 |
 |
 |
| |
Preliminary Study of Fosomax for Bone Mineral Loss in HIV
Reported by Jules Levin
|
| |
| |
Researchers at thw Washington University In St Louis School of Medicine (Tebas et al) reported on a study at the 10th Retrovirus Conference (Feb 10-14, Boston, MA) in which they conducted a 48 week prospective, randomized, open-label study to evaluate the effects of alendronate (Fosomax), vitamin D, and calcium suuplementation on bone mineral density in patients with HIV.
Osteopenia and osteoporosis are frequent complications of HIV infection and/or its treatment. Alendronate is the only bisphosphonate approved for the treatmentof osteoporosis in men and women.
Patients were on antiretroviral therapy for a minimum of 6 months. They had evidence of osteopenia/osteoporosis in the lumbar spine (t-score < -1). The patients randomized to receive alendronate or no alendronate:
--70 mg of alendronate weekly for 48 weeks, calcium (1000 mg daily as calcium carbonate), and vitamin D supplementation (400 IU daily) (15 patients in this group)
or
--Vitamin D plus calcium, without alendronate. (16 patients in this group).
The primary endpoint of this study were changes in bone mineral density (BMD). They powered the study to detect 3% changes in BMD in the lumbar region within treatment arms at 48 weeks (30 patients).
Characteristics at baseline for the patients:
87% male
80% white
29% smokers
16% moderate drinkers
61% on PI based regimens
avg age 44 yrs
avg BMI (body mass index) 25 kg/m2
length of HIV-infection: 8 yrs
84% had <400 HIV viral load copies/ml
current CD4 count: 561
avg nadir CD4 count: 167
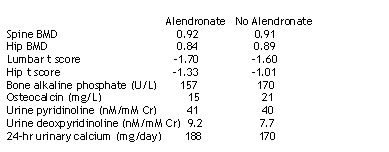
median lumbar t score: -1.52
median hip t score: -1.02
BASELINE
|
|
 |
| |
alcohol (yes/no): 7/8 in alendronate arm, 4/12 in vitaminD+calcium arm
BMI: 25.3 in alendronate arm, 24.6 in vit D+calcium arm
Otherwise, characteristics appeared comparable at baseline between the two study groups.
RESULTS
The average lumbar BMD improved right away in the patient group receiving alendronate much more than the patients in the group not receiving alendronate.
There was 1 discontinuation in each arm for unrelated reasons. There were no serious adverse effects. There were no alterations in LFTs.
The authors concluded alendronate, vitamin D, and calcium are safe and potentially useful in the treatment of osteopenia/osteoporosis associated with HIV. The 48 week lumbar spine BMD change was 5.2% (range 1.3 to 6.4) in patients receiving alendronate, vitamin D, and calcium (the change from baseline was statistically significant (p<.0001), and 1.3% (range -2.4 to 4.0) in the patients receiving vitamin D plus calcium. The difference between the arms was statistically significant (p=.005). These results are consistent with those seen in HIV-negative individuals.
There was a significant reduction in bone alkaline phosphate in the aldronate arm from baseline to week 48: mean change = -31 U/L (p=.001). One patient had elevatedtriglycerides at follow-up.
The authors cautioned that although the study results are positive the study is small and "thou shalt not base therapeutic decisions on small studies"; there were very few women in the study; the study population included patients with osteopenia that might not need treatment; further studies are needed and ACTG 5163 will be look further at this.
The authors also said osteopenia/osteoporosis affects 40-60% of HIV-infected individuals, and suggested the following therapeutic approaches in their opinion: for osteopenia--life style changes (presumably exercise, don't be sedentary), vitamin D and calcium supplementation; for osteoporosis-perhaps alendronate but we're not sure yet.
|
|
| |
|
 |
 |
|
|