| |
Adefovir-lamivudine combination therapy and hepatitis B viral kinetics
|
| |
| |
Nucleoside and
nucleotide analogues are potent inhibitors of HBV replication. Lamivudine (LAM)
and adefovir (ADV) are licensed for the treatment of patients with chronic HBV
infection, and monotherapy with each agent effects profound reduction of serum
HBV titre. However, prolonged therapy is associated with the selection and
emergence of resistant HBV species. Adopting the paradigm of HIV treatment,
studies are now exploring the potential benefit of combination nucleos(t)ide
therapy for HBV. Likely combinations will include drugs that have complementary
HBV resistance profiles. For example, LAM-resistant virus (YMDD variant) has
the M204V/I substitution. Recent data suggest that A181V/T and N236T
substitutions are important for HBV resistance to ADV. Consistent with these
observations, LAM-resistant HBV is effectively suppressed by ADV, and
ADV-resistant HBV appears to be sensitive to LAM [1,2]. In addition,
preliminary data show that combination LAM/ADV, in comparison with LAM
monotherapy, can reduce the rate of emergence of YMDD virus [3]. However, it
will prove more difficult to show that the combination reduces the rate of
emergence of ADV-resistant virus. During ADV monotherapy, resistant species
emerge at a very low rate (1-2% per annum) during the early years of treatment.
Very large controlled studies with prolonged follow-up will be required to
demonstrate with statistical significance that the addition of LAM to ADV can
reduce that failure rate. Hopefully, those studies will be carried out.
As an
alternative to large controlled studies with important and clinically relevant
end-points, it may be informative to examine the kinetics of serum HBV titres
during combination nucleos(t)ide therapy. In this issue of the Journal, Mihm
and colleagues examine the viral kinetics observed during ADV treatment of a
small cohort of patients with LAM-resistant HBV infection [4]. As a comparison
group that received ADV as monotherapy, they chose a cohort of patients treated
by Tsiang and colleagues [5]. The small size of the index cohort and the selection
of a comparison cohort with fundamentally different baseline virological
characteristics (and who received a different dose of ADV) limit the
conclusions that can be drawn from their data. A larger study with a design
that incorporated an appropriate control group(s) would have been more
informative. Moreover, the authors' aim was to describe short-term viral
kinetic data. The relevance of their observations for the development of
clinical protocols for antiviral treatment of HBV-infected patients remains
speculative. The authors acknowledge the limitations of their data and
analysis.
The two cohorts
can be compared and contrasted, and the kinetic data require close examination.
Mihm added ADV (10mg/day) to LAM for 8 patients with documented LAM-resistant
HBV infection, including patients with HBeAg-positive (n=5) or HBeAg-negative
(n=3) infection. Tsiang describes the treatment with ADV (30mg/day) of 15
patients with HBeAg-positive infection, none of whom had received antiviral
therapy during the 6 months before ADV treatment. Both studies apply
well-recognised mathematical methods to describe the viral kinetics during ADV
treatment. They examined the clearance rate of free virus (c), the drug's
efficiency for blocking virus production (e), and the rate of infected cell loss (d).The principal observations are that
there was dose-dependent efficacy in blocking viral production (lower e observed for ADV 10mg compared with
30mg/day), and that a greater rate of loss of infected hepatocytes (d)was observed during combination therapy.
For those who do not have a degree in advanced mathematics the formulae are a
bit frightening. However, study design can be scrutinised, and the inclusion in
both publications of actual data points enables critical analysis.
The observed
differences between combination therapy and monotherapy are described, but the
impact on the comparison of inclusion of HBeAg-negative patients in the index
cohort, and the administration of a higher dose of adefovir to the comparison
group needs to be considered. In addition, the observations need to be
reconciled with other published comparisons of combination versus monotherapy.
The registration studies for ADV examined the effect of ADV for treatment of
HBeAg-positive [6] and HBeAg-negative [7] treatment-naive patients. Though
detailed kinetic analysis was not reported, the decline in serum HBV titre at
monthly intervals during treatment of these cohorts was presented. The
population data showed that ADV 30mg/day effected greater reduction of serum
HBV titre than did ADV 10mg/day in HBeAg-positive patients. The same
observation has been made by Werle and colleagues [8]. Consistent with these
studies, Mihm and colleagues report a lowe e (efficacy for blocking viral production)
for the 10mg, than for the 30mg treatment dose. In other words, there is
dose-dependent inhibition of viral replication. Of course, in larger
pre-registration studies, nephrotoxicity was more commonly observed at the
higher dose. Thus, the dose of 10mg/day was chosen for licensing. Of interest,
Tenofovir (300mg/day, the licensed dose for treatment of HIV infection) was
recently shown to have significantly greater efficacy than ADV (10mg/day) for
suppression of HBV infection [9]. Indeed, the effect of Tenofovir on serum HBV
titre appears quite similar to that of ADV 30mg/day.
Though not
subjected to statistical comparison in the phase 3 studies, it appears that
greater reduction of HBV titre was achieved during ADV (10mg/day) treatment of
HBeAg-negative infection than was observed for HBeAg-positive infection [6,7].
In this context, it is useful to review the actual data, not just an average of
deltas, presented by Mihm and colleagues. The highest rates of infected cell
loss (d)were
observed for patients 6 and 7, both HBeAg-negative. That correlates with the
steep second phase decline observed for both patients (in Fig. 1 of the
manuscript). If the comparison made by Mihm and colleagues had been confined to
the HBeAg-positive patients (n=5 only), then the d values may not have differed from those
described by Tsiang and colleagues. That there is no significant difference is
consistent with the observations made by Peters and colleagues who examined the
response to ADV (10mg/day) of LAM-resistant HBeAg-positive infection [1]. In that
double-blind randomised study, comparison could be made between the combination
of LAM/ADV and ADV monotherapy. During a 48-week observation period, the
decline of serum titre observed during combination treatment was not different
from the decline effected by ADV monotherapy. Therefore, the higher d observed by Mihm and colleagues for the
combination treatment group may be partly explained by their inclusion in the
study cohort of patients with both HBeAg-positivity and HBeAg-negativity at
baseline (whereas the comparison cohort were entirely HBeAg-positive).
Taken together,
published studies show that, during treatment of HBeAg-positive infection, the
reduction of titre achieved by ADV is dose-dependant, is probably unaffected by
the presence of YMDD, is not enhanced by continuation of LAM in patients with
YMDD, and may be inferior to the reduction observed during treatment of
HBeAg-negative infection.
However, it
cannot be concluded from the cited studies that LAM should be discontinued at
time of commencement of ADV for YMDD infection. As stated above, more important
than a detailed analysis of viral kinetics during short-term therapy is the
impact of LAM maintenance as a component of combination therapy on the
subsequent emergence of ADV resistance during long-term therapy. In my opinion,
this is a likely justification for the combination LAM/ADV instead of ADV
monotherapy for treatment of YMDD infection. Also, the cited studies tell us
nothing of the likely differences between LAM/ADV combination and ADV
monotherapy given ab initio to treatment-naive patients. Preliminary data
presented at last year's AASLD annual meeting imply that, in comparison with
monotherapy, the combination may enhance the infected cell loss rate (d), and may effect a greater decline of
serum HBV titre during short-term therapy [10].
In conclusion, I
think that the differences between the population kinetics observed by Mihm and
Tsiang probably reflect simple differences between quite small cohorts at
baseline, and do not necessarily imply a beneficial effect of LAM on second
phase kinetics during ADV treatment of YMDD infection. In this case, the raw
data are more informative than the population delta. Nevertheless, other
published and presented data (and the paradigm of HIV treatment) make me favour
the maintenance of LAM after commencement of ADV for treatment of YMDD. For
treatment-naive patients, the combination probably retards the emergence of
YMDD species and may delay or prevent the emergence of ADV-resistant virus. Perhaps,
trials in progress will show that combination treatment for HBV is superior to
monotherapy, and will confirm that the treatment paradigm for HIV patients is
relevant to the treatment of our patients with chronic hepatitis B infection.
References
1. Peters MG,
Hann HW, Martin P, Heathcote EJ, Buggisch P, Rubin R, et al.. CL for the
GS-00-461 study Group. Adefovir Dipivoxil alone or in combination with
lamivudine in patients with lamivudine-resistant chronic hepatitis B.
Gastroenterology. 2004;126:91-101.
2. Villeneuve
JP, Durantel D, Durantel S, Westland C, Xiong S, Brosgart CL, et al.. Selection
of a hepatitis B virus strain resistant to adefovir in a liver transplantation
patient. J Hepatol. 2003;39:1085-1089.
3. Sung JJY, Lai
JY, Zeuzem S, Chow WC, Heathcote E, Perrillo R, et al.. A randomised
double-blind phase 2 study of lamivudine compared to lamivudine plus Adefovir
dipivoxil for treatment-naive patients with chronic hepatitis B: week 52
analysis. J Hepatol. 2003;38:25-26.
4. Mihm U,
Ga¬rtner BC, Faust D, Hofmann WP, Sarrazin C, Zeuzem S, et al. Viral kinetics
in patients with lamivudine-resistant hepatitis B during adefovir-lamivudine
combination therapy. J Hepatol. 2005;43:217-224.
5. Tsiang M,
Rooney JF, Toole JJ, Gibbs CS. Biphasic clearance kinetics of hepatitis B virus
from patients during Adefovir Dipivoxil therapy. Hepatology. 1999;29:1863-1869.
6. Marcellin P,
Chang TT, Lim SG, Tong MJ, Sievert W, Shiffman ML, et al.. Cl for the Adefovir
Dipivoxil 437 study group. Adefovir dipivoxil for the treatment of hepatitis B
e antigen-positive chronic hepatitis B. N Engl J Med. 2003;348:808-816.
7. Hadziyannis
SJ, Tassopoulos NT, Heathcote EJ, Chang TT, Kitis G, Rizzetto M, et al.. CL for
the Adefovir Dipivoxil 438 Study Group. Adefovir Dipivoxil for the treatment of
hepatitis B e antigen-negative chronic hepatitis B. N Engl J Med.
2003;348:800-807.
8. Werle B,
Cinquin K, Marcellin P, Pol S, Maynard M, Trepo C, et al. Evolution of
hepatitis B viral load and viral genome sequence during adefovir dipivoxil
therapy. J Viral Hepatitis. 2004;11:74-83.
9. Van Bommel F,
Wunsche T, Mauss S, Reinke P, Bergk A, Schurmann D, et al.. Comparison of
adefovir and tenofovir in the treatment of lamivudine-resistant hepatitis B
virus infection. Hepatology. 2004;40:1421-1425.
10. Lau G,
Cooksley H, Ribeiro RM, Powers KA, Bowden S, Mommeja-Marin H, et al..
Randomised, double-blind study comparing adefovir dipivoxil plus emtricitabine
combination therapy versus adefovir alone in HBeAg+chronic hepatitis B: efficacy
and mechanisms of treatment response. Hepatology. 2004;40:272A.
Viral
kinetics in patients with lamivudine-resistant hepatitis B during
adefovir-lamivudine combination therapy
Ulrike Mihma,
Barbara Christine Grtnerb, Dominik Faustc, Wolf Peter Hofmanna, Christoph
Sarrazina, Stefan Zeuzema, Eva Herrmannde
ABSTRACT
Background/Aims:
Mathematical analysis of viral kinetics during lamivudine-adefovir combination
therapy has not yet been performed in patients with lamivudine-resistant
hepatitis B.
Methods: In 8
patients with lamivudine-resistant hepatitis B, adefovir dipivoxil (10mg/day)
was added to ongoing lamivudine. Viral decay during the first 8 weeks of
combination therapy was described by a biphasic model to determine the
efficacy: ε, of
blocking viral production, the clearance: c, of free virus, and the loss of
infected cells: δ.
Results: Median ε was 98%, median c was 0.7/day, and
median δ was
0.07/day. No significant association was found between viral kinetic and
baseline parameters and virologic and biochemical treatment response. When
compared with viral kinetic constants reported for higher dose adefovir
dipivoxil monotherapy, ε was
lower (P=0.026) and δ was
higher (P=0.008) in this study whereas c did not differ between both studies.
Conclusions:
Although a recent study did not show any differences in the reduction of HBV
DNA comparing monotherapy with adefovir dipivoxil to adefovir-lamivudine
combination therapy in patients with lamivudine-resistant chronic hepatitis B,
mathematical analysis of early viral kinetics suggests an additional effect of
lamivudine on the infected cell loss during adefovir-lamivudine combination
therapy.
Discussion
The present
study is the first to mathematically analyze viral kinetics during combination
therapy with adefovir dipivoxil added to ongoing lamivudine in patients with
lamivudine-resistant chronic hepatitis B. Viral decline was described
convincingly by a biphasic model as used before in patients with hepatitis B
and nucleoside/nucleotide analogue therapy [23,26-28,30,33] but also more
complex staircase decay profiles had been reported [24]. Motivated from a
recent in vitro study suggesting only an incomplete inhibition of de novo
infection during therapy with adefovir and lamivudine [34], viral kinetics were
modeled with both, the assumption of incomplete (η=0.5) and, as in most previous studies,
of complete (η=1)
inhibition of de novo infection. However, differences between the resulting
curve fits and between estimated individual kinetic constants were negligible.
Patients'
characteristics at the beginning of combination therapy and viral kinetic
constants were investigated for an association with treatment outcome after 3
and 6 months of combination therapy. For treatment with lamivudine in patients
with HBeAg chronic hepatitis B, it has previously been reported that high
pretreatment ALT levels are favorable with respect to HBeAg seroconversion
[38,39]. Different patient races, HBV genotype, and HBeAg status did not lead
to differences in HBV DNA decline during therapy with adefovir dipivoxil [40].
In the present study, no association of ALT levels or HBeAg status at the time
of addition of adefovir dipivoxil to ongoing lamivudine with treatment outcome
was observed. Moreover, no significant association of viral kinetic parameters
with treatment outcome after 3 or 6 months of combination therapy was
established either. Note that the sample size may not provide enough
statistical power to detect underlying differences in the viral kinetic or
patients' baseline characteristics with respect to different treatment
outcomes. Correlation of higher infected cell loss δ with lower HBV DNA levels at month 3 of
combination therapy suggest some associations and the patient with the lowest δ never fell below 1.9×104IU/ml of HBV DNA and later developed
resistance to adefovir dipivoxil.
In the present
study, the viral kinetic constants determined from mathematical analysis of
viral decay in the first 8 weeks during combination therapy of adefovir
dipivoxil and lamivudine were compared to the viral kinetic parameters reported
by Tsiang et al. [30] in 10 patients treated 84 days with adefovir monotherapy.
Whereas in the present study adefovir was added to ongoing lamivudine in
patients with lamivudine resistance, patients in the study of Tsiang et al.
[30] had not received anti-HBV treatment 6 months prior to therapy with
adefovir. Furthermore, the patients in the present study were given 10mg
adefovir dipivoxil daily, whereas the patients in the study of Tsiang et al. [30]
took 30mg adefovir dipivoxil daily. Clearance rates c of free virus were highly
comparable in both studies. Efficacies ε of
inhibition of new virus production were significantly higher in the study of
Tsiang et al. [30] than in the present study and rates of infected cell loss δ were significantly higher in the
lamivudine-adefovir combination treatment cohort compared to the patients
receiving adefovir monotherapy [30]. Higher efficacies ε of inhibition of new virus production in
the study of Tsiang et al. [30] result most likely from the higher daily
adefovir dose in this patient group as rising efficacies ε of inhibition of new virus production
with rising doses of adefovir were described before [31]. Higher rates δ of infected cell loss in the
lamivudine-adefovir combination treatment cohort compared to those in patients
receiving adefovir monotherapy are more difficult to understand. Of course it
cannot be excluded that the differences in the two patient cohorts contribute
to this effect. Nevertheless, patients in the adefovir monotherapy group [30]
had a mean baseline HBV DNA level of 8.9×107IU/ml
compared to a mean HBV DNA level of 2.3×108IU/ml
prior to addition of adefovir dipivoxil to lamivudine in the
adefovir-lamivudine combination therapy group, indicating an equivalently high
disease activity in both patient groups. Thus, a potentially supposed higher
disease activity or a flare due to lamivudine resistance cannot explain the
differences in the rates of infected cell loss during the adefovir [30] or
adefovir-lamivudine therapy. For both, lamivudine [41-44] and adefovir [45,46],
effects on the host immune system have been reported. In chronic hepatitis B,
hyporesponsiveness of T cells was demonstrated in vitro [44,47,48] and is
believed a major mechanism for viral persistence [49]. At least a transient
restoration of anti-viral CD4 and a longer lasting restoration of HBV-specific
CD8 reactivity was demonstrated following lamivudine treatment [41-44].
HBV-specific CD8 T cells can eliminate intracellular HBV by destruction of
infected hepatocytes as well as by non-cytolytic mechanisms, thereby reducing
the number of infected cells [50,51]. Taking this into account, it is tempting
to speculate that a restoration or stimulation of the host immune system by
lamivudine leads to an enhanced clearance of infected hepatocytes during
lamivudine-adefovir combination therapy compared with adefovir monotherapy.
Note also that such a hypothesis corresponds well with the observation of
significantly faster infected cell loss reported for monotherapy with 600mg
lamivudine (n=10, median δ:
0.13/day) compared to monotherapy with 150mg lamivudine (n=11, median δ: 0.09/day), even so that difference lost
significance for a multivariate piecewise linear model [28]. Nevertheless, such
a trend was also suggested by another recent study [52]. Recently, monotherapy
with adefovir dipivoxil (n=19) was compared with adefovir dipivoxil added to
ongoing lamivudine (n=20) for 48 weeks in patients with lamivudine-resistant
chronic hepatitis B [19]. In that study no differences in HBV DNA suppression
after 48 weeks could be observed between the two treatments. Nevertheless, in
the present study, results of a different methodological approachÑmathematical
modeling of viral kineticsÑimply that there may be advantages in long-term
outcome in patients with lamivudine-adefovir combination therapy compared with
adefovir monotherapy due to an additional effect of lamivudine on infected cell
loss.
In conclusion,
the data of the present study strongly suggest further evaluation of possible
correlations between infected cell loss δ and
treatment response and further investigation of potential benefits of a
lamivudine-adefovir combination therapy compared with an adefovir monotherapy
for long-term outcome in patients with chronic hepatitis B.
Introduction
Chronic
hepatitis B is a major health problem due to its high prevalence and
association with severe liver disease and hepatocellular carcinoma. A recent
study reports sustained HBeAg loss 26 weeks after 52-week of peginterferon-α-2b in 36% of patients with
HBeAg-positive chronic hepatitis B [1]. For patients with HBeAg-negative
chronic hepatitis B sustained HBV DNA suppression below 2×104c/ml 24 weeks after the end of a
48-week treatment was observed in 43% of patients treated with peginterferon-α-2a and in 29% of patients with
lamivudine monotherapy [2]. Despite these encouraging results, treatment with
(peg)interferon-α is
limited by its side effects. By treatment with the nucleoside analogue
lamivudine, seroconversion rates of HBeAg-positive patients to anti-HBe from 17
to 47% depending on the treatment duration (1-4 years) can be obtained [3-8].
In HBeAg-negative patients with chronic hepatitis B, treatment with lamivudine
leads to suppression of HBV replication in the majority of patients. However,
in this patient group, viral relapse is observed in almost all patients after
discontinuation of therapy [9]. A major problem with prolonged lamivudine
therapy is the increasing rate of viral resistance due to mutations within the HBV
polymerase [4,6-8,10]. Treatment with adefovir dipivoxil remains effective in
patients with resistance against lamivudine [11-19]. However, recently,
resistance against adefovir dipivoxil has been observed as well [20,21],
however, without cross-resistance towards lamivudine [20,21].
Therefore, the
question arises if, in patients with lamivudine resistance, adefovir-lamivudine
combination may be superior to adefovir monotherapy by avoiding further liver
damage during potential development of adefovir resistance. Furthermore in
vitro and in vivo investigations suggest a higher anti-viral potency of
nucleos(/t)ide combination therapies compared with monotherapies [22,23].
Nevertheless, no differences in HBV DNA suppression after 48 weeks of
monotherapy with adefovir (n=19) and adefovir-lamivudine combination therapy
(n=20) in patients with lamivudine resistance have been reported in a recent
study [19].
Mathematical
modeling of viral decay summarizes additional information about the course of
disease during anti-viral therapy by determination of individual viral kinetic
parameters as the clearance rate c of free virus, a drug's efficiency ε of blocking virus production, and the
rate δ of
infected cell loss. These viral kinetic parameters can be used, e.g. for
efficient comparison of treatment groups even for relatively small sample
sizes. Whereas viral kinetic models have been applied to lamivudine [23-29] and
adefovir monotherapy [30,31] in patients with chronic hepatitis B, mathematical
analysis of adefovir plus lamivudine combination therapy has not been done so
far.
Therefore, in
this study, mathematical viral kinetic analysis was performed in patients with
lamivudine-resistant chronic hepatitis B who received adefovir dipivoxil added
to ongoing lamivudine. Individual viral kinetic parameters were determined and
compared to those previously reported for monotherapy with adefovir dipivoxil
[30]. Furthermore, patients' characteristics at the beginning of combination
therapy and viral kinetic constants were investigated for an association with
treatment outcome.
2. Patients
and methods
2.1. Patients
In the present
study, viral kinetics were analyzed in 8 patients (Table 1) with
lamivudine-resistant chronic hepatitis B. Diagnosis of chronic hepatitis B was
based on elevated serum aminotransferase (ALT) levels, the consistent detection
of hepatitis B surface antigen (HBsAg) for at least 6 months and positive HBV
DNA (Amplicorª HBV Monitor, Roche Diagnostics, Mannheim, Germany). After an
initial response to lamivudine monotherapy, in the course of treatment all
patients developed lamivudine-resistant hepatitis B, indicated by a rise in HBV
DNA to levels ³1.9×105IU/ml
and ALT levels ³1.2×upper
limit of normal (ULN) despite ongoing lamivudine therapy for at least 6 months
and good therapy compliance. Lamivudine resistance was confirmed by detection
of drug-induced mutations in the HBV polymerase in all patients (Table 1;
INNO-LiPA HBV DR, Innogenetics, Gent, Belgium). All patients tested negative
for antibodies to human immunodeficiency virus 1 and 2, hepatitis C and D.
Adefovir
dipivoxil (10mg/day p.o.) was added to ongoing lamivudine (100-150mg/day p.o.).
Patients were treated in the outpatient clinics of the Saarland University and
University Hospital in Frankfurt/Main. The study was approved by both local
Ethics Committees of Medical Research in accordance with the 1975 Declaration
of Helsinki and written informed consent was obtained from each patient prior
to enrollment.
2.2. HBV DNA
measurement and genotyping
Serum HBV DNA
was measured at baseline and daily in the first week of combination therapy,
then weekly in the first 4 weeks and then monthly. HBV DNA in each sample was
quantified independently with different assays, the Cobas Amplicorª HBV Monitor
test (lower detection limit: 57IU/ml) and with the Cobas TaqManª (lower
detection limit: 6IU/ml). Some samples were also quantified with the Amplicorª
HBV Monitor test (lower limit: 190IU/ml; all assays: Roche Diagnostics,
Mannheim, Germany. For Cobas TaqManª 1IU/ml=5.82c/ml, for Cobas Amplicorª and
Amplicorª HBV Monitor 1IU/ml=5.26c/ml was used for conversion of c/ml to
IU/ml). For mathematical modeling, results of all assays were used in parallel
taking into account the correlation of the quantification results for the same
sample.
Genotyping of
HBV was performed by the INNO-LiPA HBV GENOTYPING assay (Table 1; Innogenetics,
Gent, Belgium).
2.3.
Mathematical modeling of viral dynamics
Viral decline
during the first 8 weeks (including day 58 in two patients with no
quantifications at day 56) of lamivudine-adefovir combination therapy was
described by a biphasic model as used previously [23,24,26-28,30,32,33]
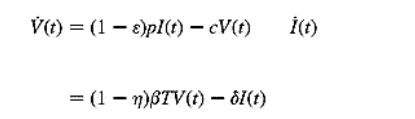
where V denotes
serum viral load, I productively infected cells, ε the efficiency factor of blocking virus
production, p the viral production rate, c the viral clearance rate, η the efficiency factor of blocking de
novo infection, β the de
novo infection rate, T uninfected target cells, and δ the rate of infected cell loss. Previous
works [23,26-28,30,33] assumed a complete block of de novo infection of
susceptible cells during anti-viral therapy, resulting in η=1 and
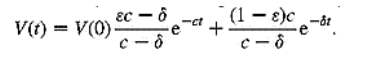
However, a more
recent in vitro study suggests an incomplete inhibition of de novo infection
during therapy with adefovir and lamivudine [34]. Therefore, besides using the
assumption η=1 the
assumption η=0.5 was
used for comparison (see also Ref. [24]) where the differential equation for
the compartment of uninfected target cells was modeled as in [35] in the latter
case. Solution of differential equations and non-linear least squares fitting
was performed with Matlab software (MathWorks Inc., Natick, MA, USA) and
estimates for c, δ, and ε were determined.
2.4.
Statistics
Data were
analyzed by Mann-Whitney U-test, Spearman and Pearson correlation,
respectively. All tests were two-tailed. P-values less than 0.05 were
considered significant.
3. Results
3.1. Viral
kinetic constants
In 8 patients
with lamivudine-resistant hepatitis B, adefovir dipivoxil was added to ongoing
lamivudine. Viral load was monitored daily in the first week of combination
treatment, then weekly in the first month and monthly thereafter. The majority
of samples was quantified with both, the Cobas Amplicorª HBV Monitor test and
the Cobas TaqManª. The two assays correlated well with a linear correlation
coefficient of 0.971 (n=83). For some samples, quantifications with the
Amplicorª HBV Monitor (correlation coefficient with Cobas Amplicorª HBV
Monitor: r=0.97; n=183; Ref. [36]) were also available and included in the
analysis. Serum HBV DNA declined in a similar biphasic pattern in all patients
and was modeled well with the proposed mathematical model (Fig. 1).
Efficiency of
blocking viral production ε, clearance
rate c of free virus, and rate for infected cell loss δ were estimated for each patient using
serum HBV DNA data of the first 8 weeks of combination therapy (Table 2). The
efficiency ε of the
combination therapy of adefovir plus lamivudine of blocking viral production
ranged from 86.3 to 99.7% with a median of 97.8%. For the clearance rate c of
free virus, a median of 0.7/day (range: 0.6-1.1/day) was calculated resulting
in a median half-life of free virus of 1 day (range: 0.6-1.2 days). The median
rate of infected cell loss δ was
0.07/day (range: 0.01-0.16/day) resulting in a median half-life of infected
cells of 9.5 days (range: 4.5-92.7 days). Kinetic constants derived under the
alternative assumption η=0.5 were
highly similar (Table 2) and the resulting fits did not differ visibly (data
not shown).
3.2.
Association of patient baseline characteristics and viral kinetic constants
with treatment outcome
After 3 months
of combination therapy with adefovir and lamivudine HBV DNA fell below 1.9×104IU/ml (Cobas Amplicorª HBV Monitor or
Amplicorª HBV Monitor) in 5 of 8 patients. None of the initially five
HBeAg-positive patients lost HBeAg at this time. ALT levels had normalized
after 3 months of combination therapy in 2 of 8 patients (Table 1).
After 6 months
of combination treatment 5 of 8 patients showed a virologic treatment response
with suppression of HBV DNA below 1.9×104IU/ml
(Cobas Amplicorª HBV Monitor or Amplicorª HBV Monitor). Two of the five
initially HBeAg-positive patients lost HBeAg, one of those became positive for
anti-HBe. ALT levels lay within normal ranges in 6 of 8 patients after 6 months
of combination therapy (Table 1). On the base of monthly ALT quantifications
during the first 6 months of combination therapy, 6 of 8 patients showed a
decreasing ALT pattern while 2 patients showed a transient increase of ALT at
month 1 of combination therapy [37].
No significant
association of patient characteristics (gender, age, weight, HBeAg-status, HBV
genotype, viral load, ALT, AST, GGT, bilirubin, plasmatic coagulation) at the
time of addition of adefovir to lamivudine with treatment response (HBV DNA
below 1.9×104IU/ml,
HBeAg loss, ALT within normal ranges at months 3 and 6 of combination therapy)
was observed.
Similarly, no
association was observed between viral kinetic parameters and ALT decline
pattern or treatment response after 3 or 6 months of anti-viral treatment with
adefovir plus lamivudine. Nevertheless, Spearman correlation coefficients of
-0.68 and -0.59 (P=0.06 and 0.13) between infected cell loss δ and HBV DNA levels at month 3 and 6 of
combination treatment, respectively, suggest a possible association here.
Patient 5 received adefovir-lamivudine combination therapy for 20 weeks and was
set to adefovir monotherapy thereafter. Interestingly, this patient, who showed
the lowest rate δ of
infected cell loss and consequently the highest half-life of infected cells
(Table 2) never fell below 1.9×104IU/ml
HBV DNA during adefovir-lamivudine combination therapy or adefovir monotherapy
and developed resistance against adefovir dipivoxil after 9 months of adefovir
monotherapy.
Viral kinetic
constants were additionally examined for an association or correlation with
baseline parameters at the time of addition of adefovir. Female patients seemed
to show slightly lower clearance rates of free virus than male patients
(P=0.046). However, no association or correlation was found between viral
kinetic constants ε, c, and δ and patients' characteristics like age,
weight, duration of infection or lamivudine pretreatment, HBV viral load, viral
genotype or HBeAg status. Parameters of liver inflammation or excretory
function did not correlate with viral kinetic constants either, only normal
plasmatic coagulation may be associated with higher efficacies ε of blocking viral production compared to
impaired plasmatic coagulation (P=0.046).
3.3.
Comparison of viral kinetic constants in patients with adefovir-lamivudine combination
therapy versus adefovir monotherapy
The viral
kinetic constants ε, c, and δ of this study were compared with those
reported by Tsiang et al. [30] for monotherapy with adefovir dipivoxil 30mg/day
for 12 weeks in 10 patients who had not received anti-viral therapy within 6
months prior to starting adefovir (Fig. 2). The clearance rates of free virus
in the study of Tsiang et al. [30] were highly comparable with those derived
here (Fig. 2A). Therapy with 30mg/day adefovir dipivoxil seemed to be more effective
in blocking viral production (median 99.6%; range 97.9-99.9%; n=10; Ref. [30])
than combination therapy with 10mg/day adefovir dipivoxil plus 100-150mg/day
lamivudine (Fig. 2B). The rate of infected cells loss δ, however, was higher in the patients of
this study and consecutively half-life of infected cells was lower than in the
patients treated with adefovir monotherapy (δ: median 0.04/day; range 0.02-0.06/day;
t1/2: median 17.1 days; range 10.8-30.1 days; n=10; P=0.008; Fig. 2C).
|
|
| |
| |
|
|
|