 |
 |
 |
| |
|
SAQUINAVIR/RITONAVIR (SQV/r) PHARMACOKINETICS (PKs) IN HIV+ SUBJECTS: 1000/100 MG BID VS 1600/100 AND 2000/100 MG ONCE DAILY (OD)
|
| |
| |
Marta Boffito1, Laura Dickinson2, Andrew Hill3, Chris Higgs1, Carl Fletcher1, Cameron Johnson1, Sundhiya Mandalia1, David Back2, Brian Gazzard1 and Anton Pozniak1. 1 Chelsea and Westminster Hospital, London, UK; 2 University of Liverpool, Liverpool, UK; 3 Roche, Welwyn, UK
Poster A-1612. 43rd ICAAC- Interscience Conference on Antimicrobial Agents and Chemotherapy. Chicago, September 14–17, 2003
This report contains excerpts from this poster at ICAAC.
INTRODUCTION
The approved dosage of ritonavir-boosted saquinavir (SQV/r) in Europe is SQV/r 1000/100 mg twice daily (BID) (either Fortovase or Invirase). This dosage has shown clinical efficacy in both the MaxCmin1 (vs. indinavir/r) and MaxCmin2 (vs. lopinavir/r) trials.
However, studies have shown that the favorable pharmacokinetic (PK) profile associated with boosted SQV regimens allows for once-daily (OD) administration, which potentially may increase adherence to therapy. The level of treatment adherence has been shown to be one of the most important determinants of virologic response to protease inhibitors (PIs). Reducing the required number of medication doses per day is likely to improve patient adherence and may thus contribute to more prolonged suppression of plasma HIV replication. Central to this approach, however, is the need to maintain PI trough concentrations above the minimum effective concentration (MEC; SQV MEC = 50 ng/mL), in order to maximize ARV activity and minimize the selection of drug-resistant viruses.
OBJECTIVES
To evaluate the steady-state PKs and the safety of two different OD SQV/r regimens, 1600/100 and 2000/100 mg, in HIV-positive patients.
Study design and dosing
This was an open-label, single arm, pilot PK study performed at a single site that enrolled 18 HIV-positive subjects receiving SQV/r 1000/100 mg BID. Three days before baseline (day 1), all subjects on other formulations of SQV were switched to SQV hard-gel (200 mg capsule)/r 1000/100 mg BID, and this SQV formulation was used throughout the study period.
Subjects were given SQV/r 1000/100 mg BID with a 40 g-fat meal on day 1 and were subsequently switched to SQV/r 1600/100 mg OD on day 2 and to SQV/r 2000/100 mg OD on day 12. Protease inhibitors were administered as part of a combination antiretroviral regimen containing two nucleoside/nucleotide reverse transcriptase inhibitors (RTIs). Concomitant administration of non-nucleoside RTIs was not permitted during the study period.
Safety assessment
The safety and tolerability of the study regimens were evaluated throughout the study on the basis of clinical adverse experiences, vital signs, physical examinations and routine laboratory parameters, including liver enzymes and lipid profiles.
Blood collection and drug concentration assay
On day 1, blood was drawn pre-dose and 0.5, 1, 2, 3, 4, 6, 8, 10 and 12 hours following morning and evening doses. On days 11 and 22, blood was drawn at the same time points plus 24 hours post-dose. SQV and RTV plasma concentrations were measured by a fully validated method using high performance liquid chromatography–tandem mass spectrometry (HPLC–MS/MS).
Data analysis
The SQV and RTV PK parameters that were determined were maximum drug concentration (Cmax), trough concentration (Ctrough), half-life (t1/2) and total drug exposure, expressed as the area under the concentration–time curve from 0 to 24 hours (AUC0–24) for all regimens.
RESULTS
The baseline demographic and clinical characteristics of the 18 subjects are illustrated in Table 1.
|
|
| |
| |
 |
|
| |
| |
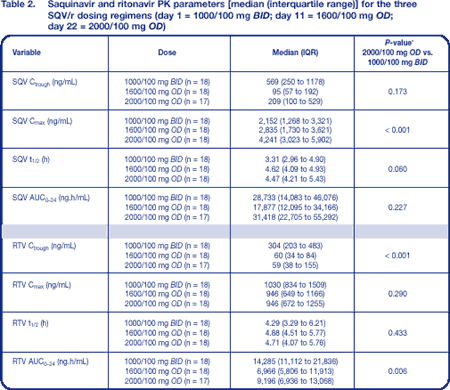
SQV and RTV PK parameters are summarized in Table 2 and median plasma SQV and RTV concentration–time profiles are shown in Figure 2.
|
|
| |
| |
 |
|
| |
| |
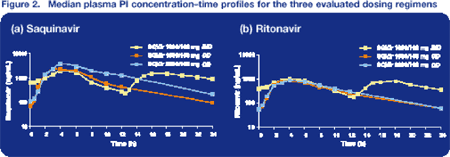
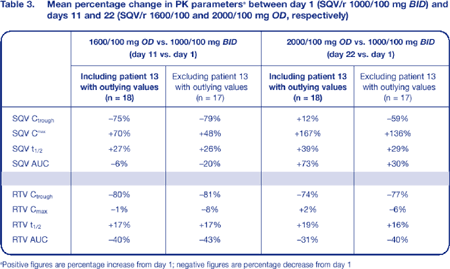
As expected, RTV Ctrough was 80% lower with the OD regimens than with standard BID dosing; despite this, a sufficient SQV-boosting effect was seen. When compared to standard SQV/r 1000/100 mg BID, SQV/r 1600/100 mg OD showed a significant increase in SQV Cmax (70%) and a significant decrease in SQV Ctrough (75%), whereas SQV/r 2000/100 mg OD was associated only with a significant increase in Cmax (167%). There was some evidence of a significant increase in SQV half-life at day 22 compared to day 1 (P = 0.06) (Table 2; Figure 3).
|
|
| |
| |
 |
|
| |
| |
The first column in each of the panels in 1000/100 mg BID dose, followed by 1600/100 mg QD, and 2000/100 mg QD.
In the 1600/100 mg and 2000/100 mg OD regimens, 17/18 and 16/17 subjects, respectively, had SQV Ctrough values above the SQV MEC of 50 ng/mL during the study period.
Interestingly, one participant in this study (subject 13) had a Ctrough of over 3000 ng/mL while on SQV/r 2000/100 mg OD. Therefore, the 12% increase in Ctrough observed with 2000/100 mg OD compared to 1000/100 mg BID was not confirmed when the analysis was performed excluding this subject. Instead, a 59% decrease in SQV Ctrough was measured with the exclusion of subject 13 from the analysis (Table 3; Figure 4).
|
|
| |
| |
 |
|
| |
| |
Reported adverse events that were potentially dose related were mild constipation after switching to SQV/r 1600/100 mg OD (n = 1), mild abdominal pain and mild diarrhea after switching to 2000/100 mg OD (n = 2) and moderate abdominal pain and moderate diarrhea after switching to 2000/100 mg OD (n = 1). No significant changes in ALT, AST, glucose, total cholesterol or triglycerides were observed during the study period (Figure 5).
|
|
| |
| |
 |
|
| |
| |
CONCLUSIONS by authors
- A once-daily dosing regimen of SQV/r 2000/100 mg OD achieved a 30% higher SQV AUC, a 136% higher SQV Cmax, but a 59% lower Ctrough than the standard 1000/100 mg BID dosage (excluding one outlying subject). One subject experienced a Ctrough value below the SQV MEC of 50 ng/mL while on this regimen.
- The PKs of SQV/r 2000/100 mg OD showed that a higher drug exposure was due to increased drug absorption.
- Routine laboratory parameters including lipids and liver enzymes did not change significantly during the study period, suggesting that an increased RTV-boosted SQV dose of 2000 mg OD does not increase risk of toxicity.
- The RTV-boosted SQV dose of 2000 mg OD was here administered for the first time to HIV-positive patients and may be a candidate for new clinical trials of once-daily SQV dosing.
|
|
| |
|
 |
 |
|
|