 |
 |
 |
| |
|
PK of Once daily Reyataz, DDI-EC, Tenofovir
|
| |
| |
Pharmacokinetic (PK) Evaluation of the Combination of Atazanavir (ATV), Enteric Coated Didanosine (ddI-EC), and Tenofovir Disoproxil Fumarate (TDF) for a Once-Daily Antiretroviral Regimen
S Kaul 1 , K Bassi 1 , B Damle 1 , J Xie 1 , J Gale 1 , B Kearney 2 , G Hanna 1. 1 Bristol-Myers Squibb Company, Princeton, NJ, 2 Gilead Sciences, Inc., Foster City, CA
ICAAC poster A-1616, ICAAC, sept 14-17, 2003, Chicago.
Introduction
The combination of enteric-coated didanosine (ddI-EC)/atazanavir (ATV)/tenofovir disoproxil fumarate (TDF) is a promising once-daily (QD) highly active antiretroviral therapy (HAART) regimen.
The recommended once-daily doses of ddI-EC (a nucleoside reverse transcriptase inhibitor), ATV (a protease inhibitor), and TDF (a nucleotide reverse transcriptase inhibitor) in HIV-infected subjects is 400 mg for subjects >60 kg (250 mg for subjects <60 kg) under fasted conditions, 400 mg with food, and 300 mg with food, respectively.
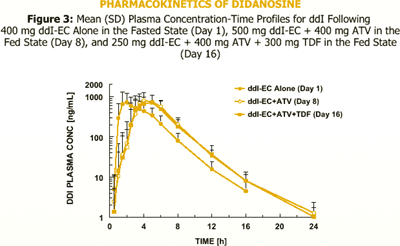
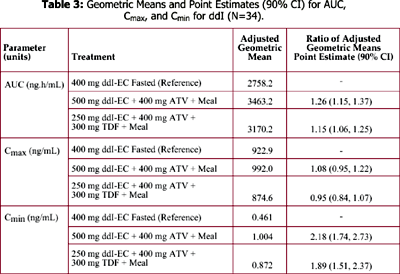
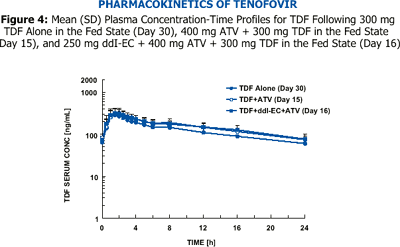
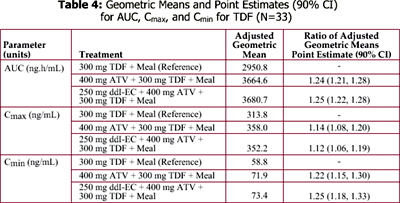
The bioavailability of 400 mg ddI-EC is reduced by about 20-25% with food. Coadministration of 400 mg ddI-EC and 300 mg TDF results in about 60% increase in ddI exposure, but the exposure of TDF is unaltered.
However, exposure of reduced dose of 250 mg ddI-EC with 300 mg TDF and food is comparable to exposure of 400 mg ddI-EC given alone in the fasted state.
Administration of 200 mg ddI chewable/dispersible buffered tablet and 400 mg ATV in the fasted state reduced ATV exposure by almost 90%, but the exposure of ddI was unchanged.
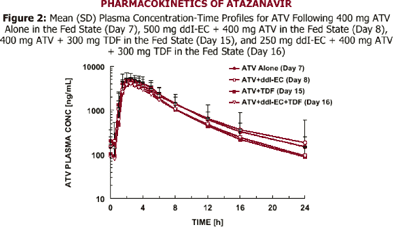
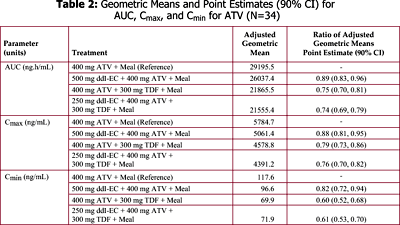
Data from CROI 2003 (PUZZLE 2 [ANSR 107]) suggest that when a ritonavir (R) (100 mg)-boosted ATV (300 mg) regimen was combined with 300 mg TDF, ATV Cmax, AUC, and Cmin values were decreased by approximately 28%, 25%, and 23%, respectively, compared to the 300 mg/100 mg ATV/R reference treatment. Despite these effects, the ATV Cmax, AUC, and Cmin values were still 1.4-, 2.3-, and 4.1-fold higher, respectively, for the ATV/R + TDF regimen, relative to historical data for 400 mg ATV/R alone.
It is unknown if PK interactions between ddI-EC and ATV, ATV and TDF, and between the triple combination of ddI-EC, ATV, and TDF exist.
Methods
--Phase I, open-label study in 36 healthy subjects weighing •60 kg. The design of this study incorporated the sequential introduction of specified drug or drug combinations.
--Serial blood samples were collected on Days 1, 7, 8, 15, 16, and 30 at pre-dose and up to 24 hours post-dose.
--Plasma or serum samples were assayed for ddI, ATV, and/or TDF by validated LC/MS/MS assay methods.
--PK parameters for ddI, ATV, and TDF were derived using a non-compartmental analysis.
--Absence of drug interaction was concluded if the 90% confidence interval (CI) for the ratio of test to reference geometric means fell within 0.80-1.25 interval for AUC; similar assessments were conducted for Cmax and Cmin. Clinical safety evaluations were performed at screening, during the study, and at the time of discharge from the study.
Results
SAFETY AND TOLERABILITY
This study enrolled 36 subjects, of which 33 completed the study. – All 3 discontinuations were for personal reasons.
There were no deaths or other serious adverse events (SAEs).
The most frequently reported AEs were: – ATV alone: diarrhea and pruritis (8.3% each) – ddI-EC+ATV and ATV+TDF: diarrhea (5.9%) – TDF alone: pharyngitis (12.1%)
Most laboratory abnormalities were Grade 1 (73.8%) or Grade 2 (19.4%). There were two Grade 3 elevations in total bilirubin (ATV alone) with normal hepatic transaminases, and one Grade 3 elevation in amylase (TDF alone) with normal lipase. These abnormalities were transient and did not require any intervention.
|
|
| |
| |
 |
|
| |
| |
 |
|
| |
| |
 |
|
| |
| |
 |
|
| |
| |
 |
|
| |
| |
 |
|
| |
| |
Author Discussion/Conclusion
|
|
| |
| |
 |
|
| |
|
 |
 |
|
|