| |
Pilot Study of the Relationship Between Histologic Progression and Hepatic Iron Concentration in Chronic Hepatitis C
|
| |
| |
Journal of Clinical Gastroenterology 2003; 37(5):406-411
Anne M. Larson, MD; Shari L. Taylor, MD; Donald Bauermeister, MD; Leonard Rosoff, Jr., MD; Kris V. Kowdley, MD
From the Department of Internal Medicine, Division of Gastroenterology, University of Washington, Seattle WA (Drs. Larson and Kowdley); GI Pathology Partners, Memphis TN (Dr. Taylor); Department of Pathology, Virginia Mason Medical Center, Seattle WA (Dr. Bauermeister); and Department of Internal Medicine, Division of Gastroenterology, Virginia Mason Medical Center, Seattle WA (Dr. Rosoff).
Comments by Jules Levin: there was no discussion about eating foods high in iron such as red meat and iron overload in this article. You can have iron levels checked, but experts I speak with do not feel there is any reason to avoid red meat in diet unless perhaps if HCV disease is very advanced, decompensated cirrhosis; in which consultation with your doctor about this is advisable.
Abstract:
Hepatic iron deposition is common in patients with chronic hepatitis C (HCV) and may play a role in progression of liver disease. This pilot study examines the relationship between hepatic iron concentration (HIC) and histologic progression over time in patients with HCV. HIC was retrospectively measured in 14 patients with HCV who had 2 serial liver biopsies prior to the era of interferon therapy. The mean interval between biopsies was 56 ± 46 months. Mean Knodell score worsened between first and second biopsies (10.0 ± 2.8 versus 12.4 ± 3.3; P = 0.007). There was increased portal inflammation (3.2 ± 0.4 versus 3.6 ± 0.5; P = 0.028) and fibrosis (1.8 ± 1.3 versus 2.7 ± 1.2; P = 0.002), but no significant change in piecemeal necrosis or lobular degeneration. There was no significant change in HIC between first and second biopsy (P = 0.66). However, HIC was noted to increase significantly among patients with cirrhosis on initial biopsy or those who progressed to cirrhosis (P = 0.009). In this pilot study, histologic progression in patients with precirrhotic HCV was not associated with an increase in HIC, whereas hepatic iron accumulation was observed in 3 patients once cirrhosis was present. The interaction between progression of hepatitis C and iron deposition warrants further study.
The liver is the primary site of iron storage and the only site for synthesis of transferrin (the plasma iron transport protein) and ferritin. Free ferrous iron is highly toxic and normally is protein-bound within the liver. Unbound, iron catalyzes the production of free radicals, which have been implicated in lipid peroxidation and hepatotoxicity. Lipid peroxidation may be the primary event causing hepatocellular injury secondary to iron overload.
Cross-sectional studies have suggested that there is a positive correlation between body iron stores and disease severity in HCV. Patients with chronic HCV who have precirrhotic disease generally have normal HIC, whereas HCV patients with end stage liver disease have increased transferrin saturation and HIC, particularly when compared with patients with end stage liver disease from other causes of chronic hepatitis. Thus, it is possible that progressive hepatic iron deposition may lead to increased disease severity in HCV. However, no previous studies have examined whether histologic progression in HCV is associated with an increase in HIC.
The purpose of this pilot study is to determine whether HIC increased over time in chronic hepatitis C infection and whether change in histology correlates with change in HIC.
Patients
Fourteen adult patients with chronic HCV infection, referred to a tertiary care center's hepatology clinic, who underwent paired liver biopsies were retrospectively identified. HCV infection was confirmed by finding HCV-RNA in serum. All patients had persistently elevated serum aminotransferases (ALT) for at least 1 year prior to liver biopsy. Liver biopsy revealed chronic hepatitis with variable levels of activity in all patients. All patients were negative for hepatitis B surface antigen. None were treated with interferon, ribavirin, or other antiviral agents (the patients had undergone liver biopsy prior to the availability of these therapies). Second biopsies were performed as part of ongoing clinical care by the hepatologist caring for the patient. However, in no case was the second biopsy done because of a concern about iron overload. A few had undergone a short course of corticosteroid therapy between their first and second biopsies. The medical record of each patient was reviewed for demographic data, route of transmission, laboratory data, and treatment history.
Liver Biopsy
Percutaneous needle biopsies of the liver were obtained on each patient at 2 separate time points ranging from 14 to 182 months. The mean interval between first and second biopsy was 56 ± 46 months. Biopsies were performed in the right lobe of the liver using the Menghini aspiration method with a 16-gauge Klatskin needle. 11 The liver tissue specimens were all more than 2 cm in length and contained at least 6 portal tracts.
RESULTS
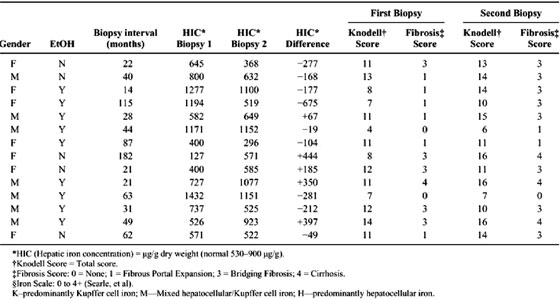
The 14 patients ranged in age from 28 to 66 years (mean 42) at the time of first biopsy; the mean age for males was 41 ± 10 years and for females, 43 ± 13 years (P = 0.77). Nine patients reported alcohol use of over 20 g a day; the remaining 5 reported rare or no use of alcohol. Risk factors for hepatitis C included transfusions (21%), intravenous drug use (22%), multiple (29%; intravenous drug use; tattoo, transfusion), unknown (21%), and sexual transmission (7%).
There was a trend toward lower serum albumin and higher prothrombin time between the first and the second biopsy, although both values remained within the normal range. There was no significant difference in alkaline phosphatase, serum bilirubin, or serum aminotransferases (AST, ALT) between the first and second biopsies. Serum iron studies were not available in enough patients to make a meaningful determination. No patient was receiving iron-containing medications.
The mean interval between biopsies was 56 ± 46 months (range 14 to 182). There was an increase in overall Knodell score between the first (10.0 ± 2.8) and second (12.4 ± 3.3) biopsies (P = 0.007), reflecting increases in portal inflammation and fibrosis. There was no significant change in mean HIC (756 ± 380 versus 719 ± 299 [mu]g/g dry weight; P = 0.659) or in HII (0.4 ± 0.2 versus 0.3 ± 0.2; P = 0.236) between first and second biopsies. The interval between biopsies was similar between those whose HIC increased and those whose HIC decreased (60 ± 69 versus 53 ± 33; P = 0.795). There was also no relationship between overall Knodell score, or individual components thereof, and HIC. However, HIC did increase significantly among patients with cirrhosis (P = 0.0014). An increase in HIC of 350 [mu]g/g dry weight between the first and second biopsy was seen in the patient with cirrhosis on both biopsies. Likewise, the two patients who progressed to cirrhosis showed increased HIC (444 [mu]g/g dry weight and 397 [mu]g/g dry weight, respectively; P = 0.00001). There was no increase in HIC among patients with fibrosis grades lower than 3 (bridging fibrosis) or among patients with bridging fibrosis who did not progress to cirrhosis (P = 0.255). Five patients had bridging fibrosis and one had cirrhosis on the first biopsy. Eight patients had bridging fibrosis, and three had cirrhosis on second biopsy. Among noncirrhotics, there was no correlation between change in HIC and change in fibrosis score between biopsies when controlled for patient age, interval between biopsies, gender, corticosteroid use, and alcohol use (P = 0.710; CI -559.4 418.5). Among cirrhotics, however, there was a significant correlation between changes in HIC between biopsies (P = 0.009; CI 200.3 966.5).
|
|
| |
| |
 |
|
| |
| |
Eight of the 11 patients (73%) had stainable iron in at least one of the two biopsies. Iron deposition was primarily within Kupffer cells in the majority of patients (5 of 8); the pattern of iron staining was mixed in the remainder. In most cases, the iron deposition was mild (grade 1-2+); only one patient had grade 3+ iron deposition and this was in a pattern involving hepatocytes and reticuloendothelial cells. HIC and HII did not approach ranges associated with hereditary hemochromatosis in any case. In 6 of 11 patients (55%), there was no change in stainable iron between the first and second biopsies. In 4 of 11 patients, there was a decrease in stainable iron. The stainable iron increased in only one patient. Knodell score worsened in 10 of 14, was unchanged in 2 of 14, and improved in 2 of 14. There was no relationship between change in total Knodell score or fibrosis score and change in hepatic iron staining. There was also no correlation between change in total Knodell score and change in HIC.
Alcohol use was reported by 9 of the 14 patients (64%). Patient characteristics were similar between those who used alcohol and those who did not, except the serum total bilirubin was higher in those who were using alcohol (P = 0.040). A greater proportion of males reported alcohol use (P = 0.091). We did not find a difference in Knodell score at first or second biopsy between patients reporting alcohol use and those without. Those with alcohol use tended to have more stainable hepatic iron that those without alcohol use (median iron staining score 1 + v 0; P = 0.126). Those with alcohol use also tended to have a greater HIC, although this was not significant and the absolute level remained within the normal range. Neither alcohol users nor non-users showed a significant difference in the change in HIC between biopsies (P = 0.583).
DISCUSSION
Hepatic iron overload may be a cofactor in the expression of many liver diseases. Hepatic iron deposition in patients with alcoholic liver disease promotes hepatocellular damage. Increased body iron stores were shown to be associated with increased risk of chronicity among persons exposed to hepatitis B. Chronic hepatitis C virus infection has been associated with mild to moderate liver iron loading. Serum ferritin, serum iron, and transferrin saturation also seem to be increased in some patients (especially men) with chronic HCV, and there is a positive association between serum ferritin concentrations and serum ALT level in this disease.
It has been postulated that iron overload may exacerbate inflammation and fibrosis in chronic HCV. However, there is controversy regarding the prevalence and severity of hepatic iron overload in hepatitis C. Several investigators have reported that hepatic iron stores are usually normal or only mildly increased, and generally remain within the "normal" range throughout the course of disease. Others have found a significantly higher HIC in chronic HCV when compared with controls. Patients who respond to interferon-alpha have lower HIC than those who do not have a response. Phlebotomy has also been shown to reduce serum aminotransferase levels in patients with chronic HCV. Furthermore, HIC may be significantly increased in end-stage liver disease associated with HCV, with levels occasionally in the range approaching hereditary hemochromatosis. Thus, the evidence seems to support a relationship between disease severity and HIC in hepatitis C.
The gene associated with hereditary hemochromatosis (HHC), entitled HFE, was identified in 1996. With the exception of one study, these have not found an association between the presence of HFE mutations, severity of liver disease, and hepatic iron level. A limitation of the current study is the lack of HFE mutation data in this cohort since this study was begun prior to availability of HFE mutation analysis. Furthermore, it is now recognized that multiple other genes may regulate iron metabolism, such as divalent metal transporter 1 (DMT1), ferroportin, and duodenal cytochrome b ferric reductase (dcytb). However, we feel that the lack of HFE mutation data does not weaken our findings for the following reasons. Previous studies have shown that body iron levels do not increase linearly over time in C282Y heterozygotes. It is likely that only one or two of our subjects would have been heterozygous for C282Y based on population studies. Furthermore, the fact that HIC increased only in the presence of cirrhosis, suggests that the presence of cirrhosis rather than the C282Y mutation is associated with increased HIC.
Histologic progression was noted most of the patients (71%) as has been described in previous reports. Although mean HIC was higher among men, there was no difference between men and women with regard to those who progressed to bridging fibrosis or cirrhosis. Patients who drank alcohol showed a trend toward higher mean HIC than those who did not use alcohol. However, alcohol use was not associated with presence of bridging fibrosis or cirrhosis. This is in contrast to previous reports. We suspect this difference is due to the small sample size.
We acknowledge that our study has several limitations, including the retrospective design, possible selection bias, lack of an iron stain on liver tissue for all patients, and small sample size. Nevertheless, we feel these findings are of interest since we are unaware of any previous study of the relationship between histologic progression and liver iron content in HCV. Our findings demonstrate that hepatic iron accumulation is observed only in advanced stages of disease (stage 4). Histologic progression in the precirrhotic phase associated with increased hepatic iron deposition. However, it remains unknown whether iron deposition leads to cirrhosis or vice versa. Prospective controlled studies are warranted to further explore this issue.
|
|
| |
| |
|
|
|