 |
 |
 |
| |
The Antiviral Efficacy of an HCV Polymerase inhibitor in the Chimpanzee Model: Genotypic and Phenotypic Analysis
|
| |
| |
Reported by Jules Levin
AASLD, Oct 27-31, 2006, Boston, MA
There were 3 posters on Abbott HCV polymerase development program. Here is the 1st of 3 reports.
Chih-Ming Chen, Yupeng He, Liangjun Lu, Hock Ben Lim, Rakesh L. Tripathi, Andrew Roth, Tim Middleton, Tami Pilot-Matias, Lisa E. Hernandez, David W. A. Beno, Michelle A. Long, Hongmei Mo, Warren M. Kati, Todd D. Bosse, Daniel P. Larson, Rolf Wagner, Robert E. Lanford*, William E. Kohlbrenner, Dale J. Kempf, and Akhter M. Molla
Antiviral Research, Global Pharmaceutical Research and Development, Abbott Laboratories, Abbott Park, IL, USA *Department of Virology and Immunology, Southwest Foundation for Biomedical Research (SFBR), San Antonio, TX, USA
AUTHOR CONCLUSION
- A-837093 is a potent inhibitor against both HCV genotypes 1a (-1.4 log viral load reduction) and 1b (-2.5 viral load reduction).
- It has excellent oral PK and liver distribution.
- Several single mutations can confer significant resistance.
- Proof-of-concept using A-837093 monotherapy was demonstrated in chimpanzees infected with 1a and 1b genotypes.
- Observed resistance mutations were similar in chimpanzee and the HCV subgenomic replicon (poster #432).
Introduction
HCV has a high replication rate in vivo and its RNA-dependent RNA polymerase has poor fidelity. This, coupled with the presence of quasispecies and the large viral pool size in patients result in the pre-existence of drug-resistant mutations which may undermine the effectiveness of drug treatment. A-837093 is a novel, specific inhibitor of the HCV polymerase with a serum-adjusted EC50 in the HCV subgenomic replicon of 42-143 nM and an excellent pharmacokinetic profile in animals. Since chimpanzee remains the most reliable animal model susceptible to HCV infection, we sought to validate the efficacy of A-837093 in the chimpanzee model and to examine the resistance profile resulting from A-837093 monotherapy.
Study Objectives
- To validate the anti-HCV efficacy of the non-nucleoside HCV polymerase inhibitor A-837093 in the chimpanzee model
- To investigate the presence of potential drug resistance mutations selected by A-837093
Methods
Chimpanzee study
- Oral dosing of A-837093 at 30 mg/kg BID for 14 days
- Post treatment observation for an additional 14 days
- Two chimpanzees used, one infected with genotype 1a virus and the other infected with 1b virus
- Serum samples were taken at times indicated in Figure 2
Serum sample analysis
- Viral load was determined by TaqMan® quantitative RT-PCR
- Drug level was measured by a standard method using LC/MS
- NS5B gene sequence determination was done by the following procedure:
--NS5B gene was amplified by RT-PCR reaction and cloned into a subgenomic replicon-based shuttle vector
-- 20 - 40 clones per each treatment day were selected and sequenced using BigDye chemistry
--Residues relevant to drug treatment were selected by:
Frequency change over the treatment period
Statistical analysis using Fisher's Exact test (2-tail)
- In vitro phenotype evaluation
-- Inhibitor susceptibility of pooled shuttle vector clones was determined using a transient transfection assay by measuring luciferase activity
-- Detailed protocols can be found in Tripathi et al., 2006 Antiviral Research (in press)
RESULTS
Compound Properties & PK
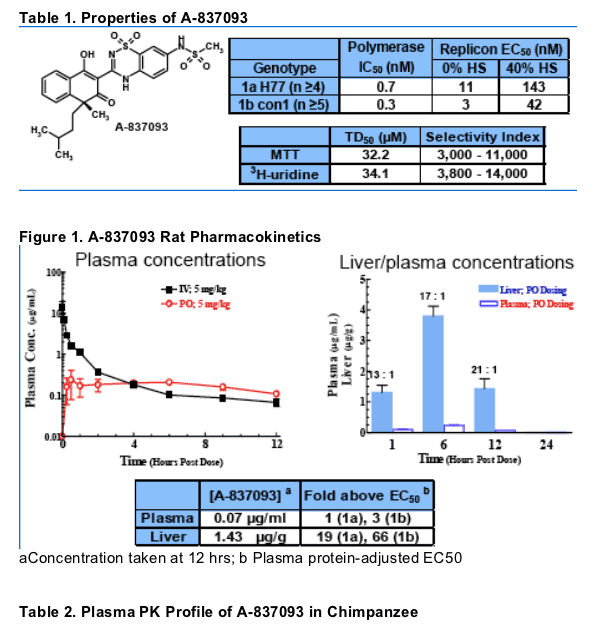
- A-837093 is a potent HCV polymerase inhibitor against both genotypes 1a and 1b (Table 1)
- A-837093 maintains a high liver to plasma ratio at 12 hrs post dosing in rat (Figure 1)
- The rat liver concentration of A-837093 is 19-fold and 66-fold over plasma protein-adjusted EC50 against genotype 1a and 1b replicons, respectively (Figure 1)
- In infected chimpanzees, A-837093 maintains a high plasma level at least 9- and 34-fold over plasma protein-adjusted EC50 (Table 3) against genotype 1a and 1b replicons, respectively
Fisher's Exact test (2-tail) was used to analyze the significance of variants occurrence over days of A-837093 monotherapy. Residues with p-value less than 0.05 are listed.
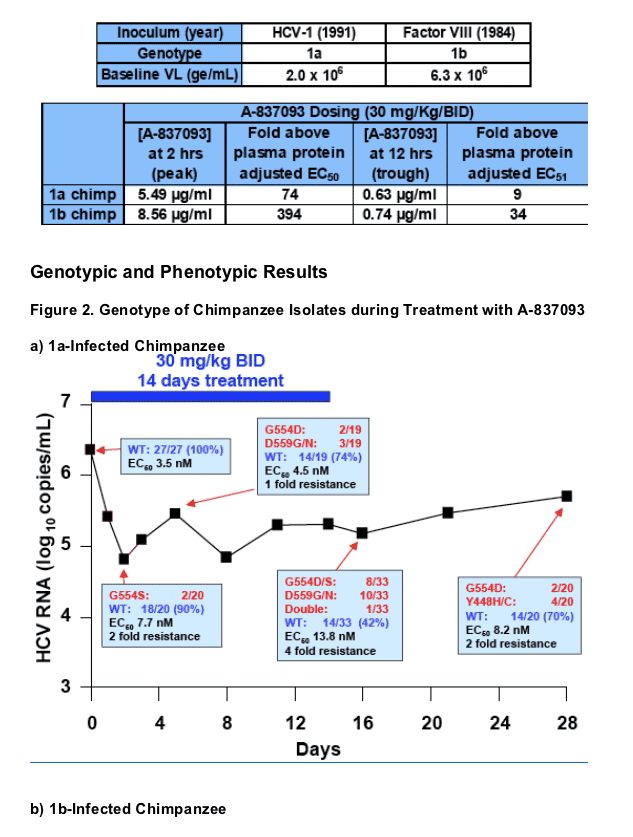
1a-Infected Chimpanzee Results
- A 1.4 log10 reduction in viral load was observed at day followed by a partial rebound (Figure 2a)
- Fisher's exact test (2-tail) indicated the changes in proportion of WT to variant over the study period are statistically significant at residues 63, 110, 206, 448, 554 and 559 (Table 3a)
- At days 5 and 16, resistance mutations were detected at G554 and D559, while at the end of the recovery period mutations at Y448 appeared and the D559 mutations were no longer seen (Tables 3a; Figure 2a)
- Wild-type species remain the major population throughout the study period
- None of the samples displayed a significant level of phenotypic resistance
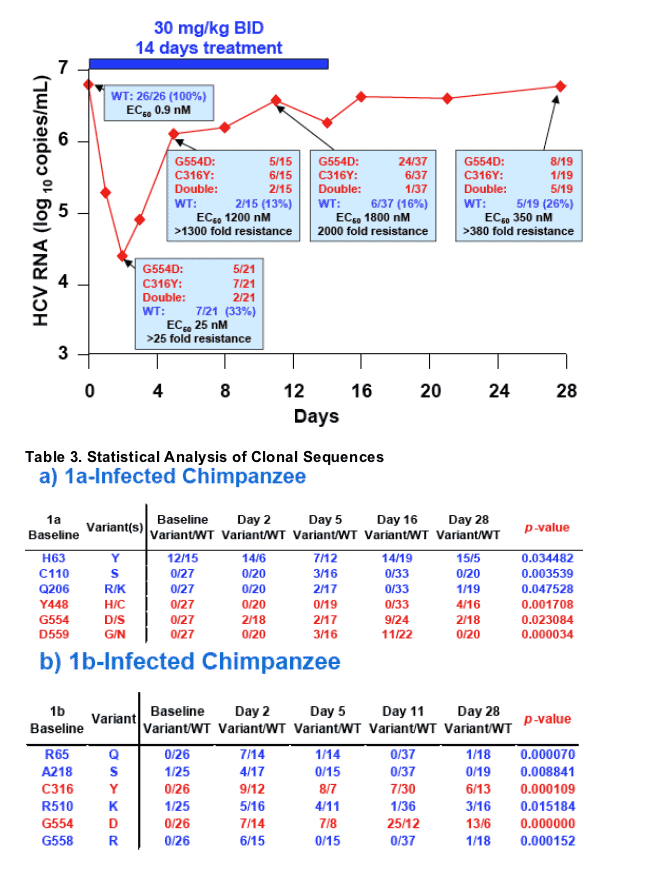
1b-Infected Chimpanzee Results
- A 2.5 log10 reduction in viral load was observed at day 2 followed by rebound to pretreatment levels (Figure 2b)
- The changes in proportion of wild-type to variant over the study period were statistically significant at residues 65, 218, 316, 510, 554 and 558 (Table 3b)
- Resistance mutations at C316 and G554 were detected at a significant level by day 2 and persisted throughout the 14 day treatment and 14 day follow-up periods (Table 3b; Figure 2b)
- In contrast to the 1a-infected animal, the proportion of wildtype species was reduced throughout the study period in the 1b-infected chimpanzee (Figure 2)
- The day 11 and day 28 samples displayed a high level of phenotypic resistance (Figure 2b)
|
| |
|
 |
 |
|
|