (14% greater adherence at 6 months, 11% greater at 12 months).
CLINICAL SUPPORT FOR ONCE-DAILY HAART
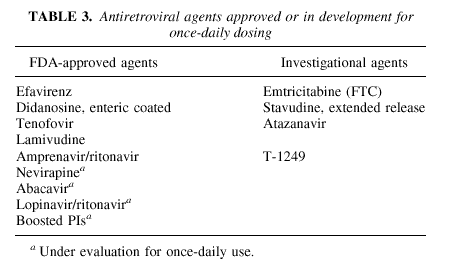
Table 3 lists antiretroviral agents that are available or are being studied as once-daily medications. Currently, four antiretroviral drugs are approved by the U.S. Food and Drug Administration (FDA) for once-daily dosing: efavirenz,
enteric-coated didanosine, tenofovir, and lamivudine. In addition, the combination of amprenavir and ritonavir is FDA approved for once-daily dosing. Both efavirenz and tenofovir were designed for once-daily administration. (At the time of this presentation, recommended efavirenz dosing was 3 ? 200-mg capsules once daily. Since then, a 600-mg tablet has been approved by the FDA that permits dosing of one tablet once daily.) Enteric-coated didanosine represents a new formulation of didanosine, which was previously approved as a buffered tablet for twice-daily dosing. The enteric-coated formulation of didanosine offers improved gastrointestinal tolerability, less frequent dosing, lower pill burden (one capsule vs. up to four tablets), and fewer buffer-related drug interactions.
A randomized, open-label, multicenter study of 511 treatment-naive subjects showed that a triple-drug regimen consisting of once-daily, enteric-coated didanosine plus stavudine and nelfinavir provided antiviral activity similar to that of a reference triple-drug regimen consisting of Combivir plus nelfinavir . The subjects in each group achieved similar median decreases in HIV RNA levels at 48 weeks: -2.59 log10 copies/mL for those in the enteric-coated didanosine group and -2.61 log10 copies/mL for those in the reference group. Similarly, a comparable percentage of patients in each group achieved HIV RNA levels <400 copies/mL at 48 weeks: 56% for those receiving enteric-coated didanosine and 53% for those receiving the reference regimen. Increases in CD4+ T-cell counts were also similar: 120 cells/mm3 and 162 cells/mm3 for the enteric-coated didanosine and the reference regimens, respectively. Discontinuations because of adverse events were comparable in both treatment groups. Nausea was more frequent in the reference arm (36% vs. 24% for the enteric-coated didanosine arm), and peripheral neurologic symptoms were more common in the enteric-coated didanosine arm (20% vs. 11% for the reference arm).
Tenofovir is a nucleotide reverse transcriptase inhibitor, the first of this class of HIV drugs. Tenofovir has been compared with placebo in a study of highly treatment-experienced subjects who were randomly assigned to receive
tenofovir 75, 150, or 300 mg once daily, or placebo, in addition to their stable antiretroviral regimen. Subjects were followed for 48 weeks. All placebo patients were switched to tenofovir 300 mg after their first 24 weeks of treatment. Viral load reductions in the tenofovir treatment group were maintained throughout the course of the study. Patients switched from placebo to tenofovir achieved a viral load reduction at 48 weeks comparable to that seen in patients who had received tenofovir from the beginning.
Trials with other established agents, including but not limited to lamivudine, abacavir, and extended-release stavudine, also indicate the potential of once-daily dosing with these agents. In a prospective open-label trial designed to evaluate the utility of once-daily lamivudine, subjects were randomized to receive either lamivudine 150 mg twice daily or 300 mg once daily along with stavudine and a PI (indinavir or nelfinavir). Viral suppression was maintained in both treatment arms by the 78 subjects who completed the 24-week study.
A pilot study of 151 subjects tested the activity of lamivudine and abacavir as once-daily agents. In this study, a reference regimen consisting of zidovudine 300 mg twice daily, lamivudine 150 mg twice daily, and abacavir 300 mg
twice daily was compared with two other treatment schedules over 48 weeks of treatment. The comparator arms consisted of zidovudine 300 mg twice daily/lamivudine 300 mg once daily/abacavir 300 mg twice daily and zidovudine 300 mg twice daily/lamivudine 150 mg twice daily/abacavir 600 mg once daily. Both lamivudine and abacavir given once daily as part of this triple-drug combination therapy maintained viral suppression with a level of tolerability that was comparable to that of the reference treatment.
An extended-release formulation of stavudine is under evaluation for use as a once-daily medication. In a randomized, double-blind study, subjects received either extended-release stavudine 100 mg once daily or the standard,
immediate-release formulation 40 mg twice daily as part of their HAART regimen. The virologic and immunologic responses at the end of the 48-week study were similar in the two treatment arms. The percentage of patients with an HIV RNA level <400 and <50 copies/mL was 78% and 50% in the extended-release group and 67% and 49% in the immediate-release group. Although the safety profile was similar for both treatment arms, there was a substantially
lower incidence of neuropathy in the extended-release group (1% of patients vs. 11% in the immediate-release group). Based on the efficacy and safety of extended-release stavudine, an NDA was filed with the FDA in December 2001.
Evidence is also building for the potential of once-daily dosing of PIs. An open-label phase II study of atazanavir, an investigational PI, compared two doses, 400 mg once daily or 600 mg once daily, with nelfinavir 1250 mg twice daily
(16). All three treatment groups also received stavudine and lamivudine. Similar proportions of patients in the three groups achieved and maintained viral load reductions below quantifiable levels. Larger phase III trials are currently underway comparing atazanavir with efavirenz and lopinavir/ritonavir.
Once-daily dosing of boosted PIs is another strategy to simplify PI-containing treatment regimens. One study evaluated subjects after they were switched from a twice-daily regimen of saquinavir to a once-daily regimen of ritonavir-boosted saquinavir. The subjects in the study initially received saquinavir 1400 mg combined with Combivir or stavudine and didanosine, and after the switch they received once-daily saquinavir 1600 mg/ritonavir 100 mg plus the
same NRTIs. Subjects who had maintained HIV RNA levels <50 copies/mL for 112 weeks with the initial regimen were eligible to be switched to the once-daily boosted regimen. The results showed that the once-daily saquinavir/ritonavir combination was effective in maintaining viral load suppression. More than 90% of patients maintained HIV RNA levels
<50 copies/mL at 24 weeks after the switch, and the remaining had HIV RNA levels <400 copies/mL. CD4+ T-cell counts also increased by 24 weeks after the switch. None of the patients dropped out of the study after the switch
because of adverse events. Trough concentrations of saquinavir exceeded the IC50 of wild-type virus at the end of the 24-hour dosing interval.
Another study evaluated a similar type of switch, among 39 patients who were stable on a triple-drug regimen of amprenavir 1200 mg twice daily in combination with abacavir and lamivudine. In this 12-week study, subjects were switched to either amprenavir 1200 mg/ritonavir 200 mg once daily or amprenavir 600 mg/ritonavir 100 mg twice daily. Antiviral activity was maintained in each of the boosted amprenavir arms, whereas the amprenavir pill burden was reduced from 16 pills per day to 8 (19).
Boosted indinavir has also been studied as a once-a-day PI option. In one trial, the combination of indinavir 1200 mg/ritonavir 200 mg once daily combined with stavudine and lamivudine twice daily in 40 subjects reduced viral loads
to <400 copies HIV RNA/mL in 80% of individuals (20). The mean increase in CD4+ T-cell counts was 157 cells/mm3 at 24 weeks. No discontinuations because of adverse events occurred, and no incidence of nephrolithiasis was reported.
The studies described above evaluate agents dosed once daily in combination with others dosed more frequently. Combination therapy in which all of the agents are taken only once daily has also been investigated. In a 48-week,
single-arm, open-label study, 75 subjects received buffered didanosine 300 mg, lamivudine 300 mg, and efavirenz 600 mg once daily, with all drugs administered simultaneously at bedtime. In an intent-to-treat analysis, 77% of
subjects achieved HIV RNA levels <50 copies/mL (median reduction >3.4 log10 copies/mL), and mean CD4+ T cell counts increased from 251 cells/mm3 at baseline to 459 cells/mm3 at 48 weeks. In a second pilot study, emtricitabine, an investigational NRTI, was dosed together with didanosine and efavirenz (22). Again, all three drugs were taken simultaneously at bedtime. After 64 weeks of therapy, 90% of the patients achieved viral loads <400 copies/mL. These
results support the premise that once-daily dosing of antiretroviral combinations will be effective. Trials comparing combinations that are dosed once daily with twice-daily combinations will be important to demonstrate that the improved adherence afforded by once-daily dosing will outweigh the consequences of missing a dose of a combination that is only taken once per day.
Not all drugs that are potentially available as once-daily agents can be assembled into a once-daily combination. Food restrictions may require some drugs to be taken in a staggered fashion. For example, it is recommended that didanosine and efavirenz be taken on an empty stomach, whereas tenofovir and lopinavir/ritonavir should be taken with food. Also, there are pharmacokinetic interactions between didanosine and tenofovir; tenofovir boosts didanosine concentrations. Such drugdrug interactions require further evaluation to establish the appropriate doses of these drugs when administered together.
DISADVANTAGES OF ONCE-DAILY THERAPY
The most common reservation concerning once-daily antiretroviral therapy is the consequence of missing a dose and the potential of being off medication for an entire 24-hour period. Some clinicians argue that there may be a smaller risk of resistant virus selection if a patient misses a dose in a twice-daily combination because the period of suboptimal drug exposure would be theoretically shorter than that for a patient missing a once-daily dose. However, the "forgiveness factor," the period of time during which drug concentrations remain above the viral IC50 level, depends upon the
pharmacokinetic properties of the drug, not the number of times a day it is dosed.
For example, when amprenavir 1200 mg is administered with ritonavir 200 mg once daily, the plasma concentration of amprenavir exceeds the IC50 of wild-type virus by approximately 6-fold 12 hours after a missed dose and by
approximately 3.6-fold 24 hours after a missed dose. In contrast, if a patient misses a single dose of amprenavir in a 1200 mg twice-daily regimen, the plasma concentration of drug would fall below the IC50 within several hours after the missed dose. These pharmacokinetic data suggest that amprenavir is a more forgiving drug when dosed once daily in combination with ritonavir than when administered as a single PI twice daily. Not all boosted PIs may be as forgiving as boosted amprenavir. When saquinavir 1600 mg is co-administered with ritonavir 100 mg once daily, concentrations of saquinavir are only 0.73 times the IC50 12 hours after a missed dose and are 0.17 times the IC50 24 hours afterward. The corresponding values for indinavir, following a once-daily dose of 1200 mg with ritonavir 100 mg, are 0.37 and 0.05 times the IC50. Similar data using lopinavir/ritonavir are needed.
Another important consideration when evaluating the consequences of missing a dose in a once-daily or twice-daily combination is the opportunity to make up for a missed dose. If one dose of a twice-daily regimen is missed, the patient
may not be able to make up the missed dose. Conversely, a once-daily dose missed at bedtime could still be made up the next morning, thereby maintaining therapeutic coverage.
CONCLUSIONS
Adherence remains one of the greatest challenges for patients receiving antiretroviral treatment. Increased simplification of treatment regimens should assist in improving the long-term adherence to therapy and maintaining treatment efficacy. In the future, once-daily regimens will become the standard of care for HIV infection. At present, several currently available antiretroviral drugs can be used once daily or have the pharmacokinetic potential for once-daily dosing, and others are in development.
| |
| |
|
|
|