 |
 |
 |
| |
CLINICAL PHARMACOKINETICS OF TELBIVUDINE, A POTENT ANTIVIRAL FOR HEPATITIS B, IN SUBJECTS WITH IMPAIRED HEPATIC OR RENAL FUNCTION
|
| |
| |
Reported by Jules Levin
Xiao J Zhou, Maureen Myers, George Chao, Gloria Dubuc, Nathaniel A Brown, Idenix Pharmaceuticals, Cambridge, MA
Telbivudine (LdT), an L-nucleoside with potent anti-HBV activity, is being evaluated in large randomized clinical trials in patients with chronic hepatitis B. Telbivudine is eliminated predominantly via renal clearance as unchanged drug, with minimal hepatic elimination. Telbivudine pharmacokinetics were investigated in subjects with impaired hepatic or renal function to assess the potential need for dose adjustment in patients with these conditions.
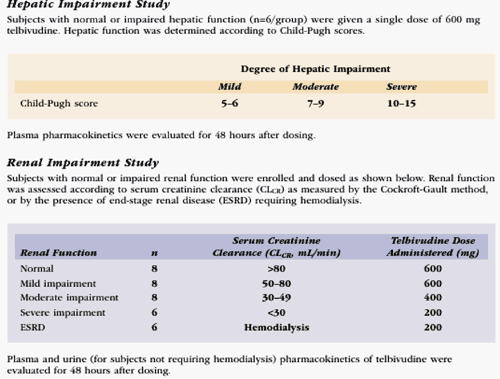
The hepatic impairment study enrolled 24 subjects with normal (n=6) or impaired hepatic function (Child-Pugh score 5-6, 7-9 or 10-15, n=6/category). Subjects received a single dose of telbivudine 600 mg.
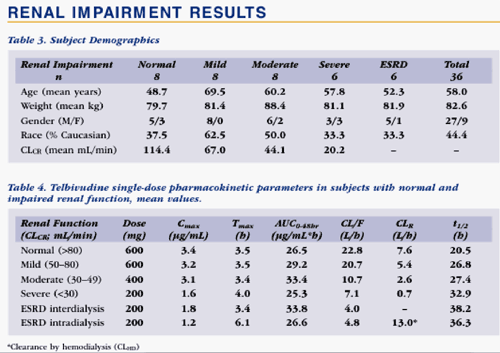
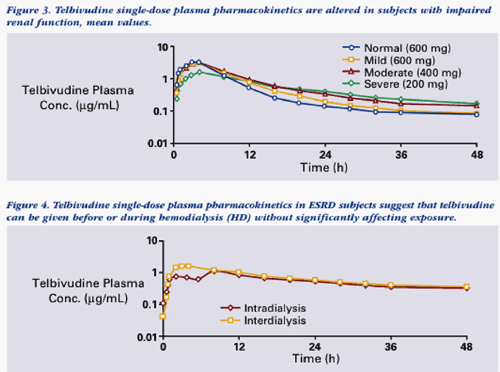
The renal impairment study enrolled 36 subjects with normal or impaired renal function, as defined by serum creatinine clearance (CLCR). Normal: CLCR >80 mL/min, n=8. Mild, moderate or severe impairment: CLCR 50-80 mL/min, n=8; 30-49 mL/min, n=8; <30 mL/min, n=6, respectively. Six subjects with end-stage renal disease (ESRD) requiring hemodialysis were also enrolled. Subjects received a reduced dose in proportion to decreased renal function.
Results
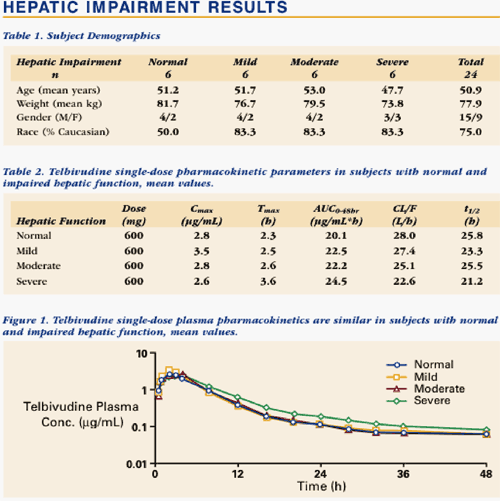
Telbivudine was well tolerated in all subjects. In subjects with impaired hepatic function, telbivudine pharmacokinetics were similar to those in normal subjects with respect to peak (Cmax) and overall drug exposure (AUC).
However in patients with impaired renal function, plasma exposure of telbivudine was dependent upon the degree of impairment. Subjects with moderate or severe renal impairment treated with a single dose of telbivudine 400 or 200 mg, respectively, exhibited overall plasma exposure comparable to that seen in normal subjects, or subjects with mild renal impairment, who were given telbivudine 600 mg. Similarly, ESRD subjects receiving telbivudine 200 mg 2 hours prior to a 4-hour hemodialysis (intra-dialysis dosing) had overall plasma exposure comparable to normal patients treated with a single dose of 600 mg.
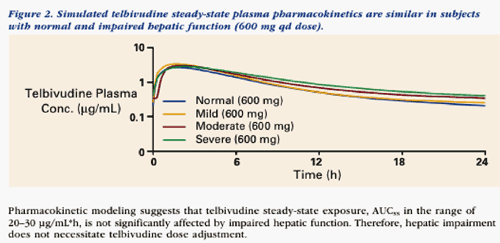
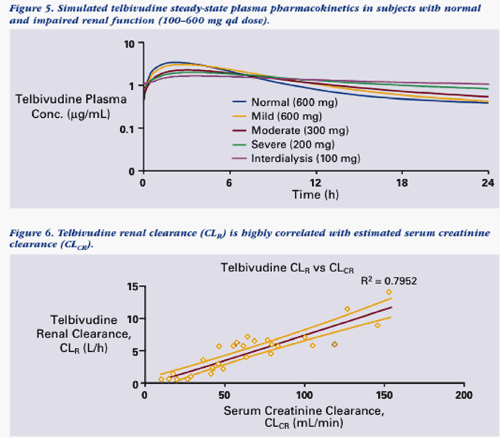
The data from this study can be viewed below & support a rationale for dose adjustment in the range of 100-600 mg qd for patients with varying degrees of renal impairment (moderate or severe) and ESRD patients undergoing hemodialysis. The goal is to maintain consistent daily LdT exposure comparable to that observed in patients with normal renal function who are receiving 600 mg qd (eg, AUC in the range of 20-30 ug/mL*h).
LdT appeared to be well tolerated by all subjects with no clinically significant adverse events or severe AEs reported in either study. No subject discontinued either study due to an AE.
Authors concluded:
--Telbivudine pharmacokinetics were not altered by hepatic dysfunction, suggesting that dose adjustment will not be necessary.
--However, telbivudine clearance was reduced in subjects with impaired renal function, suggesting that adjustment of daily dose will be necessary for treatment of patients with moderate or severe renal impairment, or patients with ESRD receiving hemodialysis.
--PK of LdT 600 mg in subjects with mild renal impairment (CL-CR 50-80 mL/min) are similar to those in subjects with normal renal function, suggesting that patients with mild renal impairment can be treated with the standard 600 mg dose.
--A reduced daily dose is needed in subjects with moderate or severe renal impairment (CL-CR <50 mL/min) and in ESRD subjects requiring hemodialysis to achieve steady-state exposure comparable to that in normal subjects receiving LdT 600 mg once daily.
--Hemodialysis (4 hours) removed about 23% of the LdT dose, suggesting that hemodialysis patients receiving LdT can be managed with a reduced dose given before or during hemodialysis.
|
|
| |
| |
 |
|
| |
| |
|
|
| |
| |
 |
|
| |
| |
|
|
| |
| |
 |
|
| |
| |
|
|
| |
| |
 |
|
| |
| |
|
|
| |
| |
 |
|
| |
| |
|
|
| |
| |
 |
|
| |
| |
|
| |
|
 |
 |
|
|