 |
 |
 |
| |
Hepatitis B Report from DDW Conference: adefovir, tenofovir, clevudine
Reported by Jules Levin
|
| |
| |
Hepatitis B is a global health concern despite the availability of a vaccine. Although vaccines for hepatitis B and hepatitis A are available, they are often not administered to at-risk individuals. In particular, the rates of vaccination for Hepatitis A and Hepatitis B in HIV-infected and in HCV/HIV coinfected individuals are stunningly low. Treatment options for HBV are growing. Drugs used to treat HBV were interferon and lamivudine, and in recent years adefovir and tenofovir were added. Several studies updating us on adefovir and tenofovir were presented at the recent DDW Conference, and are discussed below. A number of additional potential treatments for HBV are in various stages of early development including Clevudine (L-FMAU), which is a new drug for HBV in early development and a 28-day dose ranging study is reported below.
Comparison of Viral Replication Fitness of Wild-Type and Lamivudine Resistant HBV in Patients
In order to address whether lamivudine-resistant virus mutations (YMDD) are less fit and therefore have less clinical impact for patients compared to wild-type HBV, Delaney and colleagues and Gilead Sciences presented data at an oral talk comparing HBV DNA and ALT in patients who were lamivudine-resistant vs those with wild-type HBV DNA.
Lamivudine-resistant HBV has been described as replication defective based on early clinical observations and in vitro data for single mutations of M204V or M204I in the YMDD motif of HBV polymerase. However, recent data suggests that compensatory mutations also emerge and patients with long-term lamivudine resistance undergo disease progression.
This goal of this study was to compare levels of serum HBV DNA and ALT in patients with lamivudine-resistant and wild-type HBV. And to determine the prevalence of compensatory mutations in YMDD-mutants and their contributions to replication fitness.
Patients analyzed were enrolled in trials of adefovir dipivoxil for the treatment of wild-type (2 trials, n=695) or lamivudine-resistant HBV (3 trials, n=203). Patients were required to have serum HBV DNA levels ³5 log10 copies/mL at entry. Serum HBV DNA was measured by Roche Amplicor PCR. DNA sequencing was used to identify resistance mutations at baseline.
At baseline, patients with lamivudine-resistant HBV had similar serum HBV DNA (medians 8.2, 8.1, and 8.8 log10 copies/mL) compared to patients with wild-type HBV (medians 7.1 and 8.4 log10 copies/mL). As well, ALT levels were elevated for both patient groups and only slightly higher for wild-type: median ALT levels were 79-82 IU/L ( x ULN) in lamivudine-resistant and 94-98 (2.3 ULN) IU/L in wild-type patients.
Compensatory mutations (V173L, L180M) that enhance the replication of YMDD-mutant HBV in vitro were found in the majority (88%) of lamivudine-resistant patients. At baseline, four major mutational patterns were identified in patients with lamivudine-resistant HBV (L180M+M204V [58%], V173L+L180M+M204V [17%], M204I [12%], L180M+M204I [11%]); serum HBV DNA did not vary significantly across the different mutational patterns.
The authors concluded that lamivudine-resistant HBV is capable of replicating to levels comparable to wild-type HBV and causing abnormally high ALT. Exclusion of patients with lower serum HBV DNA levels and normal ALT from eligibility preclude an assessment across a greater spectrum of viral load. The four patterns of lamivudine resistance mutations all had similar levels of serum HBV DNA.
Delaney W, Westland C, Yang H, Gilead Sciences, et al. Comparison of viral replication fitness of wild type and lamivudine-resistant HBV in patients. Gastroenterology. 2003;124. DDW Conference, May 17-22, 2003, Orlando, FL [Abstract #339]
Safety of Adefovir in HBV: 48 and 109 weeks follow-up
Tong and Gilead Sciences reported on the safety and tolerability of adefovir for patients with chronic hepatitis B followed for 48 weeks with a continued follow-up of a smaller number of patients receiving ADV for 109 weeks
ADV is a nucleotide analog with activity against wild-type, precore and lamivudine-resistant hepatitis B virus (HBV) resulting in significant histological, serological, and biochemical improvement.
The objective of this study was to report safety results of treatment with ADV 10 mg once daily for up to 109 weeks in HBeAg+ or HBeAg- chronic hepatitis B patients with compensated liver function. Data from two phase III studies (study 438 and study 437) were integrated to look at 294 patients who received ADV and 228 who received placebo. Clinical and laboratory adverse events (AEs) were analyzed out to 96 weeks and 180 patients out to 108 weeks.
Patients were 78% male, 48% Caucasian, 47% Asian, median age 40 years (range 16 to 67 years); median ADV exposure: 49 weeks (range 0.4-109.4 weeks). Median HBV DNA was 7.46 log10 copies/mL; median ALT was 1.95 x ULN; 35% of patients had bridging fibrosis or cirrhosis.
The authors reported that the frequency and nature of AEs was similar to that seen with ADV and placebo in the first 48 weeks. AEs leading to drug discontinuation in 3% of patients and Serious AEs were reported in 5% of patients from week 0-48, and 5% for patients on ADV from week 0-96. From weeks 0-96 84% of the 492 patients on ADV 10mg had an AE. From week 0-48 45% reported AE related to study drug on ADV 10mg, 50% on placebo, and for patients on ADV 10mg from 0-96 weeks 36% reported AE related to study drug. From week 0-48 total discontinuations were 6% in placebo arm, 5% in ADV arm, and from week 0-96 for patients on ADV 10mh 8%.
No single SAE occurred in > 2 patients (< 1% each). One unrelated death was reported. Median baseline serum creatinine was 0.8 mg/dL and median change was 0.0 mg/dL. Two patients (<1%) had a confirmed serum creatinine increase ³ 0.5 mg/dL from baseline; both resolved. Median baseline serum phosporous was 3.4 mg/dL and median change was 0.0 mg/dL. No patients reported serum phosphorous decrease to <1.5 mg/dL.
There were no differences from week 0-48 between placebo and ADV 10mg for (grade 3/4 lab abnormalities) ALT >5 x ULN, AST >5 ULN, Hematuria (³3+), CK >4 x ULN, Amylase >2 x ULN, Glycosuria (³3+). Patients on ADV from week 0-96 had similar rates of these grade 3/4 abnormalities, there were no reported changes in percent of patients reporting this.
Of 111 patients who switched from ADV to placebo in the second 48 weeks, 25% had ALT elevations > 10 x ULN; none experienced liver decompensation.
The authors concluded that safety and tolerability of ADV 10mg was similar to placebo through 48 weeks, increases in ALT and AST were more frequent on placebo. Safety appears similar with extended dosing up to 109 weeks, with more limited amount of patients on drug, ADV 10 mg once daily was well tolerated in up to 109 weeks of dosing. Serum creatinine elevation was rare. After stopping therapy there is chance for ALT flares (25% in this study), so patients who discontinue HBV therapy, including ADV, should be closely monitored for ALT flares.
M. Tong, M. Shiffman, Brosgart,et al. Long-Term Safety Beyond 48 Weeks of adefovir Dipivoxil 10 mg Once Daily For Chronic Hepatitis B: an integrated analysis of Two Phase III Studies. Gastroenterology. 2003;124. DDW Conference, May 17-22, 2003, Orlando, Fl.
Changes in ALT and YMDD Resistance After Switching From Lamivudine to Adefovir
Peters and Gilead Sciences reported in this study that YMDD mutations often reverted after switching from lamivudine to adefovir; ALT changes after switching from lamivudine to adefovir are described below.
YMDD mutations in hepatitis B virus (HBV) polymerase emerge in 24% (range 16 to 32%) of CHB patients after one year of LAM therapy, increasing to 69% after 5 years. Studies have shown ADV has potent activity against YMDD mutant and wild type (WT) HBV. This study reported on an evaluation of the impact of switching from lamivudine to ADV or lamivudine+ADV on changes in ALT, and to evaluate the impact of switching from lamivudine to ADV or lamivudine+ADV on YMDD mutations (3TC/lamivudine resistance mutations).
Study GS 00 461 evaluated 3 regimens: continued lamivudine100 mg, adding ADV 10 mg to continued lamivudine or switching from lamivudine to ADV. Eligible patients were: HBeAg+ with CHB; ALT ³ 1.2 x upper limit of normal (ULN); HBV DNA > 6 log10 copies/mL (Roche Amplicor PCR) despite ongoing lamivudine therapy for >6 months; confirmed YMDD mutant HBV.
Fifty eight patients received ADV (n=19), ADV + LAM (n=20), or LAM (n=19). Treatment duration was 48 weeks: ALT was evaluated during therapy by treatment arm for possible associations with change in regimen (ALT elevations £ week (W) 12) and/or viral genotype. YMDD mutations were monitored by DNA sequencing at baseline, week 16, week 32 and week 48. Median age was 44-46, males 74% to 89%, caucasians 45% to 74%, asians 26% to 37%, and median prior lamivudine use was 24 to 37 months. Median HBV DNA was 7.9 to 8.4 log10 copies/ml, median ALT 73 (ADV+LAM), 70 (LAM), 101 (ADV). 100% of patients had YMDD mutations.
|
|
 |
| |
Median change from baseline to week 48 in serum ALT was 0 for Lam, -25 IU/L for ADV+Lam from a baseline of 74 IU/L, and -51 IU/L for ADV from a baseline of 101 IU/L.
PATIENTS with ALT ELEVATIONS THROUGH WEEK 48
5 patients on ADV (5/20) through week 4-12 had 5-10 x ULN ALT, 0 in Lam and Lam+ADV arms. Through week 16-48 1 in ADV group had 5-10 x ULN ALT, 0 in Lam and Lam+ADV groups. Regarding elevations in ALT of >10 ULN, during week 4-12 1 patient experienced this in ADV+Lam group, 0 in ADV group, and 0 in Lam group; from week 16-48 3 patients in Lam group experienced >10 X ULN ALT and 0 in Lam+ADV and ADV groups.
Post-Baseline ALT Flares to Week 48
|
|
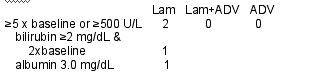 |
| |
1
YMDD MUTATIONS REVERSIONS to WILD-TYPE
YMDD sequencing (position rtM204) demonstrated that by W16, W32, and W48 of therapy, 21%, 32% and 37% of the ADV patients reverted to wild type, respectively. No lamivudine patients and 1 of 20 ADV + Lam patients lost YMDD mutations through week 48. Reversion to WT HBV occurred without increases in serum HBV DNA or ALT, except 1 patient who lost YMDD had increase in ALT of >5xULN at week 4 (ALT levels remained <5xULN at all subsequent visits). Study authors reported that serum HBV DNA decreased similarly in ADV treated patients who reverted to WT HBV or retained YMDD: for 4 patients with Lam-resistant HBV with L180M+M204V who lost mutation on ADV median change in HBV DNA was -3.5 log; for 1 patient with Lam-resistant mutation (V173L+L180M) median change in HBV DNA on ADV was -4.0 log; for 1 patient with V173L+L180M+M204V/M204I on ADV median change in HBV DNA was -5.9 log; for 1 patient with lam-r HBV (L180M+M204I) median chamge in HBV DNA on ADV was -4.9 log; and for 1 patient with L180M+M204V/I HBV DNA at week 48 was not available.
Most commonly reported (in at least 10 patients across all treatment groups) adverse events did not appear to be different for Lam continued vs ADV groups: asthenia (32% Lam-42% ADV), headache (26% Lam-21% ADV), abdominal pain (21% lam-16% ADV), pharyngitis (21% lam- 26% ADV).
The study authors concluded that ADV, alone or in combination with LAM, significantly reduced serum HBV DNA and reduced and normalized ALT in patients with LAM resistant HBV. Change in HBV DNA and ALT was similar for ADV and ADV/Lam. Reversion to WT HBV from YMDD occurred more frequently in patients receiving ADV monotherapy, and reversion to wild-type was associated with changes in HBV DNA and ALT similar to those seen in ADV treated patients maintaining YMDD. Switching to ADV from Lam resulted in transient self-limited elevations in ALT for some patients (see data above), but were not associated with signs or symptoms of hepatic decompensation; neither directly switching to ADV from LAM nor loss of YMDD mutations through 48 weeks was associated with severe ALT flare or evidence of hepatic decompensation.
M Peters, P Martin, C Brosgart et al. Changes in ALT and YMDD Mutation Profile Associated with Switching from Lamivudine to either Adefovir or Combination Lamivudine Plus Adefovir in Patients with Chronic Hepatitis B with Lamivudine Resistance. Gastroenterology. 2003;124.
Tenofovir Treatment in Hepatitis B Monoinfected Patients With Lamivudine-Resistant Virus
Kulig and colleagues reported changes in ALT and HBV DNA by the use of tenofovir 150 mg once daily in treating 6 HBV monoinfected patients previously treated with lamivudine.
Lamivudine resistant strains of hepatitis B (YMDD mutations) emerge during lamivudine therapy in patients with chronic hepatitis B (HBV). Tenofovir dipivoxil, has been shown to suppress HBV replication in HIV co-infection.
Six patients previously treated with lamivudine with documented YMDD (3TC/lamivudine resistance) mutations (Polymerase Chain Reaction [PCR] amplification/Restriction Fragment Length Polymorphism [RFLP] Assay, Roche Molecular Systems, Inc., Branchburg, NJ) were treated with tenofovir 150 mg/day. Patients continued lamivudine therapy, 75 to 150 mg. Liver tests and HBV-DNA were monitored at monthly intervals.
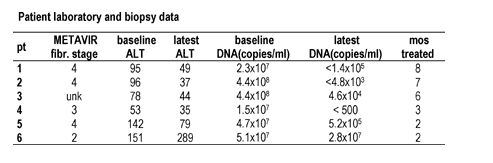
Four males and two females, age range 33 to 66 years, had been treated with lamivudine for 1 to 4 years. YMDD mutations were as follows: patient 1: M552I, codon 528 wild type (WT); patient 2: M552I, L528M; patient 3: M552V/I, codon 528 WT/L528M mix; patient 4: M552V, L528M; patient 5: M552I, 528 WT, and patient 6: M552V, codon 528 WT/L528M mix.
HBV DNA was quantitated by hybridization capture using the Digene HC II¨ assay with both the standard method (dynamic range: 1.42x105 to 1.7x109 copies/ml) and ultra-sensitive method (dynamic range 4.7 x 103 to 1.7 x109 copies/ml)(Digene, Inc., Gaithesburg, MD, 1 pg = 283,000 copies of HBV DNA) or Amplicor¨ PCR (Roche, Inc., detection threshold = 500 copies/ml).
Patient 6 developed elevated liver tests, rash and nausea/vomiting and discontinued tenofovir after 2 months. For the remaining five patients, the mean pre-treatment HBV-DNA level was 1.93 x 108 copies/ml, which decreased by approximately 3 log to 1.42 x 105 copies/ml. Mean pre-treatment ALT, 93 IU/l, decreased 52% to 48 IU/l. No elevation of serum creatinine >0.3 mg/dl above baseline was noted. Patient laboratory, biopsy and treatment duration data are depicted in the Table below.
The authors concluded that tenofovir may be an effective treatment for lamivudine-resistant HBV. We speculate that tenofovir may also be effective in wild-type HBV infection and renal insufficiency.
|
|
 |
| |
C Kulig, J Trotter, G Everson. Treatment of Lamivudine-Resistant Chronic Hepatitis B With Tenofovir. Gastroenterology. 2003;124. DDW Conference, May 17-22, 2003, Orlando, FL.
Clevudine: 28-day study of new once daily HBV drug
Clevudine (CLV, L-FMAU) is a potent inhibitor of HBV replication in vitro. In woodchucks, CLV produced a potent and sustained viral suppression following a 12 weeks dosing period.
Marcellin and colleagues reported the findings from a multicenter, open-label, dose escalation study evaluating 10, 50, 100 and 200 mg CLV once daily for 28 days (n=5, 10, 10 and 7/arm, respectively). Patients were followed post-treatment for 24 weeks. Eligible patients had chronic HBV infection, Baseline HBV DNA levels (viral load ³3x106) copies/mL, were nucleoside treatment naive and without HIV or HCV co-infection.
VL was assayed using Digene Hybrid Capture II (with a lower limit of detection of 4700 c/mL) and genotype by di-deoxy sequencing.
32 patients were enrolled, 81% were male, 81% Asian, 88% HBeAg positive. At Baseline, median viral loads were 7.3, 8.0, 8.8 and 8.4 log10 c/mL and median ALTs were 55, 119, 106 and 64 IU/L in the 10, 50, 100 and 200 mg QD cohorts, respectively.
After 28 days of dosing, the median log10 viral load change from baseline was -2.5, -2.7, -3.0 and -2.6 and median change in ALT from Baseline was -13, -14.5, +37 and -14 U/L, in the 10, 50, 100 and 200mg cohorts, respectively.
At 6 months post-dose, sustained biochemical and virologic responses were observed: 71% of the patients overall had normal ALT levels and median log10 viral load changes from Baseline were -1.2, -1.4, -2.7 and -1.6 in the 10, 50, 100 and 200mg arms, respectively.
During the study, 8 patients lost HBeAg (30%) of whom 5 seroconverted to HBeAb (19%). Authors reported CLV was well tolerated, without dose related adverse events. A transient increase in ALT was observed in the 100 mg cohort but not in the 200 mg group.
The pharmacokinetics of CLV were dose proportional with a long plasma half-life supporting a once daily regimen. No treatment emergent mutations in the HBV DNA pol domain were observed 5 months after treatment.
The authors concluded that these results confirm the antiviral activity of once daily clevudine and further demonstrate, in humans the uniquely sustained post-treatment antiviral effect seen previously in woodchucks. Despite the short (4 weeks) duration of therapy very favorable rates of HBeAg loss and seroconversion were observed without evidence to date of acquired resistance on CLV therapy. Further study is needed to confirm these early findings.
|
|
|
 |
 |
|
|