 |
 |
 |
| |
|
Lamivudine in Adolescents with Hepatitis B
|
| |
| |
Researchers from China and GlaxoSmithKline reported results from this study at the DDW Conference in May 2003 in Orlando. The study authors reported that the antiviral effect of lamivudine is similar in both adults and adolescents with chronic hepatitis B in this study after 12 months treatment. And the incidence of adverse events were similar in both groups. They also reported that retardation of growth was not observed in these lamivudine-treated adolescents. They concluded that lamivudine is effective, well-tolerated and safe for the treatment of chronic hepatitis B in adolescent patients.
The authors reported that the efficacy of lamivudine for chronic hepatitis B (CHB) has been documented in Chinese patients age >16 in a previous phase III clinical trial. Adolescent HBeAg positive CHB patients age >12 were included for evaluation in a large scale open-label phase IV study, which recruited up to 2200 patients. The objective of this study was to assess the efficacy and safety of lamivudine in Chinese adolescents with HBeAg positive CHB. All patients received 100 mg lamivudine daily for 12 months. Serum HBV markers were assayed using Abbott IMX kits, HBV DNA was determined by solid hybridization assay (sensitivity 1.0 pg/ml).
Between November 2000 and March 2001, a total of 98 HbeAg and HBV DNA positive adolescent patients aged 12 to <16 years and 1828 adult patients were enrolled into this study from 45 sites in China. 76 adolescents (78%) were HBV DNA and HBeAg positive and ALT abnormal, 22 adolescents (22%) were HBV DNA positive and HBeAg negative with ALT normal. Overall, 94 (96%) adolesecent patients completed 1 year of treatment. No adolescent patient withdrew from the study due to an adverse event.
RESULTS
The adolescent patients were at least as likely to have a virologiv response to 1 year of lamivudine therapy treatment as adult patients. The negativity of HBV DNA in ado;secent patients with abnormal ALT was 80% at month 3, 93% at month 6, and 92% at month 12. The HBV DNA negativity rate in adult patients was 60% at month 3, 87% at month 6, and 78% at month 12.
In ALT normal patients, the negativity of HBV DNA was similar between adolescents and adults. Study authors reported the rate of HBeAg seroconversion in adolescent patients was associated with their pre-treatment ALT levels, which is consistent with the observation in adult patients. The data appeared better than that in adults: 22% of adolescents had seroconversion vs 16% of adults. HbeAg loss was experienced by 24% of adolescents vs 19% of adults. HBeAg seroconversion/HBV DNA negativity was experienced by 22% of adolescents vs 14% of adults.
The study authors reported that in adolescent patients, 80% (75/94) were ALT normal and 69% (65/94) achieved both ALT normal and HBV DNA negative at month 12, compared with 74% and 59% in adult patients.
|
|
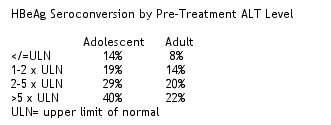 |
| |
In patients with abnormal ALT at baseline, ALT normalization was at month 3 58% in adolescents vs 51% in adults, at month 6 68% in adolescents vs 66% in adults, and at month 12 77% in adolescents vs 71% in adults.
SAFETY
The authors reported that during the 12 months treatment, 18 adolescents were reported to have at least one non-serious adverse event. A total of 26 adverse events were reported, among which 73% were reported as not related or unlikely related to the study drug and 58% as reported being mild. 8 adolescent patients were reported to have laboratory abnormalities with clinical significance as defined by the protocol. Compared with adult patients in this study, there was no significant difference observed in the adolescent patients in regard to the AE frequency and severity.
The authors concluded that 1 year lamivudine treatment results in significant HBV DNA suppression and increased HBeAg seroconversion and HBeAg loss in adolescent patients. Lamivudine is well-tolerated by adolescent patients.
|
|
|
 |
 |
|
|