 |
 |
 |
| |
Atazanavir Maintained Patient Utility and Improved Quality of Life, Comparing Favorably to LPV/RTV: 24-Week Data from BMS 043
|
| |
| |
Virologic Determinants of 24-Week Efficacy of Atazanavir With or Without Ritonavir in Patients with Prior Failure of a Protease Inhibitor
Oral presentation at 9th EACS, Warsaw, Poland, October 2003
C Zala, A Lazzarin, A Casiro, B Grinsztejn, L Nietro, R Salazar, E Ledesma, T Kelleher, R McGovern, A Rightmire, C McLaren. Buenos Aires, Argentina; Milan, Italy; Rio de Janiero, Brazil; Mexico; Lima, Peru; BMS, Wallingford, CT
ATV 300 mg boosted with RTV 100 mg increases AUC and Cmin. The objective of this study was to determine the extent to which baseline viral mutations correlate to antiviral effect of unboosted atazanavir (ATV 400 mg QD) and boosted atazanavir (ATV 300 mg QD plusRTV 100 mg QD).
In BMS 043 patients who were currently failing PI-based regimen were randomized to ATV 400mg QD plus 2 NRTIs (n=150) or LPV/rtv 400/100mg BID plus 2 NRTIs (n=150). BMS 045 patients who failed at least 2 regimens including ≥1 PI, ≥NRTI, ≥NNRTI were randomized to ATV/rtv 300/100mg QD plus tenofovir (TDF) plus 1 NRTI, LPV/rtv 400/100mg plus TDF+1 NRTI, or ATV/sqv 400/1200mg QD plus TDF plus 1 NRTI.
About 50% in 043 and 22% in 045 were Latino; 43% in 043 and 60% in 045 were White; 6% in 043 and 16% in 045 were Black. About 25% had AIDS, median Cd4 count was about 280-290. These numbers were comparable across 043 and 045.
Median viral load was 4.14-4.18 in 043 and 4.44 to 4.47 in 045. The patient population appeared to have less advanced HIV in 043: 69% in ATV 400mg QD and 59% in LPV/r 400/100mg BID had <30,000 copies/ml; 16% and 23%, respectively, had 30,000-<100,000 c/ml; 15% and 17%, respectively, had >100,000 c/ml. In 045 appeared to have more advanced HIV: viral load was higher, more patients had viral load >30,000 and >100,000 c/ml; as well, more patients in 045 had >4 PI mutations and >4 NRTI mutations.
|
|
| |
| |
| BMS 043 |
| |
ATV |
LPv/r |
| |
400mg QD |
400/100mg BID |
| HIV-RNA |
4.18 |
4.14 |
| <30,000 |
69% |
59% |
| -100K |
16% |
23% |
| >100K |
15% |
17% |
| Baseline susceptibility |
| |
ATV, n (%) |
108 (72%) |
| |
LPV |
129 (86%) |
| Baseline phenotypic susceptibility determined as <2.5 fold change (Virologic Phenosense) |
| >4 mutations at baseline |
| PI |
39 (26%) |
35 (23%) |
| NRTI |
24 (16%) |
24 (16%) |
|
|
| |
| |
| BMS 045 |
| |
ATV |
LPv/r |
| |
300/100mg QD |
400/100mg BID |
| HIV-RNA |
4.44 |
4.47 |
| <30,000 |
53% |
50% |
| 30K-100K |
23% |
27% |
| >100K |
23% |
23% |
| Baseline susceptibility |
| |
ATV, n (%) |
88 (74%) |
| |
LPV |
91 (77%) |
| Baseline phenotypic susceptibility determined as <2.5 fold change (Virologic Phenosense) |
| >4 mutations at baseline |
| PI |
39 (33%) |
45 (37%) |
| NRTI |
47 (39%) |
53 (43%) |
|
|
| |
| |
TREATMENT HISTORY (prior ARV use, median)
Prior NRTI use weeks (range) was greater in 045: In 043, patients taking ATV had 159 weeks use (0.1-645) and patients taking LPV/r had 167 weeks (26.1-630) prior NRTI use; in 045, patients taking ATV/r had 269 weeks (40-782) and patients taking LPV/r (0.1-679) had prior 265 weeks prior NRTI use.
Prior PI use was about the same in 043 & 045 -weeks (range): In 043, patients taking ATV had 133 weeks (0.1-338) prior PI use and patients taking LPV/r had 136 (0.1-536) weeks prior PI use; in 045, patients taking ATV/r 133 (0.1-321) weeks prior PI use and patients taking LPV/r had 136 (0.1-346) weeks prior PI use.
Prior NNRTI use weeks (range): In 043, patients taking ATV had 133 (1.6-210) weeks prior NNRTI use, and patients taking LPV/r had 85 (1.6-158) weeks prior NNRTI use. In 045, patients taking ATV/r had 78 weeks (0.1-227) prior NNRTI use and patients taking LPV/r had 69 weeks (0.1-304) prior NNRTI use. In 043 100% of patients had prior PI use, in 045 60% had prior NNRTI use and 37% had prior PI use.
|
|
| |
| |
| |
BMS 043 |
BMS 045 |
| |
ATV |
LPV/r |
ATV/r |
LPV/r |
| #randomized (n) |
150 |
150 |
120 |
123 |
| Prior ARV use (median) Prior NRTI use (wks) |
159 |
167 |
269 |
265 |
| Prior PI use (wks) |
133 |
136 |
133 |
136 |
| Prior NNRTI use (wks) |
133 |
85 |
78 |
69 |
|
|
| |
| |
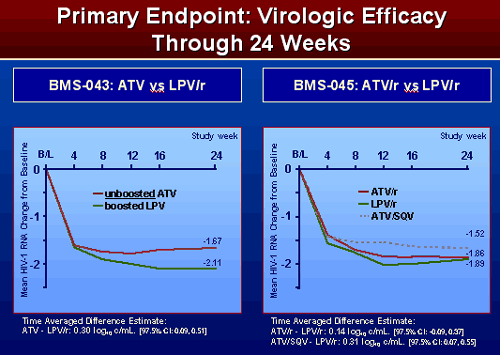
PRIMARY ENDPOINT: VIROLOGIC EFFICACY THROUGH WEEK 24
Mean HIV-1 change from baseline
In 043, viral load was reduced by 1.67 in ATV unboosted vs 2.11 in LPV/r. In 045, viral load was reduced by 1.52 in ATV/sqv arm, -1.86 in ATV/r, and 1.89 for LPV/r.
|
|
| |
| |
 |
|
| |
| |
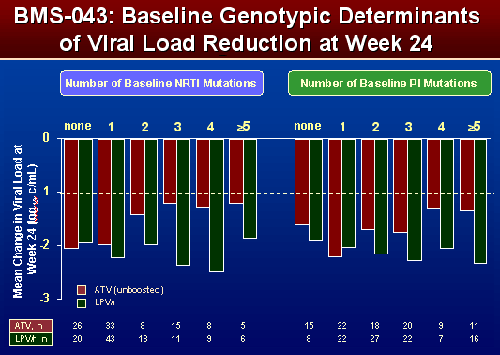
EFFECT OF NRTI and PI SUBSTITUTIONS on VIRAL LOAD REDUCTIONS
BMS 043
For patients with 2, 3, 4, and 5 or more PI mutations viral load reductions appeared greater when taking LPV/r. For patients with 0 or 1 PI mutation the viral load changes appeared comparable whether taking LPV/r or ATV unboosted. Patients with 2 to >5 NRTI mutations appeared to have greater viral load reductions when taking LPV/r. Patients with none, or 1NRTI mutation appeared to have comparable VL reductions whether taking ATV unboosted or LPV/r.
|
|
| |
| |
 |
|
| |
| |
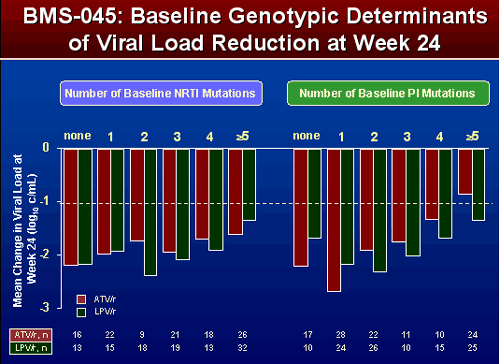
BMS 045
For patients with none or 1 PI mutation ATV/r viral load reductions appeared greater, for patients with 2, 3, 4, 5, or >5 PI mutations VL reductions appeared greater for patients taking LPV/r. Similar responses were seen when examining NRTI mutations, except when there were 5 or more NRTI mutations patients receiving ATV/r had more reduction in VL.
|
|
| |
| |
 |
|
| |
| |
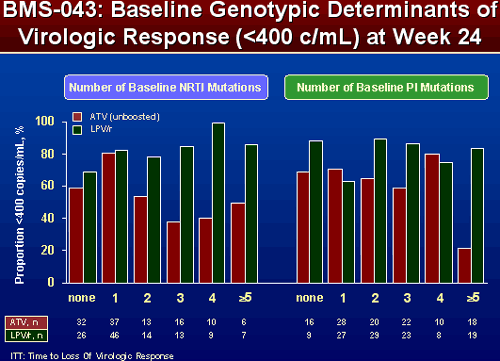
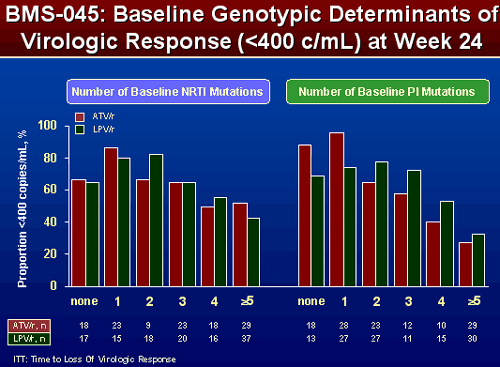
EFFECT OF NRTI and PI SUBSTITUTIONS ON PROPORTION OF SUBJECTS <400 COPIES/ML AT WEEK 24
When patients in 043 had more NRTI and PI mutations, the percent of patients with <400 c/ml appeared greater when taking LPV/r. Results were slightly different in 045. The number of NRTI mutations didn’t appear to matter that much. But the number of PI mutations did appear to matter. Patients with 2 or more mutations appeared to have a higher percent <400 c/ml when taking LPV/r, patients with none or 1 PI mutation appeared to have greater VL reductions when taking ATV/r.
|
|
| |
| |
 |
|
| |
| |
 |
|
| |
| |
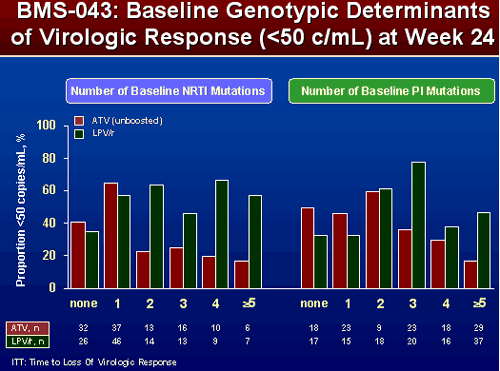
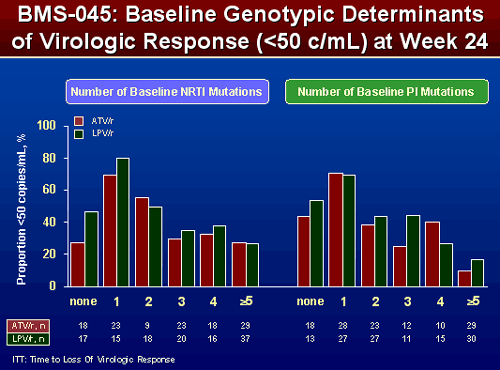
EFFECT OF NRTI AND PI SUBSTITUTIONS ON PROPORTION OF SUBJECTS <50 COPIES/ML
There are some differences between the effect of ATV/r and LPV/r but differences are not as great and are not as consistent. For patients with ≥2 NRTI mutations response appeared similar whether taking ATV/r or LPV/r. For patients with 2 or more PI mutations they did appear to do better with LPV/r, but again differences weren’t as great and less consistent.
|
|
| |
| |
 |
|
| |
| |
 |
|
| |
| |
AUTHOR’S CONCLUSIONS
The efficacy of ATV- and LPV-containing regimens decreased with increasing numbers of PI and NRTI mutations.
The relative magnitude of this effect was greatest in non-boosted ATV regimens.
The effect of PI and NRTI mutations was similar in ATV/r and LPV/r regimens.
These results indicate that RTV-boosted ATV may be a more appropriate regimen than unboosted ATV for patients with multiple PI and NRTI mutations.
|
|
| |
|
 |
 |
|
|