| |
Treatment considerations in patients with hepatitis C and cirrhosis
|
| |
| |
J Clin Gastroenterol. 2003 Nov-Dec;37(5):395-8.
E Jenny Heathcote
University Health Network, Toronto Western Hospital, Toronto, Ontario, Canada.
ABSTRACT
Patients with cirrhosis due to hepatitis C have a high chance of dying from progressive liver disease and thus have much to gain from successful
antiviral therapy.
The highest sustained virologic responses in patients with cirrhosis have been achieved using pegylated interferon alfa plus Ribavirin; 43% or more remain with undetectable virus 6 months after the cessation of 48 weeks of treatment.
In those who achieve a sustained virologic response, the degree of fibrosis is less as judged on post-treatment liver biopsy; cirrhosis may even regress. In those individuals with cirrhosis who achieve a sustained virologic response, the risk of developing hepatocellular carcinoma is significantly reduced and it is likely that their chance of developing liver failure is less.
Patients who do not achieve sustained virologic response can still show histologic improvement as demonstrated on liver biopsy post-therapy as compared to baseline.
Patients with compensated cirrhosis can benefit from therapy while those who are decompensated are prone to more safety issues. Thus, individuals with any evidence of hepatic decompensation should generally not be given interferon-based antiviral therapy, but treatment should be encouraged for those whose status is Child Class A.
BACKGROUND
Successful antiviral therapy in patients with cirrhosis due to hepatitis C is potentially lifesaving, but those with cirrhosis are unfortunately a "difficult to treat" patient population. They are difficult to treat because not infrequently they have contraindications to current anti viral therapies, have a high side effect profile, and a lower rate of sustained virologic response to interferon (IFN)-based treatment compared with those without cirrhosis. Liver biopsy plays a vital role in the pre treatment assessment of liver disease severity. Without liver biopsy, the presence of underlying cirrhosis will often go unrecognized.
CONTRAINDICATIONS TO ANTI-VIRAL THERAPY IN CIRRHOSIS CAUSED BY HEPATITIS C
Peripheral Blood Count
Cirrhosis gives rise to portal hypertension that is frequently complicated by features of hypersplenism, specifically thrombocytopenia with or without leukopenia. Although thrombocytopenia may on occasion be immune-mediated in individuals with hepatitis C, it is most often a manifestation of hypersplenism. It is, however, extremely unusual for the platelet count to fall (with or without antiviral therapy) to such an extent that it promotes a bleeding disorder, although easy bruising and gum bleeding (often promoted by periodontal gum disease) complications may occur. Spontaneous episodes of septicemia are
well-recognized in patients with cirrhosis. Such events are thought most often to be secondary to intra- and extrahepatic shunting of bacteria delivered to the liver via the portal vein. Although the precise role of leucopenia in promoting episodes of spontaneous sepsis in patients with cirrhosis remains undefined, IFN has a known bone marrow-suppressive effect and could produce a severe enough neutropenia to put the patient in danger. It is for these reasons that guidelines with regards to minimal acceptable numbers of circulating absolute neutrophils and platelets have been proposed. Most industry-initiated studies have prohibited the start of anti viral therapy in patients with cirrhosis with a platelet count of less than 70 x 106/mL or an absolute neutrophil count of less than 1.5 x 106/mL. In addition, guidelines recommending dose reduction and possible treatment discontinuation if the platelet count falls below 50 x 106/mL or the absolute neutrophil count falls to less than 0.5 x 106/mL. These guidelines have not been formally validated.
Hepatic Decompensation
Although the data are scant, there is good evidence that IFN-based therapy is inadvisable in individuals with decompensated cirrhosis due to hepatitis C.1 Early complications, mostly due to sepsis have been described and such individuals generally tolerate the treatment poorly. In addition, treatment may promote hepatic decompensation.
Tolerance of Antiviral Therapy in Patients With Cirrhosis
Intolerance, particularly due to neuropsychiatric side effect of IFN therapy, has been best described in individuals with cirrhosis due to hepatitis B.2 All forms of IFN therapy may be associated with a wide array of neuropsychiatric side effects. Although never formally examined, it is possible that anti viral therapy in cirrhotics could accentuate subclinical hepatic encephalopathy. The latter, depending on the method of assessment has been reported to be common in otherwise asymptomatic individuals with cirrhosis.3 Recent information suggests that individuals infected with hepatitis C virus (HCV), even in the absence of
underlying cirrhosis, have significant neuropsychiatric deficiencies, particularly in the field of cognition.4, 5 It possible that this may in part explain the poor tolerance of IFN by individuals with hepatitis C.
Efficacy of Anti Viral Efficacy in Cirrhosis caused by Hepatitis C
Sustained Virologic Response
The early studies using standard IFN monotherapy showed disappointing results in patients with cirrhosis.6 When the data from 6 European trials were pooled, the likelihood of a sustained virologic response (undetectable HCV RNA 6 months after completing therapy) was negligible in treatment-naive individuals with cirrhosis infected with HCV genotype 1. The response rates were somewhat improved once the combination of IFN alfa 2b plus ribavirin was introduced. Sustained virologic responses were reported in as many as 20% of patients with cirrhosis infected with HCV genotype 2 or 3.7
Early studies with peginterferon alfa-2a (40KD) (PEGASYS) indicated that this long-acting form of IFN, even when given as monotherapy, markedly enhanced the sustained virologic response in individuals with cirrhosis or bridging fibrosis. In one study that recruited only patients with cirrhosis or bridging fibrosis, the overall sustained virologic response was 30% when peginterferon alfa-2a 180 [million units]g was given once weekly for 48 weeks. This represented a marked improvement over the 8% rate achieved with unpegylated IFN alfa-2a.8 Efficacy was poorest in those infected with HCV genotype 1 (sustained virologic response of 12%), whereas in those with HCV genotype non-1 infections, the sustained virologic response was 51%.
In a large, randomized study, Pegylated IFN alfa-2b 1.5 [million units]/kg weekly (PEGINTRON) plus Ribavirin 800 mg per day for 48 weeks produced a sustained virologic response of 44% in the subset of individuals with bridging fibrosis or cirrhosis: a similar rate of 41% was seen in those treated with IFN alfa-2b plus Ribavirin.9
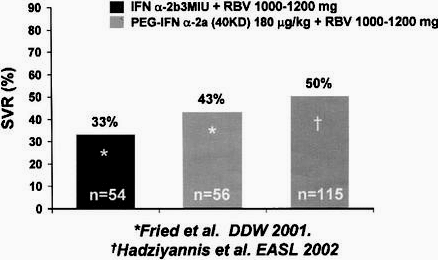
Treatment with peginterferon alfa-2a (40 KD) (PEGASYS) in combination with Ribavirin improves sustained virologic response, relative to standard IFN plus Ribavirin, in patients with cirrhosis. In a recent study, once-once weekly peginterferon alfa-2a 180 [mu]g plus Ribavirin 1000 to 1200 mg per day, administered for 48 weeks, produced a sustained virologic response of 43%; in
another study using the IFN alfa-2b plus Ribavirin the SVR was 33% (Fig. 1).10 Another study using the same peginterferon alfa-2a plus Ribavirin regimen showed a sustained virologic response in 50% of patients with bridging fibrosis or cirrhosis (See Fig. 1).11
figure 1.
|
|
| |
| |
 |
|
| |
| |
Histologic Response
Comparison of posttreatment with pretreatment liver biopsies in individuals who have undergone a course of IFN-based therapy shows an improvement in total histologic activity index (HAI) scores, both in patients with a sustained virologic response and in some who do not clear virus but who experience a fall in viral titer and improvement in liver biochemistry during treatment. Improvements in the necroinflammatory component of this score have generally been greater than the degree of improvement in fibrosis.
In a trial in patients with advanced fibrosis or cirrhosis, 54% of those treated with peginterferon alfa-2a (40KD) (PEGASYS) monotherapy (180 [mu]g once weekly) had an improvement of 2 points or more in their total HAI scores, which was a significant improvement over the 31% histologic response rate seen in patients treated with standard IFN alfa-2a (P = 0.02).8 In addition, the degree of improvement in HAI score was significantly greater (P = 0.02) with peginterferon alfa-2a (40KD) (-2.6 points) than with standard IFN alfa-2a (-0.8).12
A recent study specifically assessed changes in liver fibrosis observed in 153 patients with cirrhosis at baseline who were subsequently treated with either standard IFN alfa-2b plus ribavirin or pegylated IFN alfa-2b plus Ribavirin.13 Regression of fibrosis, assessed by the 5-point METAVIR scoring system, where
0 = no fibrosis and 4 = cirrhosis, was observed in 75 patients. In 23 patients, the METAVIR stage fell by 1 point; in 26 patients, by 2 points; in 23 patients by 3 points; and in 3 patients, no fibrosis was seen on the posttreatment biopsy. Whereas one may question the reliability of a change from 4 to 3, there is little difficulty distinguishing stage 2 from stage 4 using the METAVIR scale. In only 1/3 of patients was this improvement associated with a sustained virologic response. These changes were noted shortly after the cessation of therapy and it is reasonable to assume that further improvement in the degree of fibrosis is likely over longer follow-up periods in patients with a sustained virologic response, as was reported by Shiratori et al.14
Long Term Survival
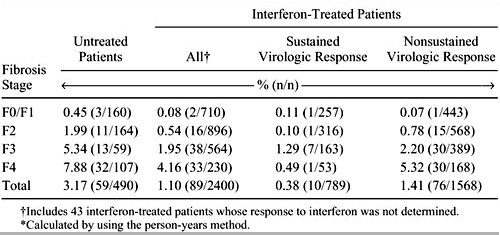
Early reports suggested that IFN-based antiviral therapy, when given to individuals with cirrhosis, did not result in any long-term benefit, as the rate of hepatic decompensation and the incidence of hepatocellular carcinoma were not affected. However, sustained virologic response was rarely achieved in these early studies.6 Subsequently, long-term follow-up of large numbers of treated patients indicated that a significant reduction in the rate of hepatocellular carcinoma was achieved in those individuals with cirrhosis who achieved a sustained virologic response (Table 1).15 Benefit in terms of a reduction in rates of hepatocellular carcinoma may also be seen in patients who have a sustained biochemical response to therapy even though
viremia may persist. Unfortunately, these data were not obtained from long-term follow-up of randomized controlled trials. Data in support of reduced rates of hepatic decompensation in patients with cirrhosis patients treated with IFN-based therapy is hard to interpret for the same reason, (ie, lack of randomization). As IFN therapy for chronic hepatitis C has now been licensed
for a decade, randomization of patients to an untreated control arm in a prospective trial would be considered unethical. Thus, in many long-term follow-up studies, which suggest that death from liver failure is reduced in patients with hepatitis C treated with IFN-based therapy, the comparison group has always been those, who, for whatever reason, did not receive treatment as the
result of some circumstance other than randomization. Such patients may have had contraindications to therapy due to the severity of their liver disease or were denied treatment because of serious comorbidities, (eg, psychiatric conditions). In the study by Fattovich et al,16 individuals treated with IFN had an apparent survival benefit, but when the untreated patients were matched for baseline signs of liver function (eg, bilirubin) no apparent difference in survival was observed between treated and untreated individuals. In another study by Serfaty et al,17 668 patients with compensated cirrhosis due to hepatitis C were
followed for a mean of 40 months. Using multivariate analysis, nontreatment with IFN was the only independent risk factor for both hepatocellular carcinoma and hepatic decompensation.
Table 1. Annual Incidence of Hepatocellular Carcinoma (liver cancer)
|
|
| |
| |
 |
|
| |
| |
In terms of progression to hepatocellular carcinoma and survival, no long-term data are available following treatment with IFN plus ribavirin. As this therapy results in a markedly enhanced rate of sustained virologic response compared with standard IFN monotherapy, and as follow-up studies beyond 2 years suggest that the relapse of viral infection does not occur, it is highly likely that the combination therapy will demonstrate a long-term survival benefit, particularly in individuals who already had developed cirrhosis prior to the onset of antiviral therapy. Therapy based on Pegylated IFNs could be expected to further
improve the outcome; however, information is needed regarding the long-term benefits of treatment with Pegylated IFNs on liver disease progression and survival. The hepatitis C antiviral long-term treatment against cirrhosis trial (HALT-C), an ongoing NIH-sponsored study, is assessing the effect of long-term treatment with peginterferon alfa-2a (40KD) on progression to cirrhosis, hepatocellular carcinoma, and liver transplantation in patients with fibrosis at the start of treatment.18 In the trial, patients with significant hepatic fibrosis who did not respond to previous therapy (IFN with or without ribavirin) are
administered peginterferon alfa-2a (40KD) plus ribavirin for 20 weeks. Those with persistent HCV viremia are subsequently randomized to continue therapy with peginterferon alfa-2a (40KD) alone for an additional 42 months, or to stop treatment. Preliminary data from this trial show that 59 of 138 patients (43%) treated for up to 20 weeks with peginterferon alfa-2a (40KD) plus ribavirin achieved a virologic response, and there was no significant difference between responders and nonresponders in terms of cirrhosis on liver biopsy (32% and 43%, respectively) but we have yet to know whether there will be a difference in the sustained virologic response rates.19
SUMMARY
Despite the fact that individuals with cirrhosis may be more at risk for developing troublesome neuropsychiatric complications and laboratory events such as leukopenia or thrombocytopenia, these patients have the most to gain from successful antiviral therapy. There are good recent data to suggest that the rate of hepatocellular carcinoma is reduced by effective antiviral therapy.
It remains unproven but likely that survival free of liver failure is improved in this particular patient population treated successfully for hepatitis C. Even though antiviral therapy in individuals with cirrhosis due to hepatitis C seems to have a
marked benefit, regular surveillance for liver cancer, portal hypertension and liver failure should probably be maintained lifelong
REFERENCES
- Crippin JS, McCashland T, Terrault N, et al. A pilot study of the tolerability and efficacy of antiviral therapy in hepatitis C virus-infected patients awaiting liver transplantation. Liver Transpl. 2002; 8:350-355.
- Renault PF, Hofnagle JH, Park Y, et al. Psychiatric Complications of long-term Interferon alfa therapy. Arch Int Med. 1987; 147:1577-1580.
- Groeneweg M, Moerland W, Quero JC, et al. Screening of subclinical hepatic encepherlopathy. J Hepatol. 2000; 32:748-753.
- Forton DM, Thomas HC, Murphy CA, et al. Hepatitis C and cognitive impairment in a cohort of patients with mild liver disease. Hepatology. 2002; 35:433-439.
- Hilsabeck RC, Perry W, Hassanein TI. Neuropsychological impairment in patients with chronic hepatitis C. Hepatology. 2002; 35:440-446.
- Valla DC, Chevallier M, Marcellin P, et al. Treatment of hepatitis C virus-related cirrhosis: a randomized, controlled trial of interferon alfa-2b versus no treatment. Hepatology. 1999; 29:1870-1875.
- Schalm SW, Weiland O, Hansen BE, et al. Interferon-ribavirin for chronic hepatitis C with and without cirrhosis: analysis of individual patient data of six controlled trials. Eurohep Study Group for Viral Hepatitis. Gastroenterology. 1999; 117:408-413.
- Heathcote EJ, Shiffman ML, Cooksley WG, et al. Peginterferon alfa-2a in patients with chronic hepatitis C and cirrhosis. N Engl J Med. 2000; 343:1673-1680.
- Manns MP, McHutchison JG, Gordon SC, et al. Peginterferon alfa-2b + ribavirin compared with interferon alfa-2b + ribavirin for initial treatment of chronic hepatitis C: a randomised trial. The Lancet. 2001; 358:958-965.
- Fried MW, Shiffman ML, Reddy KR, et al. Pegylated interferon alfa-2a (Pegasys) in combination with ribavirin: efficacy and safety results from a phase III, randomized, actively controlled, multicenter study [abstract]. Gastroenterology. 2001; 120:A55.
- Hadziyannis SJ, Cheinquer H, Morgan T, et al. Peginterferon alfa 2a (40KD)(PEGASYS) in combination with ribavirin (RBV); efficacy and safety results from a phase III randomized, double-blind, multicentre study examining effect of duration of treatment adn RBV dose [abstract 1]. J Hepatol. 2002; 36( 1): 3.
- Balart LA, Lee SS, Schiffman M, et al. Histologic improvement following treatment with once weekly Pegylated interferon alfa-2A (PEGASYS TM) and thrice weekly interferon alfa-2A (Roferon) in patients with chronic hepatitis C and compensated cirrhosis [plus oral presentation]. Gastroenterology. 2000; 118 (Suppl 2): 961.
- Poynard T, McHutchinson J, Manns M, et al. Impact of Pegylated Interferon alfa-2b and Ribavirin on liver fibrosis in patients with chronic hepatitis C. Gastroenterology. 2002; 122:1303-1313.
- Shiratori Y, Imazeki F, Mariyania M, et al. Histologic improvement of fibrosis in patients with hepatitis C who have a sustained response to Interferon therapy. Ann Int Med. 2000; 132:517.
- Yoshida H, Shiratori Y, Moriyama M, et al. Interferon therapy reduces the risk of hepatocellular carcinoma: national surveillance program of cirrhotic and non-cirrhotic patients with chronic hepatitis C in Japan. Ann Int Med. 1999; 131:174-181.
- Fattovich G, Giustina G, Degos F, et al. Effectiveness of interferon alfa on incidence of hepatocellular carcinoma and decompensation in cirrhosis type C. European Concerted Action on Viral Hepatitis (EUROHEP). J Hepatol. 1997; 27:201-205.
- Serfaty L, Aumaitre H, Chazouilleres O. et al. Determinants of outcome of compensated hepatitis C virus-related cirrhosis. Hepatology. 1998; 27:1435-1440.
- Di Bisceglie AM. BOnkovsky HL, Deinstag JL, et al. Design of HALT-C trial (hepatitis C antiviral long-term treatment to prevent cirrhosis) [abstract]. Gastroenterology. 2000; 118 (Suppl. 2): 1435.
- Shiffman ML. Retreatment of interferon and interferon-ribavirin non-responders with peginterferon alpha-2a and ribavirin: Initial results from the lead-phase of the HALT-C. Hepatology. 2001; 34:A243
|
|
| |
| |
|
|
|