| |
Anemia in HIV Infection: Clinical Impact and Evidence-Based Management Strategies
|
| |
| |
Clinical Infectious Diseases May 2004
Paul A. Volberding,1, Alexandra M. Levine,2, Douglas Dieterich,4 Donna Mildvan,5 Ronald Mitsuyasu,3 and Michael Saag,6 for the Anemia in HIV Working Groupb
1University of California, San Francisco, and 2Keck School of Medicine, University of Southern California, and 3CARE Center, University of California, Los Angeles; 4Mt. Sinai School of Medicine, New York, New York; 5Beth Israel Deaconess Medical Center, Boston, Massachusetts; and 6University of Alabama at Birmingham
ABSTRACT
Anemia in human immunodeficiency virus (HIV)infected patients can have serious implications, which vary from functional and quality-of-life decrements to an association with disease progression and decreased survival. In 2002, 16 members of the Anemia in HIV Working Group, an expert panel of physicians involved in the care of HIV-infected patients that met first in 1998, reconvened to assess new data and to translate these data into evidence-based treatment guidelines. The group reached consensus on the prevalence of anemia in the highly active antiretroviral therapy era; the risk factors that are independently associated with the development of anemia; the impact of anemia on quality of life, physical functioning, and survival; the impact of the treatment of hepatitis C virus coinfection on anemia in HIV-infected patients; evidence-based guidelines for treatment of anemia in HIV-infected patients, including the therapeutic role of epoetin alfa; and directions for future research.
Received 16 July 2003; accepted 6 December 2003; electronically published 27 April 2004. Financial support: The consensus conference and associated logistics were supported by an educational grant from Ortho Biotech Products. Ortho Biotech Products provided support in convening the meeting and obtained the services of an independent company (The Spellman Group) to assist with transcription and summarization of the meeting discussion.
Anemia is an important clinical problem in patients with HIV infection and those with AIDS. In 1998, the Anemia in HIV Working Group issued a consensus statement addressing the impact of anemia on HIV-infected individuals, as well as treatment strategies and future research directions. The Anemia in HIV Working Group reconvened in 2002 to evaluate recently available data and to determine the implications of those data for patient management.
The consensus statement that follows is based on evidence in the published literature, clinical experience, and the expert opinion of the panel. The chairpersons selected panelists from among the participants in the 1998 Anemia in HIV Working Group meeting and other experts who are involved in HIV study and who specialize in the hematological complications of the disease.
WHAT CAUSES ANEMIA IN HIV-INFECTED PERSONS?
An obvious cause of anemia in patients with HIV infection is blood loss. Blood loss may be associated with such conditions as neoplastic disease (e.g., Kaposi sarcoma in the gastrointestinal tract) or gastrointestinal lesions that accompany opportunistic cytomegalovirus infection. Other than blood loss, the pathophysiology of HIV-associated anemia may involve 3 basic mechanisms: decreased RBC production, increased RBC destruction, and ineffective RBC production.
Decreased RBC production. Decreased RBC production may be a consequence of infiltration of the bone marrow by neoplasm or infection, use of myelosuppressive medications, HIV infection itself, a decreased production of endogenous erythropoietin, a blunted response to erythropoietin, or hypogonadism.
|
|
| |
| |
 |
|
| |
| |
|
|
| |
| |
 |
|
| |
| |
| |
| |
| |
 |
|
| |
| |
Increased RBC destruction (i.e., hemolysis). Increased or premature RBC destruction in the spleen or the circulator system may occur in patients with HIV infection. Hemolytic anemia may result from RBC autoantibodies, hemophagocytic syndrome, disseminated intravascular coagulation, thrombotic thrombocytopenic purpura, or glucose-6-phosphate dehydrogenase deficiency. Hemolysis may also develop as a consequence of the use of various medications.
Ineffective RBC production. Anemia may result from nutritional deficienciesmost commonly, deficiencies in iron, folic acid, or vitamin B12. In patients with HIV disease, folic acid deficiency is generally caused by either dietary deficiency or jejunal pathology. Vitamin B12 deficiency may result from malabsorption in the ileum or from gastric pathology caused by an array of infections or other conditions that affect the gastric mucosa in HIV-infected patients.
WHAT FACTORS ARE ASSOCIATED WITH ANEMIA IN HIV-INFECTED PERSONS?
Individuals with HIV infection who are significantly more likely to develop anemia include women and African American persons. There is also evidence of increased risk of anemia in association with zidovudine use, CD4 cell counts of <200 cells/L, increased virus load, and a number of additional factors.
|
|
| |
| |
 |
|
| |
| |
Sex. In the Anemia Prevalence Study Group, which involves nearly 10,000 patients, there was a 71% greater prevalence of anemia among women than among men when anemia was defined as a hemoglobin level of <12 g/dL in women and of <13 g/dL in men (P <.0001). Both the Women's Interagency HIV Study (WIHS) of 2625 women and the Human Immunodeficiency Virus Epidemiology Research (HER) Study of 1186 women corroborate these findings. Presumably, the increased prevalence of anemia in female HIV-infected persons, compared with male individuals, reflects the overall higher prevalence of anemia in female persons, which may be largely attributed to menstrual blood loss and to the drains on iron stores that occur with pregnancy and delivery.
Race. Multivariate analyses in several major studies, including the Anemia Prevalence Study, the WIHS, and the HER study, found that HIV-positive patients who were African American demonstrated a higher prevalence of anemia than that found in other races. In the most recent report from the Anemia Prevalence Study Group, for example, the prevalence of anemia in HIV-infected persons was 39% among African American women, 19% among white women, 31% among African American men, and 12% among white men (Anemia was defined as a hemoglobin level of <12 g/dL for women and <13 g/dL for men). African American individuals with HIV infection may be at particular risk for the development of anemia, in part as a result of the presence of inherited hematologic disorders, such as sickle cell disease and thalessemia. Dietary factors may also be involved.
Zidovudine treatment. Zidovudine treatment is associated with bone marrow suppression and an increased risk of developing anemia. The HER study demonstrated that, although the use of zidovudine was associated with an increased risk of anemia (defined as a hemoglobin level of <12 g/dL) in the pre-HAART era (19931996), use of zidovudine during the HAART era (19962000) was not significantly associated with anemia. In contrast, the WIHS recorded the presence of anemia (defined as a hemoglobin level of <10 g/dL or a physician's diagnosis) in 41.6% of subjects receiving zidovudine therapy, compared with 34.3% of those not receiving zidovudine (P < .01).
Worsening HIV disease parameters. Low CD4 cell counts (<200 cells/L) and higher HIV-1 RNA levels in plasma have each been independently associated with an increased risk of anemia in multivariate analyses.
WHAT IS THE SIGNIFICANCE OF ANEMIA IN HIV-INFECTED PERSONS?
Although no causal relationship has been documented, retrospective analyses have found an association between anemia at baseline, decreased survival, and increased disease progression in patients with HIV infection. In the EuroSIDA study, for example, the 12-month survival rate was 96.9% among nonanemic patients, compared with 84.1% among patients with anemia at baseline (defined as a hemoglobin level of <12 g/dL for women and of <14 g/dL for men; P < .0001), and it was 59.2% among those with severe anemia (defined as a hemoglobin level of <8 g/dL for both men and women; P < .0001).
In the Multistate Adult and Adolescent Spectrum of HIV Disease Surveillance Project, Sullivan et al. analyzed the medical records of 32,867 individuals with HIV infection. The median duration of survival was significantly shorter in persons with anemia (defined as a hemoglobin level of <10 g/dL) than in those without anemia, regardless of baseline CD4 cell count. Among individuals with CD4 cell counts of 200 cells/L, the relative risk of death was 148% higher in those who developed anemia. Among patients whose baseline CD4 cell counts were <200 cells/L, the risk of death was increased by 56% in the presence of anemia. Survival rates improved markedly among subjects recovering from anemia.
WHAT IS THE IMPACT OF ANEMIA IN HIV-INFECTED PERSONS ON QUALITY OF LIFE AND PHYSICAL FUNCTIONING?
Impact of fatigue. Fatigue is a common symptom of HIV infection and is associated with impaired physical functioning, psychological distress, and decrements in quality of life. Although the etiology of HIV-related fatigue may be multifactorial, anemia is considered an important contributing factor or underlying cause.
In a pre-HAART study of 112 patients (62 of whom were HIV seropositive and 50 of whom were HIV seronegative) by Darko et al., 50% of HIV-positive subjects (who at that time had disease classified as CDC stage IV) reported that fatigue interfered with their daily activities, whereas none of the comparison subjects (whose disease was classified as CDC stage III or who were HIV negative) reported problems with fatigue. Employment problems and sleep disturbances were shown to contribute to morbidity and disability in the HIV-infected group. In a later study of 427 patients by Breitbart et al., 52.7% of subjects responded positively to both the "lack of energy" item on the Memorial Symptom Assessment Scale and the "persistent or frequent fatigue" item on the AIDS-specific Physical Symptom Checklist and were subsequently classified as having fatigue. Serum hemoglobin levels (available from a subset of 176 fatigued patients) were significantly lower than in nonfatigued patients (P < .02). A substudy of 148 patients from the ongoing multinational INITIO trial of 913 antiretroviral therapynaive patients revealed an independent relationship between low baseline levels of hemoglobin and overall quality of life.
Impact of correction of anemia. Correction of anemia in patients with HIV infection has been associated with meaningful improvements in quality of life and physical functioning. Abrams et al. evaluated epoetin alfa treatment (100300 U/kg 3 times per week) in 221 HIV-positive patients with anemia (defined as a hemoglobin level of 11 g/dL). Of these patients, 207 subjects for whom both baseline and follow-up measurements were available demonstrated significant (P < .01) and sustained improvement in hemoglobin levels (mean increase, 2.5 g/dL). Small increases in the hemoglobin level (up to 2 g/dL) were associated with a beneficial effect on total quality of life (as measured by the Functional Assessment of HIV Infection scale), whereas increases of 2 g/dL were significantly associated with greater improvement in quality of life score (P < .05).
In a recent study, Grossman et al. randomly assigned 269 HIV-infected patients with anemia (defined as a hemoglobin level of <12 g/dL) to receive 16 weeks of treatment with epoetin alfa at either 100 U/kg 3 times per week or 40,000 U once per week. Quality of life was measured using 2 instruments: the Linear Analog Scale Assessment (LASA) and the validated Medical Outcomes Study HIV Health Survey (MOS-HIV). Significant increases from the baseline level in mean hemoglobin levels (P < .0001) and in quality of life scores (P < .0001) were demonstrated for both dosage regimens.
In a recently completed study of 709 evaluable HIV-positive patients with anemia (defined as a hemoglobin level of 11 g/dL) who received epoetin alfa (40,000 U once per week), the mean change in hemoglobin level from baseline was statistically significant (P < .05) as early as week 4, with a mean increase of 2.7 g/dL achieved by week 8. Each 1-g/dL increase in hemoglobin level was associated with a 6.4-point increase in the LASA energy/fatigue score, resulting in an overall mean improvement of 41% (P < .001). The MOS-HIV energy/fatigue score improved by a mean of 37% (P < .001). The greatest improvements in quality of life occurred when hemoglobin levels increased from a baseline level of 1113 g/dL.
WHAT IS THE EFFECT OF HAART ON THE PREVALENCE OF ANEMIA IN HIV-INFECTED PERSONS?
Although the prevalence of severe anemia has decreased since the introduction of HAART, mild-to-moderate anemia continues to be common. A subset of 1624 patients was evaluated as part of the EuroSIDA study. Before HAART was initiated, mild anemia (defined as a hemoglobin level of <12 g/dL for women and of <14 g/dL for men) was present in 64% of subjects, and severe anemia (defined as a hemoglobin level of <8 g/dL for both women and men) was present in 1.5% of subjects. After 6 months of HAART therapy, mild anemia was present in 52% of subjects, and severe anemia was present in 1.2%. After 12 months, further improvements were recorded, with 45.6% of patients demonstrating mild anemia and 0.6% demonstrating severe anemia.
Even with use of HAART, anemia remains strongly and consistently associated with HIV disease progression. Studies have shown that, as hemoglobin levels decrease, the risk of disease progression increases. In a EuroSIDA study cohort of 2027 HIV-infected patients, Lundgren et al. examined the relative hazard of clinical progression between patients who were anemic and those with normal hemoglobin levels (>14 g/dL in men and >12 g/dL in women). The relative hazard of clinical progression was 2.2 among men with hemoglobin levels 814 g/dL and women with levels of 812 g/dL. The relative hazard of clinical progression was 7.1 in men and women with hemoglobin levels of <8 g/dL. In a study of 19,213 patients by Sullivan et al., the risk of death increased by 148% in patients with a hemoglobin level of <10 g/dL and a CD4 cell count of 200 cells/L (P < .001). A study of 2348 patients reported by Moore et al. demonstrated an increase in the rate of death of 206% in persons with a hemoglobin level 8.09.4 g/dL and of 290% in those with a hemoglobin level of <6.5 g/dL (P < .001). A 158% increase in the rate of death was reported by Levine et al. in a study of 1525 patients with a hemoglobin level of <12 g/dL (P = .007). Mocroft et al. found similar results, with a 1-g/dL decrease in most-recent hemoglobin measurement resulting in an increased risk of death of 57% (P < .001). In the National Institutes of Healthsponsored WIHS, the development or persistence of anemia in HIV-positive subjects was independently associated with a decreased duration of survival (P < .0001). Determination of whether a cause-and-effect relationship exists between correction of anemia and improved survival is an important area for future research.
WHAT ARE THE CURRENT TREATMENT GUIDELINES FOR ANEMIA IN HIV-INFECTED PERSONS?
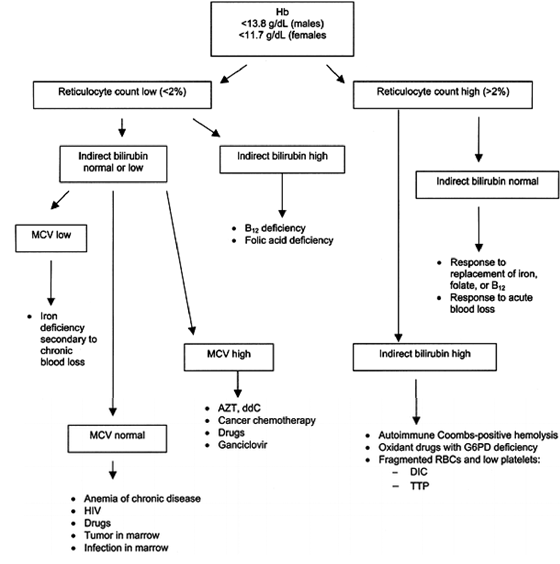
Address correctable causes of anemia. The clinical evaluation of an HIV-infected person with anemia should attempt to identify treatable underlying causes, including hypogonadism. A simplified approach to the assessment of anemia in patients with HIV infection is illustrated in figure 4. To the extent possible, treatable causes should be corrected. In patients whose anemia is severe, transfusion should be considered for alleviation of acute symptoms.
TREATABLE CAUSES OF ANEMIA
| |
| |
| |
 |
|
| |
| |
| |
| |
| |
 |
|
| |
| |
Use of HAART. HAART use may result in improvement of existing anemia. A multivariate analysis of the WIHS study found that HAART was significantly associated with correction of anemia; improvement was noted within 6 months, and a greater resolution occurred after a longer duration of HAART use (P < .0001).
Use of epoetin alfa. In multiple controlled and uncontrolled studies, epoetin alfa has been proven safe and effective for the treatment of anemia in HIV infection. In an early, combined analysis of four 12-week, randomized, double-blind, multicenter, controlled clinical trials, epoetin alfa (100200 U/kg 3 times per week) significantly improved hematocrit levels (P .05) in patients with AIDS who were receiving zidovudine, with endogenous erythropoietin levels of 500 IU/L. An increase of >1 g was seen by week 2, with furthers increases of >2 g by week 4. Treatment was also associated with significant reductions in transfusion requirements (P < .003) and improvements in overall quality of life. As has been described previously, more recent clinical studies of anemic patients with HIV infection have found that epoetin alfa may also be administered once per week (40,000 U), resulting in improvements comparable to those associated with thrice-weekly administration.
Consensus recommendations. The Anemia in HIV Working Group concluded that the following evidence-based treatment strategies should be implemented for anemia in HIV-infected patients:
Monitor hemoglobin levels routinely (e.g., concurrently with each CD4 cell count determination). Ask patients whether they are fatigued and determine whether there is impairment of physical functioning. Assess quality of life on an ongoing basis using such measures as the LASA or MOS-HIV.
If the hemoglobin level is lower than normal (<14 g/dL in men and <12 g/dL in women), or if the patient shows symptoms of anemia, rule out or correct treatable causes.
Initiate HAART if warranted.
If correctable causes of anemia have been ruled out and the hemoglobin level is <13 g/dL in men and <12 g/dL in women, initiate epoetin alfa therapy at a dosage of 40,000 U once per week.
Anticipated benefits of epoetin alfa treatment weighed against its cost must be considered by the clinician in the absence of data from rigorous cost-benefit analyses.
Continue epoetin alfa therapy until symptoms have resolved and hemoglobin levels of 13 g/dL for men or 12 g/dL for women are achieved, then maintain hemoglobin levels by dosage titration or increasing the dosing interval.
WHAT IS THE IMPACT OF TREATMENT FOR HEPATITIS C VIRUS (HCV) INFECTION ON ANEMIA IN HIV-INFECTED PERSONS?
HCV coinfection is estimated to occur in 30% of HIV-infected individuals in the United States. Ribavirin in combination with IFN or pegylated interferon IFN is the standard of care for treatment of HCV infection, but it has been shown to cause anemia, frequently leading to dosage reduction and potentially suboptimal outcomes.
When treating patients with ribavirin-related anemia, hemoglobin levels should be monitored, and epoetin alfa should be added to the treatment regimen in the presence of anemia. In patients with HCV infection alone, epoetin alfa (40,000 U once per week) has been shown to effectively treat anemia associated with ribavirin therapy, to allow maintenance of ribavirin dose, and to reduce discontinuation rates, and it may also improve quality of life. The relative percentage of patients who are able to tolerate optimal doses of ribavirin can be expected to increase by 40%50% with receipt of epoetin alfa therapy. Studies of the utility of epoetin alfa therapy for patients infected with both HIV and HCV are currently in progress.
WHAT ARE THE DIRECTIONS FOR FUTURE RESEARCH ON ANEMIA IN HIV-INFECTED PERSONS?
Future research should focus on furthering understanding of the causes of anemia, its long-term consequences and prognostic importance, the impact of various HAART regimens on the prevalence of anemia, and optimal dosing strategies for the use of epoetin alfa in special populations. Emerging data suggest that epoetin alfa has effects beyond erythropoiesis. There is evidence, for example, that epoetin alfa has antiapoptotic effects in multiple cell lines, which may have a positive impact on the immunologic response in patients with HIV infection. Moreover, in animal studies, epoetin alfa has been shown to cross the blood-brain barrier and to protect neurons and astrocytes from injury. A recent pilot study of patients demonstrated benefit in acute ischemic stroke, with an improvement in clinical outcome at 1 month. It is thus conceivable that epoetin alfa may one day prove useful in the treatment of neurologic conditions, including stroke and cognitive dysfunction.
Cost-benefit analyses are needed to determine the impact of treatment of anemia in HIV-infected patients with epoetin alfa and/or alternative therapies. Factors involved in cost-benefit studies should include identification of the predictors of a response to therapy, determination of optimal doses and schedule of treatment, calculation of the associated costs of treatment, and consideration of any effects of therapy on the natural history of HIV infection.
CONCLUSIONS
Despite use of lower dosages of zidovudine and the introduction of HAART, mild-to-moderate anemia still occurs in a substantial portion of HIV-infected persons and is associated with increased mortality, increased disease progression, and reduced quality of life. Female sex, African American race, medications used for treatment of HIV infection, high HIV-RNA levels, and low CD4 cell count are risk factors for the development of anemia in HIV-infected persons. The Anemia in HIV Working Group agreed that guidelines for the treatment of anemia should be updated. Hemoglobin levels and functional status should be monitored in a systematic manner on an ongoing basis. When anemia is present, treatable causes should be corrected. HAART should be initiated at hemoglobin levels of <14 g/dL for men and <12 g/dL for women if other criteria for institution of HAART are met. If correctable causes have been excluded, epoetin alfa should be added if hemoglobin levels decrease to <13 g/dL in men and <12 g/dL in women or if anemia does not improve after 6 months of HAART. Administration of epoetin alfa once per week is appropriate for the treatment of chronic anemia. Future research on epoetin alfa in HIV-infected patients, beyond its effects on erythropoiesis, is warranted.
THE ANEMIA IN HIV WORKING GROUP
Cochairmen are Paul Volberding (University of California, San Francisco, San Francisco Veterans Affairs Medical Center) and Alexandra Levine (University of Southern California Keck School of Medicine and Norris Cancer Hospital, Los Angeles). Other members of the Working Group are Richard Chaisson (Johns Hopkins University, Baltimore), Terri Creagh (Clinical Epidemiology Research, Atlanta), Douglas Dieterich (Mt. Sinai School of Medicine, New York), Howard Grossman (Columbia University and St. Luke's Roosevelt Hospital Center, New York), Donna Mildvan (Infectious Diseases, Beth Israel Medical Center, and Albert Einstein College of Medicine, New York), Ronald Mitsuyasu (University of California, Los Angeles, CARE Center), Michael Saag (AIDS Outpatient Clinic, University of Alabama at Birmingham), Victoria Sharp (Center for Comprehensive Care, St. Luke'sRoosevelt Hospital Center, New York), Renslow Sherer (Rush Medical Center, Cook County Hospital, Chicago), and Patrick Sullivan (HIV Vaccine Trials Network, Seattle).
|
|
| |
| |
|
|
|