| |
Fosamprenavir: new version of amprenavir; GSK Drug Pipeline; herpes & HIV
Reported by Jules Levin
|
| |
| |
GlaxoSmithKline held a community meeting in Asheville, NC for 3 days where they presented new results from studies on various diseases to about 75 attendees; most of whom work in community based organizations as case managers, service providers, and treatment educators.
GSK presented for the first time publicly the latest 48-week update from the CONTEXT Study which compares fosamprenavir (also called "908" for short), a new formulation of amprenavir. The CONTEXT Study examines 908 once and twice daily boosted by low-dose 200 mg ritonavir in comarison to Kaletra in treatment experienced patients. Fosamprenavir is an improved version compared to the older formulation called amprenavir. The pill count is much reduced, 908 pills are 700 mg in contrast to 150 mg pills for the older version of amprenavir. 908 can be taken with or without food or fluid. The GI side effects from the old version were difficult and therefore reduced tolerability. The new version, 908, has much less GI side effects, as you will see from the study data reported below. In addition, results from two other fosamprenavir studies (NEAT & SOLO), which had been previously reported and compared 908 twice daily and once daily to nelfinavir, were reviewed. Below you'll find the data from these 3 studies reviewed. There are several interesting points from these studies:
(1) similarly as seen for Kaletra, GSK researchers have reported that none of the patients taking 908/r QD (0/32) who were evaluated in this resistance substudy in the SOLO study developed primary or secondary protease mutations, while 50% (27/54) of the patients taking NFV did develop primary or secondary protease mutations;
(2) the viral response in the CONTEXT Study to 908/r (1400/200mg) BID was similar to Kaletra, but in these PI-experienced patients the 908/r once daily regimen appeared inferior; this study was designed as a non-inferiority study and the primary endpoint of the study was time-averaged change in viral load (AAUCMB), and the FDA said non-inferiority of 908/r BID or QD compared to Kaletra could not be established at week 48; 908/r taken once daily (1400 mg 908 plus 200 mg ritonavir) appears to be an effective regimen in treatment-naive patients based on the results from the SOLO Study, but based on the results of the CONTEXT Study the twice daily and Kaletra regimens performed better than the 908/r once daily regimen.
(3) in the NEAT and SOLO Studies 908 had less incidence of diarrhea than in patients taking nelfinavir, and GI related side effects for patients taking 908 were mild;
(4) elevations in cholesterol were similar between nelfinavir and 908 (1400 mg twice daily) in the NEAT Study, but LDL and total cholesterol did increase similarly for both groups over the 48 week study; and triglycerides increased more for patients on nelfinavir than on 908. In the SOLO Study where 200 mg of ritonavir was added to 1400 mg of 908 increased the incidence of grade 3/4 triglycerides (6% for patients taking 908)
(5) In the NEAT Study, 908 1400 mg twice daily was compared to nelfinavir 1250 mg twice daily, and as evaluated by percent of patients <400 and <50 copies/ml 908 performed better. In the SOLO Study 908 1400 mg plus 200 mg ritonavir taken once daily was compared to nelfinavir 1250 mg twice daily; as evaluated by percent <400 and <50 copies/ml, nelfinavir responses were better than that seen in the NEAT Study, and 908 responses were similar to that seen in the NEAT Study.
NEW DRUG PIPELINE
Before reporting results from 908 studies here is a brief review of presentations by GSK researchers at the community meeting in Asheville, NC on their new drug pipeline. GSK has active research and development programs for HIV entry inhibitors, an NNRTI for HIV resistant to efavirenz and nevirapine, an NRTI for HIV with NRTI resistance, an integrase inhibitor, a protease inhibitor for HIV with PI resistance, and vaccines for preventing HIV transmission, preventing transmission of genital herpes virus (HSV-2 vaccine), and HPV (human papiloma virus). HPV can lead to cervical and anal cancer.
GSK has two entry inhibitors in preclinical development. For HIV to enter CD4 cells to replicate itself, attachment and fusion must occur. Attachment consists of HIV attaching itself to the CD4 receptor on the CD4 cell and then attachment to a coreceptor (CCR5 or CXR4). GSK has two CCR5 inhibitors in phase I research and reported that it is researching additional CCR5 and CXR4 inhibitors. Both are orally administered and are slated for phase I human studies in healthy volunteers. GSK reported on a next generation NNRTI called "634", which they reported is potent against wild-type HIV and NNRTI-resistant HIV: resistant to efavirenz and nevirapine. They reported preclinical resistance data showing 634 was effective against viruses with the K103N mutation, which is the key NNRTI mutation. These viruses had 2 NNRTI mutations and were highly cross-resistant to efavirenz and nevirapine. 634 is entering phase I human study. GSK's first integrase inhibitor (S-1360) has been stopped due to inadequate potency in HIV+ individuals, but they are moving ahead with another integrase inhibitor that is in late-stage preclinical development (RSC-1838). GW640385X is a next generation protease inhibitor being developed by GSK & Vertex Pharmaceuticals, which GSK reported is potent against wild-type HIV and PI-resistant HIV.
Results from two studies of the GSK HSV-2 Vaccine examining the prevention transmission of genital herpes virus disease were reported. In one study HSV-1 and HSV-2 seronegative partners of HSV-2 infected persons were studied to see if the vaccine prevented transmission. In the second study HSV-1 infected partners of HSV-2 infected persons were studied to see if transmission was prevented. Results appear promising and a follow-up study is planned.. The HIV vaccines are being researched for prophylaxis and therapeutic use. Large preliminary studies are completed and results will be presented at the AIDS vaccine conference in New York City in September, plus they are planning additional studies.
Peter Leone, MD (University of North Carolina) gave an interesting and captivating talk on HSV and HIV. An estimated 60 million people in the USA have herpes, far more than HBV, HCV, HPV, syphilis, and chlamydia. The CDC estimates there are 1 million new genital herpes infections per year. There has been a 32% increase in genital herpes from 1978 to 1990. HSV is a chronic infection, there is no cure. Patients are infected for life and can experience symptomatic recurrences (outbreaks) and subclinical asymptomatic shedding. HSV is poorly recognized and infrequently diagnosed. Many infected individuals are not aware they have herpes. Transmission of HSV can occur when the infected sex partner has outbreaks or asymptomatic shedding. Studies appear to find that herpes-2 increases the risk for HIV acquisition and transmission. Leone reported that HSV-2 infection increases the per-contact acquisition rate of HIV-1 by 4-5 fold, regardless of the source contacts HIV-RNA level. Studies find that HSV-2 may accelerate HIV progression. Leone said this does not appear likely to occur when HAART fully suppresses HIV, but high frequency of HSV reactivation may influence the continued replication of HIV on HAART and influence the increasing prevalence of drug-resistant HIV being transmitted. Condoms reduce the risk for transmission of herpes, but do not eliminate the risk. The higher the CD4 count the less likely viral shedding will occur (31% shedding rate when <200 CD4s, 15% viral shedding rate when CD4s are 200-500, and 6% viral shedding rate when CD4s are >500). Leone reviewed the data reported by GSK at the FDA hearing for Valcyclovir approval to reduce transmission of HSV-2 before the FDA AIDS Drug Advisory Committee. To read a full report by NATAP from this hearing which took place on May 13, 2003, here is the link:
Valtrex For Reducing Transmission of Genital Herpes- FDA Hearing: FDA panel votes 11-0 to recommend approval
http://www.natap.org/2003/may/051503_1.htm
Summary of Valcyclovir data: GSK conducted an 8 month international study for this approval and the study showed that using 500 mg of Valtrex once daily resulted in a 75% reduction in transmission of symptomatic genital herpes infection, and also showed a 48% reduction in the acquisition asymptomatic acquisition of herpes-2 documented by HSV-2 seroconversion during the study. 4 of 743 (0.5%) study participants who received Valcyclovir (Valtrex) developed symptomatic HSV-2 acquisition compared to 16 of 741 (2.2%) study participants who received placebo in the study (p-value: 0.007). This is a 75% reduction. Acquisition of asymptomatic HSV-2 occurred in 14 of 743 (1.9%) persons receiving Valtrex 500 mg once daily compared to 27 of 741 (3.6%) persons receiving placebo who acquired asymptomatic HSV-2 (p-value: 0.038). This is a reduction of 48%. Transmission of HSV can occur during outbreaks of sores and asymptomatic viral shedding. Taking Valcyclovir reduced recurrences of outbreaks: 47% of persons infected with HSV-2 had no recurrences during the 8-month study compared to only 13% of study participants who received placebo in the study. Viral shedding was also reduced by persons taking Valtrex 500 mg once daily in the study. 82% of the study participants had viral shedding on 1 or more days compared to 49% of persons who took Valtrex at this dose who had viral shedding on 1 or more days.
Three FOSAMPRENAVIR (also called 908) Studies
|
|
 |
| |
| Judith Millard, PhD, GlaxoSmithKline, is the lead clinical researcher for
development of fosamprenavir |
| |
| |
GSK researchers presented a review of three fosamprenavir studies, which are eavh described below. In the NEAT and SOLO studies 908 was compared to nelfinavir. In SOLO, 908 plus low-dose ritonavir taken once daily was compared to nelfinavir (Viracept) in treatment-naive patients. The NEAT Study compared 908 twice a day to nelfinavir in treatment-naive patients. Nelfinavir performed better in one of these studies than in the other, as you can see in the data results reported below. The CONTEXT Study compares once and twice daily 908 boosted with low dose ritonavir to Kaletra in treatment-experienced patients.
CONTEXT Study
Vertex Pharmaceuticals reported week 48 results from this study by press release just before the GSK community meeting. GSK researchers reported the following data from this study at the GSK community meeting. In an evaluation of time-averaged change in viral load (AAUCMB), which was the primary endpoint of the study, non-inferiority of 908/r BID and 908/r QD compared to LPV/r BID could not be established at 48 weeks. However, in an analysis of the proportion of patients with viral load below 400 copies/mL, 908/r BID (58%) and LPV/r BID (61%) demonstrated comparable levels of antiviral activity. The proportion of patients who achieved vRNA below 50 copies/mL at 48 weeks was also similar in the 908/r BID (46%) and LPV/r BID (50%) arms.
In the CONTEXT Study PI-experienced 315 patients with current treatment-failure were randomized to:
-- 908/r 1400mg QD plus RTV 200mg QD
-- 908 700mg BID plus RTV 100mg BID
-- Kaletra (lopinavir 400mg plus RTV 100mg BID)
Prior ART Experience
Researchers reported median duration of prior protease inhibitor use was 149 weeks in 908/r QD arm, 149 weeks for 908/r BID arm, and 130 weeks for Kaletea arm. Patients previously taking 2 or more protease inhibitors: 57% for 908/r QD, 49% for 908/r BID, and 40% for Kaletra.
Median duration of prior NRTIs: 234 weeks in 908/r QD, 257 in 908/r BID, 210 weeks in Kaletra group. Percent of patients previously taking 3 or more NRTIs: 70% in 908/r QD, 79% in 908/r BID, and 64% in Kaletra.
Median duration of prior NNRTIs: 87% in 908/r QD, 84% in 908/r BID, and 78% in Kaletra. Percent of patients previously taking 2 or more NNRTIs: 11% for 908/r QD, 14% for 908/r BID, 8% for Kaletra.
There appears to be a trend that patients taking Kaletra had less prior experience with ART drugs.
RESULTS
Mean AAUCMBs at week 48:
-- 908/r QD: -1.49 log
-- 908/r BID: -1.53 log
-- Kaletra BID: -1.76 log
Percent of Patients <400 copies/ml (ITT rebound or discontinuation=failure) at Week 48
-- 908/r QD: 50%
-- 908/r BID: 58%
-- Kaletra BID: 61%
Proportion of Patients <50 copies/ml (ITT RD=F) at Week 48
-- 908/r QD: 37%
-- 908/r BID: 46%
-- Kaletra BID: 50%
|
|
 |
| |
Viral load reduction was similar for patients taking tenofovir and for patients not taking tenofovir, so based on this somewhat crude analysis GSK researchers said this suggests no interaction between tenofovir and 908.
NEAT Study
The NEAT Study examined 251 ART-nave patients who received open-label 908 1400 mg twice daily vs nelfinavir 1250 mg twice daily. All study participants also received abacavir 30 mg twice daily plus 3TC 150 mg twice daily. The patient population in the study was diverse: 25% white, 31% black, 43% hispanic. Patients also received abacavir plus 3TC. Average viral load was 66,000 copies/ml and Cd4 count was 212. 18% had <50 CD4s and 46% had HIV viral load >100,000 copies/ml. Enrollment for the study was 153 patients from the USA, 52 from Panama, 25 from Puerto Rico, and 21 South Africa. 55% of study participants were identified as contracting HIV by heterosexual contact, 39% as men who have sex with men, and 6-10% IDU.13% of patients had hepatitis C and 5% hepatitis B.
RESULTS
In this study efficacy was evaluated by how many patients had viral failure and non viral failure (adverse events) for each study group. More patients taking nelfinavir experienced viral failure (28% vs 14%) by week 48. Viral load rebound was seen in 10% in the 908 group vs 14% in NFV group. 4% prematurely discontinued in 908 group vs 13% in NFV group due to insufficient viral load response.
Non-virologic failure was equal for both arms (20% vs 21%). Failure due to adverse events was equal, 5-6% in each treatment group. Additional reasons for non-viral failure were lost to follow-up, other, consent withdrawn, clinical progression, death, and missing data, and there were no differences between the treatment groups
|
|
 |
| |
Drug-Related Adverse Events
Diarrhea: 18% taking NFV vs 5% taking 908
Drug hypersenstivity: 9% taking 908 vs 5% takinh NFV
Rash: 7% taking 908 vs 2% taking NFV
Nausea: 5% 908; 4% NFV
Vomiting: 2% 908 vs 4% NFV
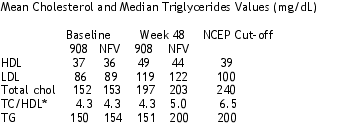
FASTING LIPIDS
There did not appear to be much difference between treatment groups in their effect on cholesterol, but triglycerides increased more in the NFV group. HDL is good cholesterol; LDL is bad cholesterol.
|
|
 |
| |
*total cholesrol-HDL ratio is used to evaluate cardiovascular risk
Grade 3/4 lab abnormalities were comparable between treatment groups: ALT 5-6%, AST 6%, triglycerides 0-1%, cholesterol 0%, glucose <1%-0%.
SOLO Study (908/r Once Daily)
606 treatment-naive patients were randomized equally to 908 1400 mg plus 200 mg ritonavir once daily, or nelfinavir 1250 mg twice daily; all patients also received abacavir 300mg plus 3TC 150 mg.
About 27% of study participants were female; 51% white, 38% black, 7% hispanic. 7% had hepatitis B, 18% had hepatitis C. 21% were CDC class C. Average HIV viral load was about 63,000 copies/ml and CD4 count was 170. 20% had <200 CD4 count.
RESULTS
In contrast to the NEAT Study there were less viral failure in this study in both study groups, 4% in the 908/r QD arm vs 15% in the NFV BID arm. This compares to 14% (908) vs 28% (NFV) in the NEAT Study. 3% had viral rebound in the 908/r arm and 7% in the NFV arm. 1% in the 908 arm never achieved viral load suppression vs 8% in the NFV arm. Prematute discontinuations: adverse events 8% in 908 arm, 5% in NFV arm; lost to follow-up 6% in 908, 3% in NFV; consent withdrawn 5% 908, 3% NFV; other 5% 908, 3% NFV.
Viral Load Response At Week 48
Nelfinavir performed better in SOLO than in NEAT. In NEAT, the patients on NFV had 51% <400 copies/ml and compared to 65% in this study; and patients had 41% <50 copies/ml in NEAT compared to 52% in SOLO.
|
|
 |
| |
CD4 Increase was on average 396 cells for patients on 908/r and 385 for NFV.
RESISTANCE
It appears that a similar story to Kaletra regarding resistance was found for the 908/r QD regimen. No protease mutations were found in patients receiving 908/r who experienced viral failure. A statistically significant difference between the 908/r and NFV arms was observed in terms of protease (0 vs 50%, p<0.0001) and RT (13% vs 56%, p<0.0001) mutations.
At the first time point checked 0/32 patients taking 908/r QD had a primary or secondary mutation detected vs 27/54 patients taking NFV. 4/32 (13%) of patients taking 908/r QD had 3TC resistance vs 30/54 (56%) taking NFV. 0/32 patients taking 908/r had abacavir resistance (K65R, L74V) and 2/54 (6%) taking NFV had abacavir resistance (2 K65R, 1 L74V).
Resistance was evaluated at a later time-point which they called (Last On-therapy Time Point": 0/11 patients taking 908/r QD had primary or secondary protease resistance mutations. 1 patient had the 3TC M184V mutation. 7 patients did not meet the original criteria for analysis (V >1000 c/ml at 2 consecutive time points after being <400 c/ml) and 0/7 had primary and secondary protease mutations. 1/7 developed 3TC mutation. The study researchers concluded that 908/r QD creates a high barrier to the development of resistance.
ADVERSE EVENTS (drug related of at least moderate severity)
Diarrhea: 9% on 908/r QD vs 16% on NFV
Nausea: 7% on 908/r vs 5% on NFV
Vomiting: 6% on 908/r vs 4% on NFV
There were no differences between treatment groups regarding other adverse events: fatigue, headache, increased ALT (2%), abdominal pain (2%), rash (2%).
Grade 3/4 Lab Abnormalities
ALT: 8% in both groups
AST:6-7%
Triglycerides: 6% in 908/r QD group vs 2% in NFV group.
Cholesterol: <1% in 908/r group vs 0 in NFV
Glucose: <1% in 908/r vs <1% in NFV group
Fasting triglycerides: increased from mean levels of 150 mg/dL in 908/r patients at baseline to about 250 mg/dL at week 48. For NFV TG increased from 150 mg/dL at baseline to 200 mg/dL at week 48. NCEP cut-off is 200 mg/dL.
Fasting total cholesterol: mean values increased the same for both groups from 150 mg/dl at baseline to about 220 mg/dL at week 48. NCEP cut-off is 240 mg/dL.
Fasting LDL (bad cholesterol): mean values increased the same for both groups from 100 mg/dL at baseline to about 125 mg/dL at week 48. NCEP cut-off is 160 mg/dL.
HDL Cholesterol: regarding the proportion of patients with (/) 40 mg/dL HDL cholesterol (NCEP cut-off) at week 48. At baseline, 40% taking 908/r and 32% taking NFV had >40 mg/dL; at week 48 75% taking 908/r and 69% taking NFV had >40 mg/dL and 69% taking NFV had
|
|
| |
| |
|
|
|