| |
Fuzeon Consensus Recommendations: clinical management with Fuzeon for treatment-experienced patients
|
| |
| |
"Clinical management of treatment-experienced, HIV-infected patients with the fusion inhibitor enfuvirtide: consensus recommendations"
AIDS: Volume 18(8) 21 May 2004 pp 1137-1146
Clotet, Bonaventuraa; Raffi, Françoisb; Cooper, Davidc; Delfraissy, Jean-Françoisd; Lazzarin, Adrianoe; Moyle, Graemef; Rockstroh, Jürgeng; Soriano, Vincenth; Schapiro, Jonathani
From the aHospital Universitari Germans Trias i Pujol and the Institut de Recerca de la SIDA-Caixa Foundation, Badalona, Barcelona, Spain, bCentre Hospitalier Universitaire Hôtel Dieu, Nantes, France, cNational Centre in HIV Epidemiology and Clinical Research, University of New South Wales, Sydney, Australia, dCentre Hospitalier Universitaire Bicêtre, Assistance Publique-Hôpitaux de Paris, Paris, France, eIstituto di Ricovero e Cura a Carattere Scientifico, San Raffaele Vita-Salute University, Milan, Italy, fChelsea and Westminster Hospital, London, UK, the gDepartment of Medicine I, University of Bonn, Bonn, Germany, the hInfectious Diseases Unit, Hospital Carlos III, Madrid, Spain, and iSheba Medical Center, Tel Aviv, Israel.
Received: 29 October 2003; revised: 13 January 2004; accepted: 27 January 2004.
Article Outline
--Use of resistance testing
--Optimal initiation of ENF (Fuzeon)
--Less-optimal initiation
--ENF failure: considerations for discontinuing the...
--ENF safety
--Injection site reactions
--Pneumonia
--Hypersensitivity
--Education, training and support
Abstract
The introduction of enfuvirtide (FUZEON) represents an important advance in the treatment of therapy-experienced patients with HIV-1 infection. However, parenteral self-administration, and the advanced disease and antiretroviral experience of patients currently most needing enfuvirtide introduce unique usage considerations.Enfuvirtide has been shown to provide clinically relevant improvements in CD4 cell counts and reductions in HIV viraemia across all subgroups of treatment-experienced patients studied, including those taking few or no other active drugs. However, optimal outcome results from initiation when the CD4 cell count is above 100 x 106 cells/l and viraemia below 1 x 105 copies/ml, as part of a newly constructed third or fourth antiretroviral regimen in combination with one or two other antiretrovirals to which the virus remains sensitive. Resistance testing should be used where available to guide background drug selection.
Where insufficient options for an effective background exist, enfuvirtide should still be considered and treatment undertaken with the aim of achieving an immunological or clinical response, despite the unlikelihood of a sustained virological outcome. Similarly, where there is no viable alternative treatment, enfuvirtide should be continued following virological failure wherever ongoing immunological or clinical benefit is discerned.
Injection site reactions (ISRs) are common on enfuvirtide and will affect almost all patients. ISRs are manageable and seldom activity- or treatment-limiting. Bacterial pneumonia and systemic hypersensitivity reactions have also been reported uncommonly.
A structured series of patient visits with a healthcare professional provides an atmosphere of ongoing training and support that may prevent 'injection fatigue', maintain adherence and minimise the incidence of ISRs. An initial investment in establishing such procedures can be expected to yield significant returns in patient confidence and benefit on enfuvirtide.
Background
Enfuvirtide (ENF; previously T-20), marketed under the name FUZEON, is a 36-amino acid synthetic peptide inhibitor of gp41-mediated fusion between HIV-1 and the target cell membrane. Administered by subcutaneous injection at a dose of 90 mg (1 ml) twice-daily (b.i.d.), ENF targets the HR1 domain of HIV-1 envelope gp41, preventing binding of the distal HR2 region and subsequent gp41 structural rearrangement that facilitates virus-cell fusion. A potent inhibitor of group M HIV-1 replication in vivo, ENF is inactive against HIV-2. The recent marketing approvals for ENF in several countries followed significant 24-week results from two multinational Phase III studies (TORO 1 and TORO 2) in highly treatment-experienced adult patients from Europe, Australia and the Americas (fig. 1). ENF has also been evaluated in paediatric therapy over 48 weeks. Pharmacokinetic studies in HIV-infected children (aged 6-16 years) have established a paediatric dose of 2 mg/kg b.i.d. (up to the maximum adult dose of 90 mg b.i.d.) to provide comparable exposure to the adult 90 mg dose independent of age group, pubertal stage, body weight or body surface area. There are currently no data on the use of ENF during pregnancy, and insufficient pharmacokinetic information currently exists to establish dosing guidelines for children under 6 years of age.
Fig. 1
|
|
| |
| |
 |
|
| |
| |
Preliminary data suggest that the activity of ENF is essentially unaffected by HIV-1 subtype. There does not appear to be an association between viral co-receptor tropism and virological response in the TORO trials although inconsistent in vitro associations between tropism and susceptibility to ENF have been reported, possibly due in part to differences in receptor density between the various target cells used. The extracellular mode of action and unique target also result in the absence of cross-resistance with other approved antiretrovirals (ARVs), a favourable systemic toxicity profile, and low potential for pharmacokinetic interactions with other ARVs.
These characteristics make ENF an important agent for the increasing number of patients for whom cumulative ARV exposure has resulted in multi-class HIV drug resistance that restricts future treatment options. However, subcutaneous self-administration, and its infrastructure of ongoing patient education and support, introduces challenges for both patients and clinicians.
The pivotal Phase III trials TORO 1 and TORO 2 were open-label studies where treatment-experienced patients were randomized in a 2 : 1 ratio to receive ENF plus an optimized background (OB) regimen of three to five ARVs or the OB alone. In the combined analysis of these studies, the change from baseline to week 48 in plasma HIV-1 RNA was -1.48 log10 copies/ml for patients receiving ENF plus an OB regimen compared with -0.63 log10 copies/ml for patients receiving an OB regimen alone (P < 0.0001; intention to treat [ITT], Last Observation Carried Forward [LOCF]). For CD4, the increase from baseline was 91 x 106 cells/l for patients receiving ENF plus an OB regimen compared with 45 x 106 cells/l for subjects receiving an OB regimen alone (P < 0.0001; ITT, LOCF). The proportion of patients with virological response < 400 copies/ml was more than twofold higher in the ENF + OB group (30% versus 12%; ITT discontinuation and virological failure = failure; P < 0.0001). Benefit on ENF was seen across all subgroups of patients analysed, including patients with no active drugs remaining in the OB regimen (genotypic sensitivity score, GSS = 0).
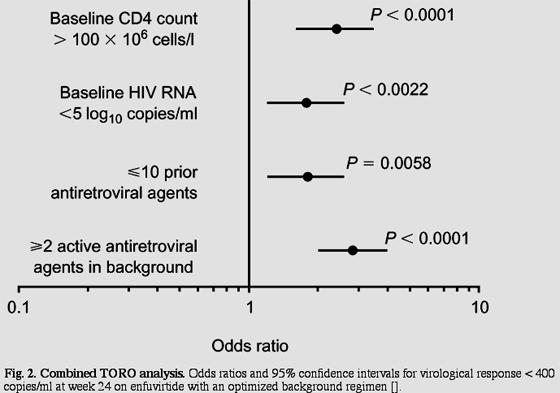
Combined TORO data also identified two or more other active ARVs in the background regimen, baseline viraemia below 1 x 105 RNA copies/ml, baseline CD4 cell count above 100 x 106 cells/l, and prior exposure to no more than 10 ARVs, all to be independently predictive of response below 400 copies/ml on ENF (Fig. 2). Eighty per cent of those with all four of these positive prognostic factors achieved < 400 HIV RNA copies/ml at week 24 in the TORO studies compared with 50% in the control regimen. For the complete study population, week 24-treatment benefit is maintained through week 48.
Fig. 2
|
|
| |
| |
 |
|
| |
| |
If effective and durable therapy is to be achieved on ENF, it is critical that clinicians be aware of how to balance a pragmatic deferral of self-injection against the likelihood of greater success with earlier use. Also, given the broad demography of patients for whom ENF now represents the only potent option, a spectrum of clinical practice should be established for both optimal and less optimal situations. Finally, the novel nature of the drug requires a clear understanding of how to manage adverse events and the supportive educational environment required to ensure its successful introduction. On 17 July 2003, the authors of this paper, all HIV-treating physicians with ENF knowledge and experience, met to discuss and agree on consensus recommendations for the optimal use of this new agent. We present here the results of this meeting, which are therefore not the views of any one individual but the consolidated views of the group.
Use of resistance testing
Resistance testing is recommended for selecting a background regimen for ENF and should be used where such testing is available. Genotyping is more widely available than phenotyping in the clinic or practice setting and should be considered as the first choice. Available clinical evidence does not suggest an additional advantage to combining genotype with phenotype, although evidence does suggest that genotype should be interpreted with the assistance of expert guidance or validated interpretation software.
The unavailability of resistance testing is not a contraindication for ENF use. Where a test cannot be obtained, a full and comprehensive clinical treatment history will also assist drug choice.
Analysis of the combined TORO trials showed a clear association between virological success on ENF and the number of active drugs in the background regimen (Fig. 3). The success rate appears to plateau at two active ARVs plus ENF, supporting the intuitive assumption that ENF will function best as a component of triple-drug highly-active antiretroviral therapy (HAART). However, like other resistance-response analyses of this type, drug sensitivity in TORO was based on a binary sensitive/resistant classification that in practice applies only to the non-nucleoside reverse transcriptase inhibitors (NNRTIs). Since resistance to most nucleoside analogues (NRTIs) and the protease inhibitor (PI) drugs is not binary, a lack of fully active options by resistance testing will not always indicate the lack of an effective combination. Expert advice should be sought where there is uncertainty about possible options.
There is as yet no rationale for basing clinical decisions on assays of ENF susceptibility: such tests are currently research tools. Although a wide range of baseline ENF susceptibility was seen in the TORO trials, no significant association was found between baseline ENF susceptibility and virological response through 24 weeks. While a number of amino acid substitutions at gp41 codons 36-45 are known to emerge in patients with virological failure on ENF their association with clinical response is unknown. Until the clinical relevance of ENF mutations is established, testing for ENF resistance need not be undertaken either pre-treatment or after ENF failure. If such testing is performed the results should be kept for future reference. One should consider storing on-therapy plasma samples in cases of ENF failure, for possible use when more advanced assays or better knowledge of ENF resistance become available.
|
|
| |
| |
 |
|
| |
| |
Optimal initiation of ENF
The optimal time to initiate ENF is when a strong and sustained virological response can be predicted on the basis of treatment history, resistance data and viro-immunological parameters. Maximum response may best be achieved in patients whose virus retains at least partial activity to at least two drugs, ideally from different ARV classes, and who present with CD4 cell counts > 100 x 106 cells/l and plasma viraemia < 1 x 105 copies/ml. While this situation may occur in a wide range of patients, these conditions are most likely to apply to triple-class-experienced patients looking to start a third or fourth regimen following failure of both NNRTI- and PI-based combinations. The precise timing will depend on the specific sequence of agents used and the cumulative pattern of resistance that emerges.
As for any new drug, constructing an OB to combine with the new agent is essential. While residual activity to two or more drugs is preferable, as demonstrated by the previously described multivariate analysis of prognostic factors for week 24 response in TORO, the availability of only a single agent deemed active by resistance testing should not be considered a contraindication for ENF use. Week 48 data from the combined TORO studies indicate that, in the context of a properly optimized OB, the use of ENF with a single active agent yields a response rate roughly comparable to its use with two or more active drugs. This illustrates the intrinsic limitation of considering resistance as a binary proposition, and shows that the use of ENF with a single agent reported as active by genotyping or phenotyping will still yield significant benefit where the regimen is constructed to maximize the remaining activity of the other agents. Some amount of residual antiviral activity may be maintained by a number of NRTIs and/or PIs even when they are scored as resistant by genotyping or phenotyping assays. Combining several such drugs may suffice as an effective background to which ENF can be added, and pharmacokinetic enhancement - such as simultaneous boosting of two PI with minidose ritonavir (r) - might also be used to improve activity. The use of lopinavir (LPV)/r was associated with improved virological outcomes in TORO patients who were LPV/r-naive, and poorer outcomes in those already LPV/r-experienced. This may have relevance for dual-boosting strategies involving LPV/r and another PI. However, since LPV/r was an experimental agent at time of TORO, prior use of LPV/r may have been a marker of greater treatment experience in these patients.
While most patients seeking to start ENF will be triple-class experienced, NNRTI-naive individuals who have already failed more than one prior HAART regimen should also be considered eligible. In combination with NRTIs and/or other agents selected for maximal activity, the use of ENF with an NNRTI in this context is likely to yield stronger and more durable responses than the use of a 'fragile' regimen containing the NNRTI without ENF. This approach is even more relevant where prior exposure to mono- or dual-NRTI therapy occurred before HAAR. The Phase II study T20-206 in PI-experienced, NNRTI-naive patients provides small-scale clinical data in support of this rationale. Similarly, PI-naive individuals with extensive class-resistance to the NRTIs and NNRTIs would also be potential candidates for ENF.
Care should be taken, with patients who already fit the criteria for good outcome, when considering a delay in initiating ENF pending the availability of new agents to use with it. An exception might be made for eligible patients presenting with high (typically > 300 x 106 cells/l) and stable CD4 cell counts for whom delaying therapy may not adversely affect subsequent clinical or virological prognosis. CD4 levels should be closely monitored and ENF initiated, with an OB, with or without a new agent before cells decline to levels associated with an increased risk of clinical events (typically 100-150 x 106 cells/l) and in particular before CD4 cell counts fall below 100 x 106 cells/l.
Less-optimal initiation
Patients presenting with extensive prior treatment and resistance that excludes viable background ARV options will largely not be treatable with the expectation of a sustained virological outcome. These patients fall into the category for whom any antiretroviral treatment would be based on the goal of improving immune and/or clinical status.
Immunological response will ideally consist of stable or increasing CD4 cell counts, but, for those without other options may also encompass a slower rate of CD4 cell count decline relative to the pre-treatment period. Subjective measures, such as improvement in quality of life, may also be valid outcomes for patients with limited clinical prospects.
Interventions to optimize whatever virological activity might be achievable by the background regimen should still be undertaken; through reassessment of treatment history, resistance data, dual-PI boosting, theoretical approaches such as preserving known fitness-reducing mutations like M184V (with lamivudine or emtricitabine), therapeutic drug monitoring, or the inclusion of experimental agents (e.g., tipranavir or TMC114 as PI, capravirine or TMC125 as NNRTI) where available as part of a clinical trial or expanded access initiative.
The importance of employing an OB, even where no activity is reasonably predictable, is highlighted by the modest (+31 x 106 cells/l) week 48 increase in CD4 cell count in the combined TORO trials for those taking an OB alone where the genotypic sensitivity score (GSS) was calculated to be zero -i.e., where no agents in the regimen were considered active by genotypic resistance testing. For those taking ENF on such a background, the CD4 cell increase at 48 weeks was approximately double this (+57 x 106 cells/l; P < 0.05 compared to OB alone), and 8% displayed a week 48 virological response < 400 copies/ml. Both of these CD4 cell count increases are likely to represent a clinically meaningful response in patients with very low baseline CD4 cell counts. The implied contribution of an ostensibly inactive OB to these results provides an appropriate caution against using ENF as true monotherapy (i.e., on its own without a background regimen) even in situations where no other drug appears to be active.
In this setting, even though the use of ENF provides greater benefit, its use will essentially be to prevent disease progression and 'buy time' for prospective new agents. Obtaining any clinical improvement or delay in progression would be indicated in this population. It is important to note that this benefit may be of limited duration.
ENF failure: considerations for discontinuing therapy
In many cases there will not be any effective alternative therapy in treatment experienced patients who fail on an ENF-containing regimen. Therefore virological failure or lack of complete response on ENF may not be an indication to discontinue therapy where immunological or clinical benefit continues. As is the case for the less optimal initiation of ENF described above, readjustment of the background regimen should be undertaken in such cases to potentially improve virological outcome.
There may be a concern regarding accumulating fusion inhibitor resistance if ENF is continued despite continuing viral replication. This could potentially be relevant for future peptide fusion inhibitors or other future agents. However, it is not clear when the second peptide fusion inhibitor will be widely available. Therefore, the possibility of accumulating cross-resistance to potential future fusion inhibitors should not be considered a factor for enfuvirtide discontinuation at this time.
ENF safety
Injection site reactions
The most common ENF-associated adverse event is a spectrum of injection site reactions (ISRs) whose clinical features comprise erythema, induration, nodules and cysts, pruritus, ecchymosis and pain or discomfort. Discomfort is generally of mild-to-moderate severity. Severe discomfort which limits activities or requires analgesia is uncommon (1-3% at any given study visit in the combined TORO trials, 11% cumulative incidence over 48 weeks).
ISRs will occur in almost every patient administering ENF (96% over 48 weeks in the combined TORO studies), but are rarely treatment limiting (4.4% over 48 weeks discontinued for ISRs). There does not appear to be a fixed pattern to their occurrence. Some patients will get ISRs regularly, while for others they may only occur sporadically or only earlier/later in the course of ENF treatment. Both practitioner and patients need to be aware that while ISRs are frequent they normally have little impact on day-to-day activities.
The precise nature of ISRs is still under investigation, though histologically they resemble localized hypersensitivity reactions. Good injection technique (including aseptic technique) in conjunction with rotating injection sites, as outlined in Table 1, may be the most effective way to minimize their incidence and severity, as well as the incidence of associated events, including infections. Conversely, poor technique that leads to intramuscular administration or injecting too rapidly may elevate ISR incidence.
A number of specific interventions, such as post-injection massage of the injection site, have been reported to ameliorate these reactions in some patients. However, specific recommendations are difficult at this time. A basic outline of good ENF injection technique, and various interventions which may alleviate some of the most common injection-related problems, is shown in Table 1. Warning signs for rare but potentially serious ISRs and incipient infections are also listed.
Table 1.
PREVENTION & MANAGEMENT SUGGESTIONS FOR INJECTION SITE REACTIONS & OTHER INJECTION SITE RELATED ADVERSE EVENTS
aEnfuvirtide is adequately absorbed from muscle (animal data) and an intramuscular injection should not be repeated subcutaneously. b45-degree angle is based on average subcutaneous fat deposits. Asthenic or highly lipoatrophic individuals may require a shallower angle of injection. cMay reduce induration, erythema and pain/discomfort. May be valuable for asthenic or lipoatrophic individuals with limited injectable area.
Good Injection Technique
(1) ensure solution is at room temperature
(2) avoid muscle(a) by beveling needle at 45-90 degrees, depending on body habitus(b) (45 degree angle is based on average subcutaneous fat deposits. Asthenic or highly lipoatrophic individuals may require a shallower angle of injection)
(3) inject solution slowly
(4) maintain sterile technique
--wash hands
--barrier gloves may be required for patients in regular contact with dirt or grease (e.g. gardeners, garage mechanics)
--clean injection area and vial caps with alcohol swabs
--never touch needle
(5) Avoid injecting into previous ISR
--feel for hard, subcutaneous bumps
--avoid indurated or erythematous areas
(6) Allow a 5-7.5-cm injection-free zone around the navel
(7) Avoid injections on the belt line
(8) Rotate sites (abdomen, thighs, arms) and never inject two consecutive doses into the same place
Other Interventions for ISR
(1) Gentle manual massage after every injection
(2) Wear loos clothing to avoid irritating sore areas
(3) Divide injection between two different sites(c)
Interventions for Injection Pain
(1) Oral analgesics pre-injection
(2) Topical anaesthetic (e.g., lidocaine gel)
(3) Allow swabbed skin and vial tops to air-dry completely before inserting needle to avoid stinging
(4) Numb area with ice or a cool pack before injecting
(5) Ensure ENF solution is at room tempurature before injection to avoid stinging
Management of Pruritis (itching)
(1) tepid (slightly warm) baths and mild, unscented soaps: oatmeal soap and bath soaks anecdotally associated with improvement
(2) Use mild laundry detergents: reduce chemical laundry additives, e.g., wrinkle reducers, easy-iron rinse additives and fabric softeners
(3) oral antihistamines
(4) emollient creams or lotions (non-alcoholic based and fragrance-free)
(5) adequate oral hydration
Itching Warning Signs that may require medical attention
(1) worsening pruritis unresponsive to simple management measures
(2) swelling & erythema larger than 8.5 cm, warm to the touch and/or draining fluid
(3) induration (injection site gets hard) that limits injectable areas
(4) continuing or worsening pain or discomfort, unresponsive to analgesics, that limit normal activities
Pneumonia
A higher rate of pneumonia (primarily bacterial, both Gram positive and Gram negative) was observed in patients taking ENF in the TORO studies than in those taking OB alone (combined 48-week data: 6.7 cases/100 patient-years versus 0.6/100 patient years).
Pending further data, patients undergoing ENF therapy should be monitored for pneumonia, but reasonable clinical judgement should be exercised since the risk remains very low. Patients with predisposing risk factors for pneumonia (low CD4 cell counts, high viraemia, intravenous drug use, smoking and previous history of lung disease) should be monitored more closely, and antimicrobial prophylaxis in applicable cases should be considered.
Hypersensitivity
Rare cases of reactions considered to indicate systemic hypersensitivity to ENF have been reported (< 1% [five cases] over 48 weeks in the TORO studies). Presentation has included instances of rash, fever, nausea/vomiting, chills, rigours, hypotension and elevated transaminases. Other potentially immune-mediated events have also been reported rarely, including primary immune complex reaction, respiratory distress, glomerulonephritis, and Guillain-Barré syndrome (one case).
Patients developing a reaction suggestive of systemic hypersensitivity should discontinue ENF. In a subsequent medical evaluation, the clinician should consider other, similar syndromes which could have been triggered by other components of the background regimen - for instance, the abacavir hypersensitivity reaction; or rash due to NNRTIs or amprenavir.
Education, training and support
Experience from TORO shows that, with training and support, the majority of patients can learn appropriate injection technique and maintain long-term administration of ENF without significant impact on activities of daily living.A multi-disciplinary approach to patient education and support is highly recommended. All health care professionals (HCP) involved in the care and training of ENF-treated patients, irrespective of previous experience with injectable agents, should undergo training themselves in order to be fully aware of all the relevant issues and how they can be addressed.
Regardless of a patient's prior experience with injectable agents, appropriate counselling and training time should be implemented. Patient training materials - such as a videotape introduction to ENF preparation and use - may facilitate the process. However, these should not replace face-to-face consultations with an HCP. For some patients a structured series of visits should be considered to ensure an environment of ongoing support and assistance. A suggested timeline and structure for these visits is shown in Fig. 4.
Fig 4. SUGGESTED SYRUCTURE & TIMELINE FOR PATIENT EDUCATION & SUPPORT VISITS
Initial patient visit: initial visit 7 day 1may be combined in one visit (repeat visaits may be needed before patient is confident to self-inject without supervision)
COUNSELING
INITIAL PATIENT VISIT
- injection pain
- injection difficulty
- ISRs and their management
- Inconvenience
- Impact on daily activities
- Incorporation into lifestyle: reducing preperation time; use of preparation time
- prior injecting drug users may require additional counseling
DAY 1
TRAINING
- training video
- preparation and storage
- sterile technique
- injection technique: practice aids
- first supervised ENF self-injection
DAY 3
TELEPHONE FOLLOW-UP
DAY 7-14
FIRST PATIENT FOLLOW-UP
- discuss early experience of self-injection
- assess ISRs and advise where necessary
- check used ENF vials
- rough adherence measure
- residual liquid indicates patient is not drawing up full dose
- consider patient self-injection under HCP observation
EVERY 3-4 MONTHS THEREAFTER
Ongoing follow-up
- reassess injection technique
- injection under observation
- retraining if necessary
- discuss experiences and problems
- assess ISR
- exchange new information
- tips on improving convenience
- ISR management
The initial visit is the most important and sufficient time should be allowed for both counselling the patient and training in ENF preparation and self-injection. The HCP should explain ISRs and their management, and review the injection schedule to help the patient determine how best to incorporate twice-daily administration into their existing routine. Time should also be spent addressing concerns over injection and the possible impact of ISRs on appearance. For some patients, more than one meeting will be required to achieve the confidence to self-inject without supervision. In particular, previous injecting drug users may have unpleasant associations with needles or fear relapse.
Although typically it takes around 20 min for ENF to dissolve fully it can take up to 45 min. Patient training should stress the importance of ensuring that ENF is fully dissolved prior to injecting, as well as pointing out simple techniques for minimizing reconstitution time. It is advisable that reconstitution time be spent productively, such as in bathing or preparing a meal, so that ENF administration becomes part of a habitual routine. Table 2 lists a number of simple interventions to reduce reconstitution time. Of these, reconstituting two doses at once is a particularly useful way to minimize the daily time spent preparing ENF.
Table 2. ENSURING RAPID ENFUVERITIDE (ENF) DISSOLUTION
Interventions to minimize ENF preparation time
- Store lyophilized ENF (liquid) and sterile injection water at room temperature
- Tap vial gently before injecting sterile water to loosen ENF powder from the glass
- Inject sterile water down the side of the vial and not directly into ENF powder, as this can cause foaming which lengthens reconsitution time
- Roll the vial between the palms after adding water, ensuring that no drug remains on the vial wall
- Tap vial gently to facilitate reconstitution
- Reconstitute two doses at once, storing the second in the refrigerator until needed (n.b., always store in the vial, never in a syringe. Always bring to room temperature before injecting)
Practice injections can be performed using a simple demonstration aid, such as an orange or a sponge ball. Once confidence has been built by successful practice, the first dose of ENF should be prepared and self-injected under medical supervision and the patient then permitted to perform subsequent injections at home.
Where patients are readily contactable, staff may find it useful to speak to them by telephone after 2-4 days on ENF to assess their progress. With or without a telephone contact, patients should be asked to return for a follow-up assessment after 1 or 2 weeks. Asking patients to return at this time with their used vials allows an assessment of adherence and, if traces of liquid remain in the vials, whether the patient is drawing up the proper dose of medication. At this time inspection of ISRs should be carried out to determine whether there are issues of injection technique or ISR management that should be addressed. The HCP may also wish to re-observe patient self-injection to ensure that good practice is established early.
Subsequent regular follow-up visits should be scheduled every 12-16 weeks (or earlier if indicated by standard of care) even for experienced patients, to monitor ISRs and ensure against 'injection fatigue'. Observation of patient self-injection, with re-training where necessary, should be made a part of this ongoing follow-up. Occasional injection by a partner, friend or family member will help reduce injection fatigue and improve technique, particularly for challenging sites such as the arm. Partner/friend training should be carried out by the HCP and include the use of protective barrier gloves and techniques to avoid needlestick injuries, as well as immediate steps to take if a needlestick injury occurs.
Peer-to-peer support and education are also valuable. Larger practices with a high volume of ENF users may wish to consider the possibility of instigating a 'buddy' system of volunteers to assist new users. Group training may also be effective where disease disclosure is not an issue for the patients concerned.
Finally, these recommendations form only the basic outline of preferred practice, and HCP should be aware that a range of more detailed resources may be available from the manufacturer, local and national government bodies, and organizations such as the Physician Assistant AIDS Network (PAAN), the Association of Nurses in AIDS Care (ANAC), the Australian AIDS Nurses Resource Group (AANRG), and the European Association of Nurses in AIDS Care (EANAC).
Conclusion
In conclusion, ENF is a valuable antiretroviral for highly treatment-experienced patients but one that requires an appreciation of how its unique mode of administration introduces a dimension of patient training and support to successful long-term use. The principle uses of enfuvirtide at present are in 'salvage' patients. For preference, ENF should be used in those for whom sufficient residual drug activity remains to construct a background regimen that allows full virological response. In more treatment-advanced patients, those without effective options at high risk of clinical progression, ENF also represents a means of 'buying time' for new agents. More information is needed on ENF resistance, particularly the genotypic and phenotypic correlates of ENF response. However, until such data are available, and until potentially cross-resistant new agents advance through development, ENF resistance considerations should not influence clinical management.
|
|
| |
| |
|
|
|