| |
Role of Structured Treatment Interruption before a 5-Drug Salvage Antiretroviral Regimen: The Retrogene Study
|
| |
| |
The Journal of Infectious Diseases 2003;188:977-985
Lidia Ruiz,1 Esteban Ribera,2 Anna Bonjoch,1 Joan Romeu,1 Javier Martinez-Picado,1 Roger Paredes,1 Marjorie Díaz,2 Silvia Marfil,1 Eugenia Negredo,1 Julia García-Prado,1 Cristina Tural,1 Guillem Sirera,1 and Bonaventura Clotet1
1Retrovirology Laboratory, IrsiCaixa Foundation and Lluita Contra la SIDA Foundation, Hospital Germans Trias i Pujol, Universitat Autònoma de Barcelona, Badalona, and 2Hospital Vall d'Hebron, Barcelona, Spain
We evaluated the efficacy of a 5-drug salvage regimen, preceded by a 12-week, structured treatment interruption (STI), in 46 multidrug-treated, human immunodeficiency virus type 1infected patients with detectable viremia. Patients were randomly assigned to receive a 5-drug salvage regimen immediately (noninterruption [NI] group; n = 24 patients) or after 12 weeks of STI (interruption [I] group; n = 22 patients). At week 48, 45% of patients in the I group and 46% of patients in the NI group had virus loads <50 HIV-1 RNA copies/mL (P = .619). No differences in CD4 cell counts were seen between groups at week 48 (P = .734). A complete reversion to wild-type genotype was detected in 35% of patients in the I group, but this phenomenon did not affect the virological response. The only overall baseline factor associated with ensuing virus suppression was a lower number of nucleoside reverse-transcriptase inhibitorresistant mutations (relative risk, 0.66; 95% confidence interval, 0.470.93; P = .021). A prior STI seems to confer no additional benefit to subsequent virological or immunological outcomes of a salvage regimen.
Introduction
Both the toxicity associated with short- and long-term antiretroviral therapy and the failure to control human immunodeficiency virus (HIV) type 1 replication with currently available regimens create the need to explore other therapeutic strategies. One such strategy is controlled interruption of the antiretroviral therapy. The objective of structured treatment interruptions (STIs) in multidrug-treated patients who have detectable plasma HIV-1 RNA levels and reduced drug susceptibility is to revert as many drug resistanceassociated mutations as possible and to "resensitize" the HIV quasi species to currently available antiretroviral drugs. For such patients, treatment options are often exhausted because of broad, intraclass drug cross-resistance. In this scenario, the benefits of combinations of 5 antiretroviral drugsknown as "megahighly active antiretroviral therapy" or "multidrug regimens"have been demonstrated extensively for selected patients, although rates of virological failure can vary widely, depending on the characteristics of the patient population and the type of salvage regimen prescribed.
Different studies have evaluated the immunological and virological evolution during STIs in multidrug-treated patients. Overall, these studies concluded that it is possible to achieve transient reversion of the resistant mutations of the reverse transcriptase (RT) and protease (PR) genes with an STI. The decrease in the percentage of mutant viruses that are resistant to antiretroviral drugs in the whole HIV population may improve, in the short term, the success of subsequent salvage therapies in multidrug-treated patients. A recent study by Katlama et al. has suggested that there is a significant benefit of an STI before the initiation of a multidrug salvage therapy in patients with advanced HIV who have experienced multiple virological failure while receiving previous antiretroviral therapies.
However, it is uncertain whether STIs for multidrug-treated patients may have a simple "cosmetic" effect on the predominating virus quasi species. For instance, wild-type HIV subpopulations may regain predominance during STIs but become less dominant thereafter, with the reinstitution of therapy. This phenomenon may occur as a result of the better replicative capacity of previously existing drug-resistant viruses under the new selective drug pressure. In this case, it is unknown whether the initial and transient reversion of drug-resistant mutant viruses to predominantly wild-type populations would exert a durable virological benefit.
In the present study, we evaluated the virological and immunological efficacy of a 5-drug salvage therapy, including a double PR inhibitor (PI), preceded or not by a 12-week STI, in multidrug-treated, HIV-1infected patients who were experiencing virologica failure.
Study design
The Retrogene study was a prospective, 2-arm, open-labeled, randomized, controlled study evaluating the virological and immunological benefit of a combined salvage antiretroviral therapy that included 2 PIs, preceded or not by a 12-week STI. Patients recruited were attending 2 tertiary-care University hospitals in Barcelona, Spain, both of which are national reference centers for the care of HIV-infected patients.
Patients
HIV-infected patients were eligible for enrollment if they had received at least 2 different 3-drug, PI-including combination regimens for at least 6 months, had experienced failure with all previously received treatments, and had 2 consecutive plasma HIV-1 RNA loads >1000 copies/mL. No restrictions were required for CD4 cell counts at the study entry. All patients gave written, informed consent before their inclusion into the study, and the study was approved by the institutional ethical committee of the public, nonprofit, academic institutions of the National Health Service in Spain.
The main exclusion criteria were as follows: not being treated with antiretrovirals at the time of inclusion, an opportunistic infection within the previous 4 weeks, a qualitative serum polymerase chain reaction (PCR) result positive for cytomegalovirus (CMV) before starting the study, pregnancy or lactation, prior treatment with lopinavir/ritonavir (Lpv/Rtv), and inappropriate adherence (defined as the consumption of <95% of medication prescribed within the 2 weeks before the screening visit, as assessed by self-reports) [24].
Patients were randomly assigned to receive a 5-drug salvage regimen without interruption (noninterruption [NI] group) or to interrupt their ongoing treatment and defer the initiation of the 5-drug regimen until 12 weeks later (interruption [I] group). The duration of the STI was established according to previous data showing that primary resistant mutations declined rapidly within 13 weeks of stopping antiretroviral therapy.
The salvage regimen included Lpv/Rtv (400/100 mg twice daily), saquinavir soft-gel capsule (Sqv-SGC; 1000 mg twice daily), lamivudine (150 mg twice daily), abacavir (300 mg twice daily), and didanosine enteric capsule (400 mg once daily for patients with weight 60 kg and 250 mg once daily for patients with weight <60 kg).
The primary therapy-outcome measure was the percentage of patients with a complete virological response, defined as the suppression of plasma HIV-1 RNA to undetectable levels (<50 copies/mL), at 48 weeks. Measurements of secondary efficacy outcomes included a 0.5-log10 copies/mL decrease in virus load (defined as "partial virological response") and changes in CD4 cell count from baseline. For the I group, week -12 was the reference point for the change in HIV-1 RNA load and CD4 T cell count from baseline analysis.
Follow-up
Patients assigned to the NI group were monitored at 3-month intervals during the ensuing 48 weeks, whereas patients in the I group were monitored on a monthly basis during the STI (12 weeks). After the onset of treatment, patients were monitored every 3 months. Plasma HIV-1 RNA levels, CD4 and CD8 cell counts, and adverse events were recorded at each study visit.
For safety purposes, patients in the I group who had a CD4 cell count 200 ¥ 106 cells/L at the time of inclusion received additional monitoring, including a qualitative serum PCR for CMV and an ophthalmoscopic examination at the selection visit; at weeks -12, -8, and -4; at baseline; and at the 3-month visit during treatment.
Plasma HIV-1 RNA load was measured by the ultrasensitive Amplicor assay (version 1.5; limit of detection, 50 copies/mL; Roche Molecular Systems). Peripheral blood CD4 and CD8 T lymphocytes were counted by flow cytometry, using commercial monoclonal antibodies (Becton Dickinson).
All mutation genotypic resistance tests were performed in the coordinating center. For all patients, at day 0 and at the time of virological failure (HIV-1 RNA load >1000 copies/mL), plasma samples were sequenced by use of the TRUGENE HIV-1 Genotyping Kit and the OpenGene automated DNA sequencing system (both from Visible Genetics), as described elsewhere [25]. For patients in the I group, genotypic-resistance testing was also evaluated at the end of the STI period. The classification of drug-resistance mutations was based on the Updated Recommendations of the International AIDS societyUSA mutations panel.
RESULTS
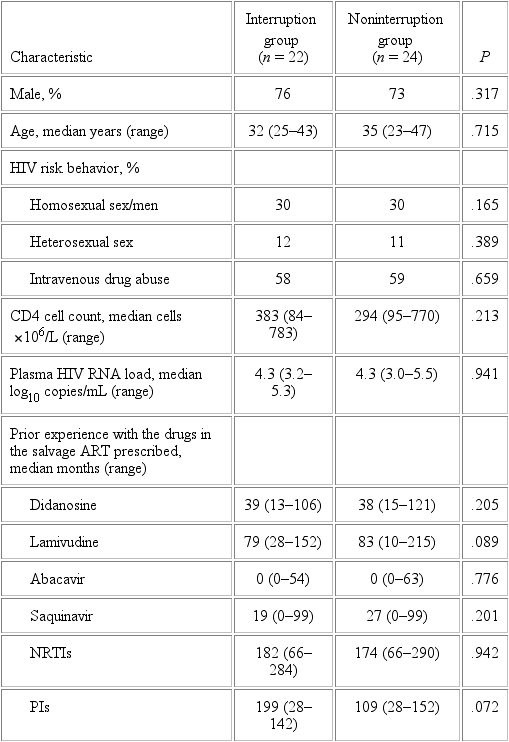
Baseline demographic data
A total of 46 HIV-infected patients were included: 22 patients in the I group and 24 in the NI group. The baseline characteristics of patients in the 2 groups were similar. Overall, the median CD4 cell count was 339 ¥ 106 cells/L (range, 84 x 106 - 783 x 106 cells/L), and the median plasma HIV-1 RNA load was 4.25 log10 copies/mL (range, 3.00-5.54 log10 copies/mL). All patients had been exposed to RT inhibitors (RTIs; nucleosides and nonnucleosides) and PIs. The median number of antiretroviral drugs that had been prescribed before the study entry was 8 (range, 5-13 antiretroviral drugs). The median total time receiving prior antiretroviral therapy was 5.73 years (range, 2.91-14.13 years).
|
| |
| |
 |
|
| |
| |
Virological responses
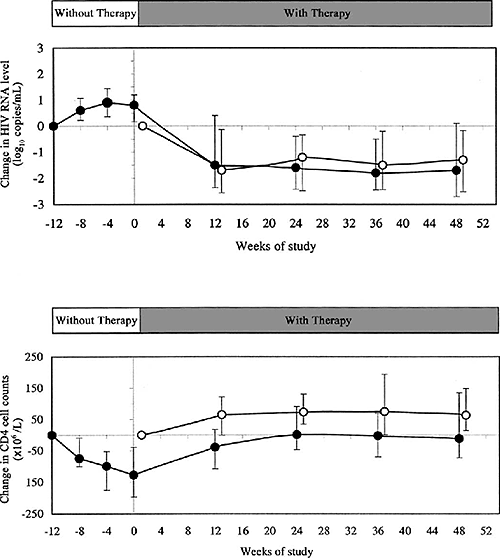
Overall, 21 (46%) of 46 patients achieved a complete virological response (limit of detection, 50 HIV-1 RNA copies/mL) at 48 weeks of follow-up. No statistically significant differences were observed in the proportion of patients with complete HIV-1 RNA suppression between groups (46% and 45% in the NI and I groups, respectively; P = .619). A total of 27 (59%) patients (14 in the NI group and 13 in the I group) had a partial virological response, whereas 19 (41%) of 46 individuals (10 patients in the NI group and 9 in the I group) did not have a virological response to the salvage therapy at week 48. The initiation of the salvage regimen led to a significant reduction in virus load, of 1.52 log10 copies/mL (range, -3.38 to 1.32 log10 copies/mL; P = .002) in the NI group and 1.74 log10 copies/mL (range, -3.48 to 0.88 log10 copies/mL; P < .001) in the I group, at week 12; such responses were sustained until the end of the study (figure 2). In patients from the I group, plasma HIV-1 RNA levels increased a median of 0.77 log10 copies/mL (range, -0.76 to 1.64 log10 copies/mL; P < .001) at the end of the STI, although similar levels of suppression of HIV replication were achieved, compared with those in patients who did not discontinue therapy at week 48 (figure 2).
Immunological responses
At week 48, the median CD4 cell count increase was 63 x 106 cells/L (range, -490 x 106 to 484 x 106 cells/L; P = .012) in patients in the NI group, whereas, in patients in the I group, the median CD4 cell count did not significantly change from baseline values (-11.50 x 106 cells/L; range, -279 x 106 - 506 x 106 cells/L; P = .958). However, no significant differences were found in CD4 cell count between groups at the end of the study (P = .734) (figure 2).
In patients in the I group, there was a significant median decrease in CD4 cell count, of 127 ¥ 106 cells/L (range, -290 x 106 - 70 x 106 cells/L; P < .001) at the end of the STI. However, after resumption of salvage therapy, this decrease in CD4 cell count reversed to the prior baseline levels (figure 2).
Figure 2.
Median changes in plasma human immunodeficiency virus (HIV) RNA levels (top) and CD4 cell counts (bottom). , Interruption group; , noninterruption group. Bars, interquartile ranges.
|
| |
| |
 |
|
| |
| |
Adverse events
Similar rates of adverse events related to the salvage antiretroviral regimen were seen in both groups. Nevertheless, no serious (grade III or IV) adverse events were reported, and, therefore, no patient had to interrupt the study because of drug toxicity. The most frequent adverse events were nausea (3 events in the I group and 1 event in the NI group), vomiting (4 events in the I group and 2 events in the NI group) and diarrhea (1 event in the I group and 3 events in the NI group). In 3 patients, the Sqv-SGC dose was reduced because of persistent vomiting (2 patients received 800 mg twice daily, and 1 patient received 600 mg twice daily).
In the I group, neither adverse clinical events nor deaths occurred during the STI period or after the institution of salvage regimen. Likewise, in the NI group, neither adverse clinical events nor deaths occurred.
Drug-resistance genotypic analysis
Genotypic data were available for 40 of 46 patients included at study entry (week -12 for the I group and week 0 for the NI group). The median number of mutations was similar between both groups: 6 and 5 mutations associated with RTI resistance in the I and NI groups, respectively, and 5 mutations associated with PI resistance in both groups (table 2). In all patients evaluated, drug-resistance mutations to at least 2 of the 3 available drug classes were detected. At study entry, 95% of patients in both groups showed mutations associated with resistance to nucleoside RTIs (NRTIs), 80% showed mutations associated with resistance to nonnucleoside RTIs, and 90% showed mutations associated with resistance to PIs.
The most frequent primary resistance mutations observed at week -12 in the I group were as follows: 41L (70%), 215Y/F (65%), 181C (50%), 210W (50%), 184V (45%), 190A/S (40%), 67N (30%), and 103N (30%) in the RT gene; and M46I/L (50%), L90M (45%), and 82A/F/T/S (30%) in the PR gene. This genotypic pattern was similar to that seen in the NI group at the time of initiating 5-drug salvage therapy. A similar genotypic-resistance pattern, which was already present before the STI, was detected in patients from both groups who experienced virological failure (data not shown).
After the 12-week STI period, the number of resistant mutations detected in the I group significantly diminished, from 6 to 1.50 in the RT gene (P = .005) and from 5 to 1 in the PR gene (P = .004). The most frequent mutations in the I group were as follows: 215Y/F (35%), 41L (30%), and 181C (30%) in RT gene; and M46I/L (20%) and 82A/F/T/S (15%) in the PR gene.
A complete reversion from a drug-resistance genotype to wild-type RT and PR genotypes occurred in 7 (35%) of 20 patients in the I group. In 9 (45%) of those patients, a partial reversion was achieved, whereas, in 4 patients (20%), no reversion to wild-type RT and PR genotypes was detected. In contrast with other reports, no significant differences in virus load or CD4 cell count at study entry were found between patients with and patients without reversion.
Baseline factors related to a better virological response to the salvage therapy. In the univariate analysis of factors related to virological suppression in both study groups, we found that patients with plasma HIV-1 RNA loads <20,000 copies/mL at the time of study entry were more likely to achieve virological suppression at week 48 than were patients with higher HIV-1 RNA loads (P = .026). In addition, patients with virological suppression at the end of the study harbored, at baseline, viruses with a significantly lower number of mutations associated with resistance to RTIs than did patients who experienced virological failure (median, 3.8 vs. 5.8 mutations, respectively; P = .021). However, in the multivariate analysis, we found that the total number of mutations associated with resistance to NRTIs at study entry was the only factor related to virological suppression at week 48 in both treatment arms (relative risk [RR], 0.66; 95% confidence interval [CI], 0.470.93; P = .021).
Other variables (e.g., demographic characteristics, CD4 cell count, reversion to a predominant wild-type population, type of mutation, or duration of prior therapy) were not related to virological response (neither in the univariate nor in the multivariate analysis). Nevertheless, there were no differences in the virological outcome between the I and NI groups, even after stratifying patients according to baseline virus load and number of RT mutations before study entry.
DISCUSSION
Multidrug regimens are an alternative salvage approach applied to patients with persistent viremia and with the presence of multidrug resistance associated mutations. Extensive prior antiretroviral therapy and drug-resistance evolution significantly limit the virological and immunological benefits of salvage regimens. On the basis of recent data suggesting that there is a short-term benefit of a prior shift from mutant to predominantly wild-type virus subpopulations on subsequent salvage regimens in patients with multidrug resistance, we investigated whether an STI provided an additional benefit to a dual PIbased salvage strategy, compared with direct multidrug treatment. Overall, the present study could not demonstrate any significant benefit of an STI on subsequent virological and immunological outcomes of a 5-drug salvage regimen strategy in multidrug-treated patients after 48 weeks of follow-up.
Nevertheless, the sample size in the present study was limited, because few prior data regarding the risk of STIs preceding multidrug salvage regimens were available. This may become a relevant limitation to the statistical power of our analyses; however, a similar study (Community Programs for Clinical Research on AIDS [CPCRA] 064 trial) has recently demonstrated that STIs do not appear to confer clinical, immunological, virological, and quality-of-life benefits to patients with virologic failure and multidrug-resistant virus. Those results, which are based on data from 270 HIV-infected patients, confirm our findings.
Similar to other studies, patients in the present study who interrupted treatment initially showed a significant increase in plasma HIV-1 RNA levels and a significant decrease in CD4 cell counts. However, no differences in virus loads and CD4 cell counts could be demonstrated between groups at weeks 24 and 48 after reinstitution of treatment. More important, although a shift to a predominantly wild-type HIV population was seen at different degrees during the STI period, we found the same genotypic-resistance pattern that was already present before the STI, in patients from both groups experiencing virological failure. This indicates that the HIV quasi species that caused the virological failure of the salvage regimen could be the same as those that predominated before the STI, and might have subsequently reemerged.
Results of a recent open-labeled, multicenter, randomized study by Katlama et al. suggested that there is a benefit from an 8-week STI followed by a multidrug salvage regimen. In their study, the virologic success rate (defined as a decrease of at least 1 log10 in plasma HIV-1 RNA load after 24 weeks of therapy) was 24% and 50% for immediate versus deferred treatment, respectively. Such results suggest that reversion to a less-mutated genotype may be of help in achieving virological suppression.
Conversely, we were not able to find such differences in the present study. Several differences in the design of both studies, regarding the number of patients included and the duration of the STI, may account for the differing results. Those patients included in the present study had higher CD4 cell counts (median, 339 ¥ 106 cells/L vs. 27 ¥ 106 cells/L, respectively) and lower virus loads (median, 4.3 vs. 5.3 log10 HIV-1 RNA copies/mL, respectively) at baseline and, probably, more experience with antiretroviral drugs than did patients in the study by Katlama et al. Moreover, we used a simpler multidrug salvage regimen, and we used the same regimen for all patients. Conversely, in the study conducted by Katlama et al. there was no fixed antiretroviral regimen, and the regimen could include 67 antiretroviral drugs.
Another possible explanation for the divergent results of the present study and that of Katlama et al. could be that some HIV-1 subspecies in our cohort remained at least partially sensitive to the subsequent antiretroviral combination. This also could have occurred in the CPCRA 064 trial, in which the population was more similar to our patients. In this context, an STI before the onset of such a combination may be useless, because the remaining antiviral potency of the salvage regimen may overcome the eventual benefit of a transient treatment withdrawal. Hence, STIs may still be useful for patients with extensive and complete resistance to all antiretroviral drug classes (similar to patients included in the GIGHAART ANRS 097 study).
Indeed, the absence of significant differences in virus load and CD4 cell count responses at week 48, between the groups in the present study, could be explained by the persistence of minority resistant HIV populations after the discontinuation of treatment, for 3 months, in the I group. STIs may significantly contribute to shifting the predominantly drug-resistant virus quasi species to a predominantly wild-type virus population. However, it has been recently demonstrated that, in patients undergoing STIs, highly resistant virus may persist as minor populations and reemerge along with the reintroduction of antiviral pressure. In fact, all patients in the present study who experienced virological failure showed the same genotypic resistancemutation pattern already present at the initiation of the study, regardless of the reversal of predominant mutant variants to wild type, during the STI period. These results suggest that the transient shift to wild-type populations that occurs during the STI does not add further virological control to the subsequent challenge with a 5-drug regimen.
Recent data have shown that even partial virus suppression in patients who remain viremic despite long-term treatment with PI-based therapy reduces CD4 cell turnover and activation, thereby resulting in sustained CD4 cell count increases, and that interruption of treatment in patients with drug-resistant viremia results in increased CD4 cell activation, increased CD4 cell turnover, and substantially decreased CD4 cell counts, which may predispose the patient toward clinical progression. These results emphasize that, in this setting, STIs should be used only in well-controlled trials, not in daily clinical care until more results are obtained.
Remarkably, we observed a significant proportion (46%) of patients with virological suppression, compared with that in other salvage therapy studies. In agreement with the results of other studies, the success of the salvage regimen was associated with a low baseline plasma HIV-1 RNA load and the number of NRTI mutations at baseline. Moreover, this virological benefit could be related to the better immunological status of our patients at baseline, as well as to the use of a dual PI as a part of the multidrug salvage regimen, which has been proven to be associated with a more-favorable virological outcome than single PI-based approaches.
In conclusion, we found no virological and immunological benefit of performing STIs before the initiation of a 5-drug salvage regimen. Therefore, we would not recommend that STIs be used routinely before multidrug salvage approaches in heavily pretreated patients with a virological and immunological status similar to the patients described here.
|
| |
|
|
|