 |
 |
 |
| |
Roche HCV Polymerase Inhibitor Shows Potency: 4 log viral load reduction
|
| |
| |
Reported by Jules Levin
AASLD, Oct 30, 2006, Boston, MA
"Results of a Phase 1B, Multiple Dose Study of R1626, a Novel Nucleoside Analog Targeting HCV Polymerase in Chronic HCV Genotype 1 Patients"
Stuart Roberts MD*, G. Cooksley, G. Dore, R. Robson,
D. Shaw, H. Berns, M. Brandl, S. Fettner, G. Hill, D. Ipe,
K. Klumpp, M. Mannino, E. O'Mara, I. Najera, Y. Tu and C. Washington
*The Alfred Hospital & Centre for Clinical Studies, Melbourne, Australia
AUTHOR SUMMARY
R1626 has shown:
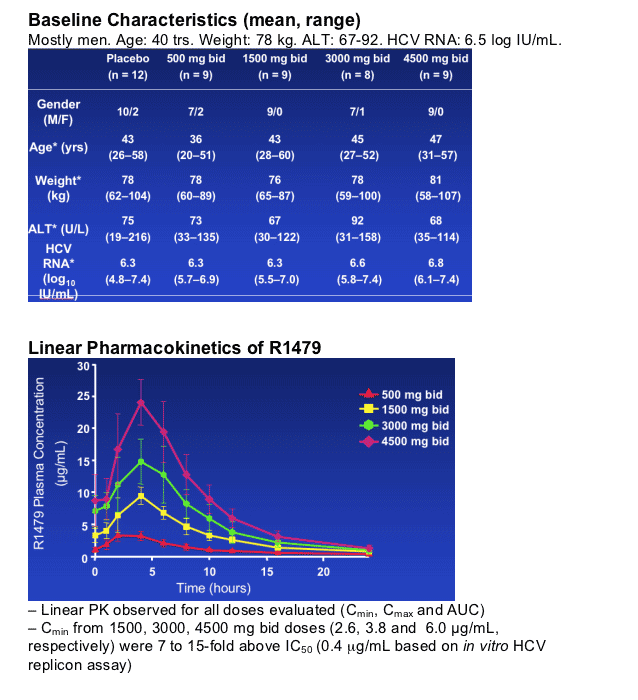
- Linear pharmacokinetics up to 4500 mg bid
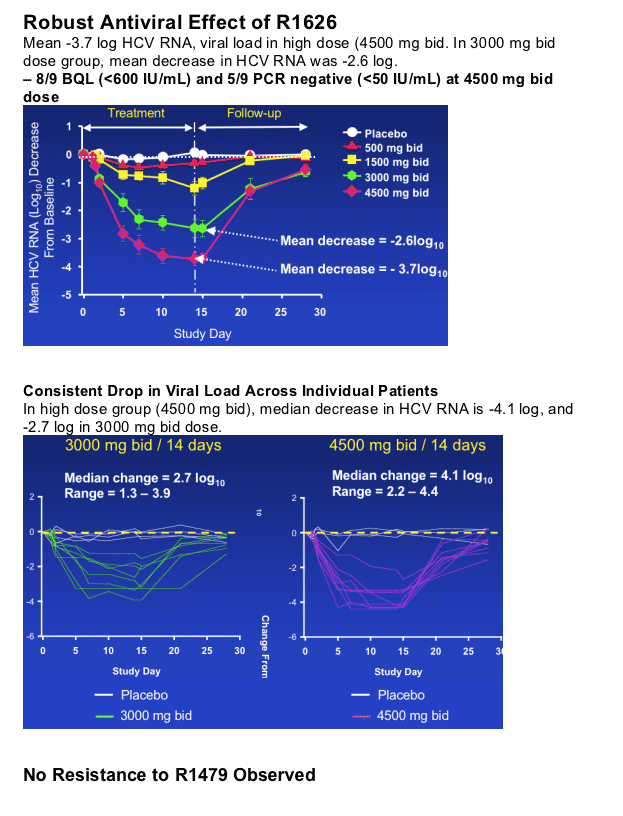
- Robust antiviral effect with mean viral load reductions of 1.2 - 3.7 log10 following 14 days of monotherapy
- No viral resistance observed
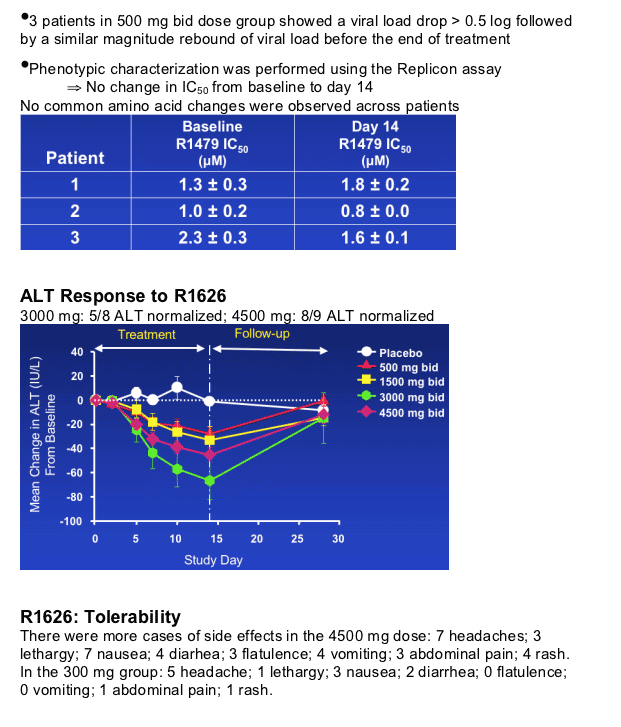
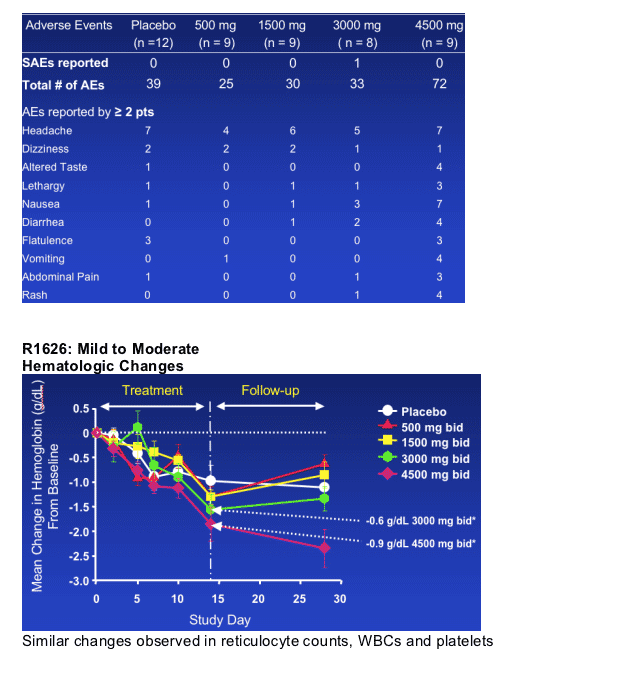
Good tolerability following twice daily doses for 14 days up to 3000 mg bid
-- Increasing adverse events at higher dose levels
-- Reversible mild to moderate hematologic changes were treatment- and time-related
R1626, a novel nucleoside analog HCV polymerase inhibitor, has shown the greatest viral load reductions in patients reported to date with this class of compounds
- Further clinical testing is needed to assess the potential benefit of the direct antiviral effect of R1626 versus potential risks
Phase 2 is underway to test the efficacy and safety of R1626 in combination with peginterferon alfa-2a and ribavirin
R1626 Characteristics
R1626 (F. Hoffmann-La Roche Ltd) is a pro-drug of the nucleoside analog, R1479, a potent and selective inhibitor of the HCV polymerase enzyme
Testing of the parent compound (R1479) in healthy volunteers and HCV patients revealed no significant safety issues R1626 was developed to significantly increase bioavailability and HCV inhibition
Study Objectives & Design
Multi-center, observer-blinded, randomized, placebo-controlled, multiple ascending dose study to evaluate:
- Safety/tolerability
- Pharmacokinetics (PK)
- Antiviral activity of R1626
Four dose levels of R1626 monotherapy twice daily (bid) for 14 days:
- 500 mg bid (9 active and 2 placebo)
- 1500 mg bid (9 active and 3 placebo)
- 3000 mg bid (8 active and 4 placebo)
- 4500 mg bid (9 active and 3 placebo)
Major Inclusion Criteria
- Treatment-naïve patients with chronic HCV genotype 1 infection
- Males and females (non-childbearing potential)
- Age: 18 to 60 years
- Metavir fibrosis stage 2
- Serum HCV RNA of ≥ 105 IU/mL
- ALT 5 times of ULN
Methods
Pharmacokinetics
- Determined on Days 1, 7 and 14
Pharmacodynamics (HCV RNA and ALT)
- Screening, pre-treatment (Days -14 , -1 and 1),
on-treatment (Days 2, 5, 7, 10, 14) and post-treatment (Days 15, 21 and 28)
Safety
- Laboratory chemistry and hematology
- Adverse events (AEs)
- Time matched ECGs
- Vital signs
|
| |
|
 |
 |
|
|