Written by Donald P. Kotler, MD
St. Luke’s-Roosevelt Hospital Center
Introduction
Of all of the changes that are included in the umbrella-term, ‘lipodystrophy’, fat loss or lipoatrophy has been the most difficult to manage. Treatment options are limited; no therapy has received FDA approval. Several studies have suggested that factors which influence the development of lipoatrophy, and those that promote fat accumulation, are different. The treatments proposed for lipoatrophy and fat accumulation differ as well. Treatments for fat accumulation that have been reported in the literature include diet and exercise, growth hormone (Serostim), and metformin (Glucophage). Treatments for lipoatrophy that have been reported include antiretroviral switches and treatment with thiazolidinediones (glitazones). The published results on these two therapies will be reviewed.
What is normal?
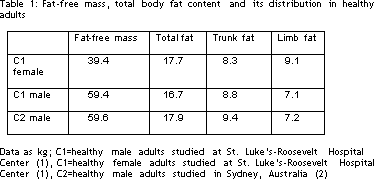
In order to put the effects of therapy in context, it helps to know what is normal fat content and distribution, quantitatively. Table 1 lists average DXA results from two groups of healthy adults. The data from control group C1 were published from our laboratory (1) and include both men and women. The data from control group C2 were taken from the initial publication about lipodystrophy by Carr and colleagues (2). Total fat content is similar in men and women but is higher as a percentage of weight in women (30%) than in men (21%). In contrast, fat-free mass is substantially higher in men, as demonstrated previously by others. There is a difference in the percentage of fat that is central (trunk) and peripheral (limb), with women having more fat peripherally than do men, a difference also shown by others. The results in the men studied by DXA in New York City and Sydney, Australia are very similar. The average man studied in New York has 36.7 pounds of fat, of which 19.4 pounds are in the trunk and 15.6 pounds are in the limbs (6.2 pounds per leg and 1.6 pounds per arm).
How different is body composition in subjects with lipodystrophy?
Table 2 lists average DXA results, at baseline, taken from subjects in three published studies in which abacavir was switched for zerit (3-5). Most or all of the subjects studied were male. Average fat-free mass, when reported, was not substantially different from the controls. Total and limb fat averaged 2.5-5 kg lower in the lipodystrophy groups than in the controls, while trunk fat was slightly higher in subjects with lipodystrophy than in the controls. The ratio of trunk-to-limb fat was about 2.5 times higher in subjects with lipodystrophy than in controls. The absolute amount of fat in the arms and legs were about one half that of the controls.

What is the effect of switching antiretrovirals?
Table 3 lists the absolute and percentage changes in fat-free mass, total fat, trunk fat, and limb fat, again estimated from the published papers, using the patients who switched from zerit to abacavir. The switch did not affect fat free mass in any study. In the study by Carr et al (MITOX), total fat mass rose by around 9%, while limb fat rose by 11% and trunk fat rose by 15% after 6 months (3). The change in limb fat was statistically significant. However, the subjects were unable to detect the change in fat content. The data were updated to a 2 year follow up, during the Fifth Workshop on Lipodystrophy (6). There was continued increase in limb fat totaling a 36% increase from baseline values. However, the changes continue to be clinically inapparent in many patients. In addition, the 36% rise represents about one third of the difference between the subjects at baseline, and controls. In this study, measures of visceral fat did not change, nor did the presence and size of buffalo humps.
John and colleagues from Perth, Australia also studied subjects who switched from zerit to abacavir to prevent or reverse lipoatrophy, and noted an increase in absolute limb fat of about 500 gm over 48 weeks (4). The change, while statistically significant compared to those who continued therapy, is the equivalent of about 4 ounces of fat per limb. No clinical correlations were reported.
Moyle and colleagues from London performed a complicated switch study, and the results in 10 subjects who switched from zerit to abacavir are lilsted in the table (5). Total fat rose by 3.5 kg, or almost 25% after one year. The increase was split relatively equally between trunk and limbs. Because of the loss of limb fat, the increase in limb fat after one year, 1.7 kg, represented almost one half of the difference between baseline and control values. Visceral fat content was unaffected by the switch. Surprisingly, self-assessment and quality of life scores did not change over 48 weeks. One might question the robustness of the measurement tool, though the group size, at 10, is very low for a subjective scale to show significant changes.
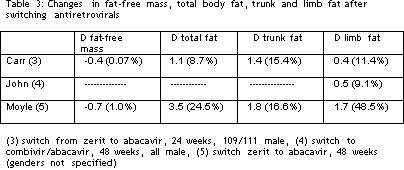
To summarize, switching from zerit to abacavir leads to a statistically significant increase in limb fat with progressive rises over periods up to 2 years. However, the degree of increase does not return limb fat to normal. In addition, there is a relative wide variation in individual results.
What is the effect of therapy with thiazolidinediones?
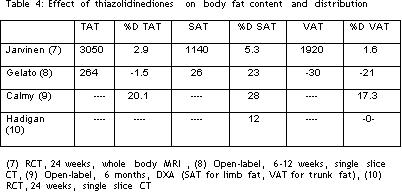
Four studies have reported the effects of therapy with thiazolidinediones, three using rosiglitazone (Avandia) and one using pioglitazone (Actos). Jarvinen and colleagues from Helsinki performed a placebo-controlled trial using 8 mg of rosiglitazone for 6 months, and was unable to show a statistically significant difference in either visceral (VAT) or subcutaneous (SAT) adipose tissue by whole body MRI (7). Gelato and colleagues from Stony Brook performed an open-label study in 8 subjects, and demonstrated a 23% increase in mean SAT and a 21% fall in mean VAT by single slice CT scanning (8). No clinical correlations were reported. Calmy and colleagues from Geneva reported on an open-label trial of pioglitazone for 6 months and documented significant increases in total fat, trunk fat, and leg fat (9). They reported that some subjects noted improvement ranging from modest to substantial, while others noted no change, and some even reported progression of the changes. Hadigan presented the results of a randomized, placebo-controlled trial of rosiglitazone at the recent 5th Workshop on Lipodystrophy. Therapy was placebo-controlled for 3 months, then followed by 3 months of open-label; therapy (10). A 5% increase in leg fat by DXA, which was not statistically significant, was noted at 3 months. However, an increase in abdominal subcutaneous fat of 8%at 3 months and 12% at 6 months were found, and were statistically significant. Trunk fat and VAT did not change. No clinical correlates were reported.
Discussion
Three points are worthy of discussion. The first is that both antiretroviral switching and thiazolidinedione therapy may lead to some reversal of lipoatrophy. The fact that these two completely different approaches may have benefit supports the contention that the development of lipoatrophy is multifactorial, or at least that treatment approaches may be multi-pronged. Furthermore, the results indicate that the process of lipoatrophy is not irreversible.
A wide intra-individual variation in treatment effects was seen in several of the studies, which suggests that medications and other factors variably affect body fat content and distribution in different subjects. In other words, one or another antiretroviral may be the major cause of lipoatrophy in some subjects while other factors, e.g., a rigorous exercise training program plus anabolic steroids may be more important in others. It is important to note that no study reported complete reversal of lipoatrophy, though it might have occurred in some subjects. This author’s experience is similar, in that many patients show little change after switching or even discontinuing antivirals, or with thiazolidinedione therapy, while a few patients have an excellent response.
An unanswered question is how much of a change is noticeable. Several of the studies did not report clinical correlations and others remarked that the increases in limb fat or SAT were unappreciated or labeled as mild by the subject. Subject perception was not a primary endpoint in any study and most of the studies were grossly underpowered for this question. Prospective studies using validated instruments in larger study groups will be needed to determine if the changes in limb fat/SAT after switching antiretrovirals or treating with thiazolidinediones are clinically relevant in addition to being statistically significant.
References
1. Engelson EE, Kotler DP, Tan YX, Wang J, Pierson RN, Heymsfield SB. Fat distribution in HIV-infected patients reporting truncal enlargement quantified by whole-body magnetic resonance imaging. Am J Clin Nutr. 1999;69:1162-9.
2. Carr A, Samaras K, Burton S, et al. A syndrome of peripheral lipodystrophy, hyperlipidaemia and insulin resistance in patients receiving HIV protease inhibitors. AIDS. 1998;12:F51-8.
3. Carr A, Workman C, Smith DE, Hoy J, Hudson J, Doong N, Martin A, Amin J, Freund J, Law M, Cooper DA; Mitochondrial Toxicity (MITOX) Study Group. Abacavir substitution for nucleoside analogs in patients with HIV lipoatrophy: a randomized trial. JAMA. 2002;288:207-15.
4. John M, McKinnon EJ, James IR, Nolan DA, Herrmann SE, Moore CB, White AJ, Mallal SA. Randomized, controlled, 48-week study of switching stavudine and/or protease inhibitors to combivir/abacavir to prevent or reverse lipoatrophy in HIV-infected patients. J Acquir Immune Defic Syndr. 2003;33:29-33.
5. Moyle GJ, Baldwin C, Langroudi B, Mandalia S, Gazzard BG. A 48-week, randomized, open-label comparison of three abacavir-based substitution approaches in the management of dyslipidemia and peripheral lipoatrophy. J Acquir Immune Defic Syndr. 2003;33:22-8.
6. Martin A, Carr A, Ringland C, Amin J, Workman C, Freund J, Hoy J, Doong N, Smith D, Cooper DA for the MITOX study. Long term changes in lipodystrophy after switching from nucleoside analogues to abacavir. Program and abstracts, Fifth International Workshop on Adverse Drug Reactions and Lipodystrophy in HIV. Paris, France, July 8-11, 2003, Abstract 16.
7. Yki-Jarvinen H, Sutinen J, Silveira A, Korsheninnikova E, Fisher RM, Kannisto K, Ehrenborg E, Eriksson P, Hamsten A. Regulation of Plasma PAI-1 Concentrations in HAART-Associated Lipodystrophy During Rosiglitazone Therapy. Arterioscler Thromb Vasc Biol. 2003;23:688-94.
8. Gelato MC, Mynarcik DC, Quick JL, Steigbigel RT, Fuhrer J, Brathwaite CE, Brebbia JS, Wax MR, McNurlan MA. Improved insulin sensitivity and body fat distribution in HIV-infected patients treated with rosiglitazone: a pilot study. J Acquir Immune Defic Syndr. 2002;31:163-70.
9. Calmy A, Hirschel B, Hans D, Karsegard VL, Meier CA. Glitazones in lipodystrophy syndrome induced by highly active antiretroviral therapy. AIDS. 2003;17:770-2.
10. Hadigan C, Yawetz S, Thomas A, Havers F, Sax P, Grinspoon S. A randomized, double-blind, placebo-controlled study of rosiglitazone for patients with HIV lipodystrophy. Program and abstracts, Fifth International Workshop on Adverse Drug Reactions and Lipodystrophy in HIV. Paris, France, July 8-11, 2003, Abstract 12.