Reported by Jules Levin
The preliminary results from COLPILOT are among the most important information to be presented at EASL: events in patients with portal hypertension- 27% for those taking colchecine vs 11% for patients taking pegIFN a-2b.
COLPILOT Study: "EFFECT OF MAINTENANCE PEG-INTRON THERAPY ON PORTAL HYPERTENSION AND ITS COMPLICATIONS: RESULTS FROM THE COPILOT STUDY".
The COPILOT clinical trial is evaluating maintenance therapy with PEG-Intron 0.5 µg/kg weekly versus colchicine (COLC) 0.6 mg twice daily in 600 patients with HCV and advanced fibrosis who have failed prior interferon treatment. It was thought that Colchecine may also provide benefit to patients with advanced liver disease, so you can view colchcine as a placebo. The study results find better effects from interferon than colchecine. Clinical endpoints include death, liver failure, transplantation, variceal bleeding and liver cancer. The 2-year evaluation suggested an improved event free survival in patients on PEG compared to colchicine (Hepatology 2004; 40: 239A). We evaluated the role of portal hypertension (PHTN) on the clinical outcomes of maintenance therapy and the role of maintenance PEG-Intron on portal pressure. Following are Summary by the presenter of the study results Nezam Afdahl and a brief introduction, and the details of the study results presented at EASL.
SUMMARY
--PegIFN a-2b is superior as maintenance therapy with reduction in annual liver related event rate from 13.5% to 5.5% in patients with cirrhosis & portal hypertension.
--PegIFN a-2b reduces HVPG by a mean of 40% after 24 weks of treatment
--This is a significant 50% reduction in development of esophageal varices at 2 years in patients on PegIFN a-2b.
AUTHOR CONCLUSIONS:
Maintenance PEG therapy may retard varices development, reduces portal pressure and prevents variceal bleeding and complications of PHTN compared to COLC. Clinical endpoints are more common in patients with PHTN. PEG should be considered in patients with cirrhosis who fail interferon and ribavirin therapy.
Afdalh speculated at the end of his presentation that the IFN is decreasing intrahepatic inflammation and reducing angiogenic factors and vasoconstrictors such as VEGF & endothelin, and may be part of the active component of portal hypertension leading in the short term phase of the study in the first two years to an effect where there is a reduction in portal pressure which results in an improvement in the reduction in two years clinical events, particularly complications of portal hypertension & variceal bleeding.
INTRODUCTION
There were two high profile study reports at EASL on Maintenance Therapy. At the final session at EASL on Hepatitis C two oral presentations on this topic closed the conference. Two studies, the COPILT & EPIC Studies are examining whether Maintenance Therapy with peginterferon provides benefit to patients by slowing disease progression for individuals with HCV. Both studies are sponsored by Schering Plough, but the results can be applied to use of either peginterferon. Roche is sponsoring another high-profile Maintenance Therapy study called "HALT-C". Preliminary results were presented from HALT-C as well. Maintenance therapy is an important treatment option for patients with advanced HCV disease. When a patient with pre-cirrhosis or compensated cirrhosis does not achieve a sustained viral response, maintenance therapy may provide a very useful benefit for the patient. Maintenance therapy is when such a patient who was unable to achieve SVR continues with a reduced dose of peginterferon for an ongoing period of time. Many studies have found that interferon can slow HCV disease progression; many studies find interferon reduces the risk for developing liver cancer and improves survival. It is widely accepted that interferon has anifirbotic effects, that it slows fibrosis progression. Three studies are ongoing to provide better evidence that Maintenance Therapy does slow disease progression. But many doctors utilize Maintenance Therapy as an important treatment option for patients due to the many studies already conducted, while waiting for the results from these 3 studies. This affect of slowing disease progression is crucial so patients can maintain a less advanced stage of HCV disease until newer & more effective antiviral drugs for HCV become available. Without the slowing of HCV disease decompensated cirrhosis, liver cancer, and death may occur. Many new HCV drugs are in early stages of development, so it is important to retain a level of health to be able to utilize these drugs. Many companies are researching HCV protease inhibitors (being studied in patients now), polymerase inhibitors (NM283 is a polymerase inhibitor & other companies are developing drugs for this target); these are the two most popular targets that companies are developing drugs for.
Preliminary data from the COPILOT Study was reported by Nezam Afdahl & it finds results suggestive of benefit from peginterferon for patients. The EPIC Study results presented at EASL only provided SVR results. While this information is interesting and provides important insights into treatment, particularly SVR results for nonresponders & relapsers, the study has not provided data results on the Maintenance Therapy stage of the study; neither has the HALT-C study provided results yet from that part of the study. The results presented at EASL from COPILOT is the first glimpse from these studies at the potential benefits in slowing disease progression from use of interferon. My next report will provide results from EPIC Study.
EFFECT OF MAINTENANCE PEG-INTRON THERAPY ON PORTAL HYPERTENSION AND ITS COMPLICATIONS: RESULTS FROM THE COPILOT STUDY
Afdalh reported this information & study results from COPILOT. It is believed the interferon (IFN) has these effects: enhanced immune response to HCV, antiviral effect, antiproliferative, antifibrotic, anticarcinogenic, inhibition of angiogenesis, regulation of Tgf-beta.
PRIMARY STUDY ENDPOINTS
Death
Liver transplant
Liver cancer — hepatocellular carcinoma
Variceal or portal hypertensive bleeding
Increase in CPT> 2 points with jaundice, encephalopathy of ascites
SECONDARY ENDPOINTS
Histology
Quality of life
Serum fibrosis markers
Development of portal hypertension
PORTAL HYPERTENSION DEFINITION
Endoscopic diagnosis of esophageal varices
--grade 1: visible veins up to 3mm
--grade II: into lumen >3mm
--grade III: obliterating lumen
Endoscopic diagnosis of portal gastropathy:
--3 of the following: snakeskinning, edema, cherry red spots, contact bleeding, erythema
Confirmatory test: ultrasound with splenomegaly and platelet count less than 130,000/cc:
--seen in >80% of patients with endoscopic diagnosis of gastropathy

RESULTS
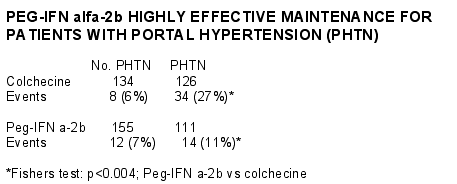
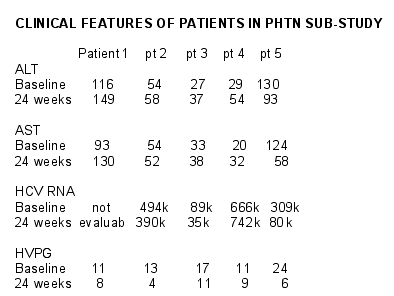
132 patients have had repeat endoscopy at 2 years and new varices were seen in 11 of 66 patients on COLC versus 5 of 66 patients on PEG (p = ns). All 5 patients had baseline elevated HVPG (Mean 15 mmHg) and all had a reduction in HVPG after 24 weeks on PEG (Mean 6 mmHg; overall HVPG reduction 41%, mean reduction in HVPG 7 mmHg). Primary event rate for patients with PHTN was 13.5% per year on COLC and 5.5% per year on PEG (p < 0.004). Primary event rate without PHTN was 3% in each treatment group. Variceal bleeding over 2 years occurred in 11 patients on COLC (9%) and 1 patient on PEG (1%). Ascites and liver failure was also more common in COLC (n = 20) compared to the group on PEG (n = 13).
The initial data immediately below was presented at AASLD 2004.
AUTHOR’S CONCLUSIONS:
Maintenance PEG therapy may retard varices development, reduces portal pressure and prevents variceal bleeding and complications of PHTN compared to COLC. Clinical endpoints are more common in patients with PHTN. PEG should be considered in patients with cirrhosis who fail interferon and ribavirin therapy.
EVENT FREE SURVIVAL: at 1440 days of study- (all patients — ITT)
80% for PegIFN a-2b vs 70% for Colchcine (p=0.007)

VARICEAL BLEED
Hematemesis or melena requiring blood transfusion
Definite: active bleeding seen from varices or stigmata of recent bleeding with no other lesion seen
Probable: large varices and no other lesion seen
Possible: varices but other lesion also present on endoscopy
10 of 11 cases definite or probable
CLINICAL FEATURES OF 11 VB PATIENTS ON COLCHCINE
7 of the 11 patients were on B-blockers
--2 no varices at index endoscopy and presented with de novo bleeding
--1 asthma
--1 grade 1 varices
time to bleeding event:
WEEK
0-24 3
24-48 4
48-96 4
Death 3 patients, OLT 2 patients
EVENT FREE SURVIVAL: PORTAL HYPERTENSION
At day 1500 of study: 0.9 for PegIFN vs 0.65 (approx, taken from view of graph)
(log rank: p<0.01)
PLATELETS <100,000: EVENT FREE SURVIVAL
At day 1500; PegIFN 0.85 vs 0.65 (aprox, taken from view of graph)
(log rank: p<0.05)
PORTAL PRESSURE SUB-STUDY
10 patients with varices randomized to PegIntron arm: 5 have completed
Portal pressure at baseline and at week 24
Standardized measurement technique for HVPG
Peripheral and hepatic venous serum and plasma
--cytokine assays
--angiogenesis and growth factor assays
DEVELOPMENT of PHTN AT YEAR 2
COLCHECINE
92 patients at risk:
--20 varices
--4 gatsropathy
2 patients developed new varices with bleeding within initial 2 years
overall 28%*
PEG IFN a-2b
95 patients at risk:
--8 varices
--4 gastropathy
overall 12%*
*Chi Square 5.44, p=0.025