 |
 |
 |
| |
Study Suggests More Than 48 Weeks Therapy for Patients with high baseline viral loads, HCV genotype 1, and cirrhosis may be necessary
|
| |
| |
"PREDICTORS OF VIROLOGIC RELAPSE IN PATIENTS WITH CHRONIC HEPATITIS C (CHC) TREATED WITH PEGINTERFERON ALFA-2A (40KD) (PEGASYS) ALONE OR IN COMBINATION WITH RIBAVIRIN (COPEGUS)"
Comments from Jules Levin: A previous study called I think the Benelux Study found that 18 months treatment may improve response rates for some patients. It's my understanding that a study examining 18 months therapy is ongoing. Some doctors are using 18 months therapy for hard to treat patients including patients with multiple characteristics for hard to treat patients including previous non-responders to interferon+ribavirin, cirrhotics, high HCV viral load, HCV/HIV coinfected. The study below suggests 18 months may be more effective than 12 months therapy but well designed safety and efficacy studies need to be conducted. The safety concerns of 18 months therapy need more characterization.
V. Balan*, 1 S. Zeuzem, 2 H. Sette, 3 M. Fried, 4 D. Jensen, 5 G. Pastore, 6 P. Marcellin, 7 F. Sedarati, 8 *Presenting Author
1Mayo Clinic, Phoenix, Scottsdale, USA 2Saarland University Hospital, Homburg, Germany 3Instituto De Infectologia, Sao Paolo, Brazil 4University Of North Carolina, Chapel Hill, North Carolina, USA 5Rush-Presbyterian St. Luke's Hospital, Chicago, Illinois, USA 6Clinic Of Infectious Diseases, University Of Bari, Bari, Italy 7Hopital Beaujon, Clichy, France 8Hoffman-La Roche Inc, Nutley, New Jersey, USA
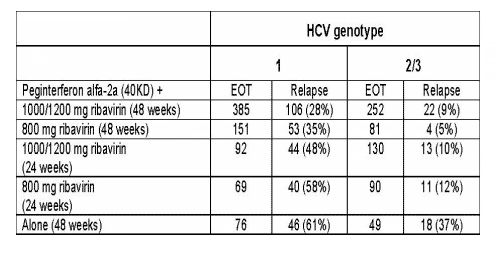
Background: Some patients treated for CHC still relapse after an end-of-treatment (EOT) virologic response (VR), particularly patients infected with HCV genotype 1.
Objectives: To determine the frequency of relapse in patients treated with peginterferon alfa-2a (40KD) (Pegasys) ± ribavirin (Copegus) and to identify baseline parameters associated with viral relapse.
Methods: Chronic hepatitis C (CHC) patients (N=1375) received peginterferon alfa-2a (40KD) 180 mg/week ± ribavirin 800-1200 mg/day for 24 or 48 weeks. Sustained viral response was defined as undetectable HCV RNA at the end of follow-up; and relapse, as detectable HCV RNA following a documented end of treatment response.
Results: High baseline viral load, and, to a lesser degree, cirrhosis, were found to be associated with relapse, particularly in HCV genotype 1 patients.
Conclusion: Treatment with peginterferon alfa-2a (40KD) (Pegasys) /ribavirin (Copegus) results in lower relapse rates than monotherapy in all patients. CHC genotypes 2/3 patients experience lower relapse, regardless of ribavirin dose given, compared with HCV genotype 1 patients whose relapse is dependent on ribavirin dose and duration of treatment. More than 48 weeks treatment for CHC patients with high baseline viral loads, HCV genotype 1, and cirrhosis may be necessary and should be investigated.
|
|
| |
 |
|
| |
|
 |
 |
|
|