 |
 |
 |
| |
Ribavirin Dose/Levels Predicts Outcome in Pegasys/RBV ACCELERATE Study Comparing 16 vs 24 Weeks Therapy
|
| |
| |
Reported by Jules Levin
AASLD, Oct 27-31, 2006, Boston, MA
"Effect of drug exposure on sustained virological response in patients with chronic hepatitis C virus genotype 2 or 3 treated with peginterferon alfa-2a (40KD) (PEGASYS) plus ribavirin (COPEGUS) for 16 or 24 weeks"
M.L. Shiffman,1 S. Pappas,2 S. Greenbloom,3 R. Sola,4 L. Nyberg,5 J-P. Bronowicki,6 D. Crawford,7 A. Lin,8 G. Hooper,9 S. Zeuzem10
1VCU Medical Center, Richmond, VA, USA; 2St Luke's Center for Liver Disease, Houston, TX, USA; 3Toronto Digestive Disease Associates, Toronto, ON, Canada; 4Servicio de Digestivo, Hospital del Mar, Barcelona, Spain; 5Kaiser Permanente Medical Center, San Diego, CA, USA;
6CHU Nancy, Vandoeuvre-Les-Nancy, France; 7Princess Alexandra Hospital, Brisbane, QLD, Australia; 8Roche, Nutley, NJ, USA; 9Roche, Welwyn Garden City, UK; 10Saarland University Hospital, Homburg/Saar, Germany
The primary outcome of ACCELERATE, a large randomized trial examining 16 and 24 weeks of peginterferon alfa-2a (40KD) (PEGASYS®) plus ribavirin (COPEGUS®) in patients infected with HCV genotypes 2 or 3, demonstrated that, overall, 24 weeks of therapy is associated with a significantly higher rate of SVR than 16 weeks of treatment (76% vs 65% of patients; p<0.0001).[5] However, some patient populations, such as those with a low baseline viral load or a rapid virologic response (RVR; HCV RNA undetectable by week 4), achieved a high rate of SVR (>80%) with 16 weeks of treatment, demonstrating
that this may be a reasonable treatment option in those patients who find it difficult to tolerate a full course of therapy.
In this analysis we assessed the relative influence of drug exposure, as well as baseline factors, on treatment outcomes in ACCELERATE.
In ACCELERATE, treatment-naive patients with chronic hepatitis C and genotype 2 or 3 infection were randomized to receive peginterferon alfa-2a (40KD) 180 _g/week plus ribavirin 800 mg/day for 16 or 24 weeks.
(Note from Jules Levin: In line with a theme emerging from several posters at AASLD, I think the finding of most interest in this analysis is that ribavirin dosing affected outcome. Because this analysis included patients in both the 16 & 24 week groups in the ACCELERATE Study, I think the finding of this analysis that >17 weeks of Pegasys therapy (duration of Pegasys therapy) predicted outcome is not very relevant. This is because since in ACCELERATE 16 weeks did not perform as well as 24 weeks you would expect >17 weeks Pegasys to predict a better outcome. Other predictors of outcome in this analysis that are of interest include: patients with baseline viral load <400,000 IU/mL did better; patients with cirrhosis did not do as well compared to patients with bridging fibrosis; patients older than 47 yrs did not do as well as patients <47 yrs; patients with genotype 2 did better than patients with genotype 3, but among patients with RVR (rapid viral response) both genotype 2 & 3 did equally well, and that's obviously understandable.
AUTHOR CONCLUSIONS
Consistent with previous studies,[2,6-8] multiple logistic regression analysis identified HCV genotype 2 infection, younger age, lower HCV RNA level, higher ALT activity and absence of cirrhosis as being associated with a greater probability of achieving an SVR.
In addition, total exposure to peginterferon alfa-2a (40KD) (PEGASYS®) and ribavirin (COPEGUS®) each had a major influence on SVR in patients with HCV genotype 2 or 3, independent of all other factors studied.
One caveat of this study is that, since all patients in this trial received a fixed dose of ribavirin 800 mg/day, those patients with the highest cumulative ribavirin dose/kg were not only those patients who received the highest doses of ribavirin but also those patients who were the lightest. Lighter patients consistently have the highest SVR rates,[2,7] and it is possible that increasing ribavirin exposure in patients with higher bodyweight would result in similar magnitudes of increases as demonstrated in these analyses. However, previous studies that have examined the use of higher doses of ribavirin or ribavirin dose according to bodyweight have failed to demonstrate such an increase.[2,3]
These findings illustrate the importance of maintaining drug exposure in HCV genotype 2 or 3 patients treated with peginterferon alfa-2a (40KD) plus ribavirin. The effect of drug exposure on SVR rates is as important as baseline factors, showing the relative importance of treatment adherence.
Baseline factors predictive of an SVR
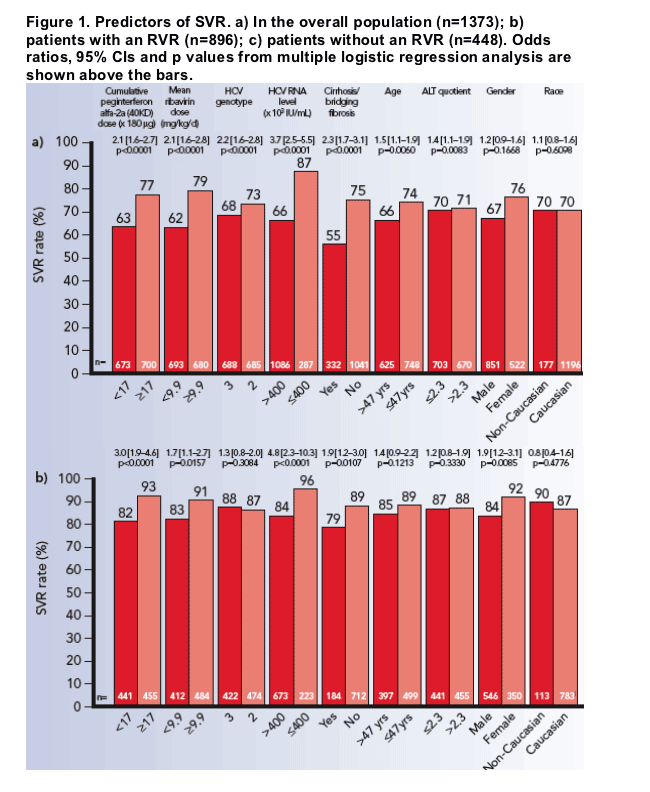
The observed effect of drug exposure and individual demographic and baseline disease factors on the development of an SVR is shown in Figure 1.
- peginterferon alfa-2a (40KD), ribavirin exposure, baseline HCV RNA level, and histologic status all strongly influenced the development of an SVR
- age, HCV genotype, and gender had moderate influence
- ethnicity and ALT quotient at baseline had minimal or no influence.
Multivariate logistic regression analysis showed that cumulative peginterferon alfa-2a (40KD) dose, mean daily ribavirin dose, HCV genotype, baseline HCV RNA level, cirrhosis status, age and baseline ALT significantly and independently predicted SVR (Figure 1a).
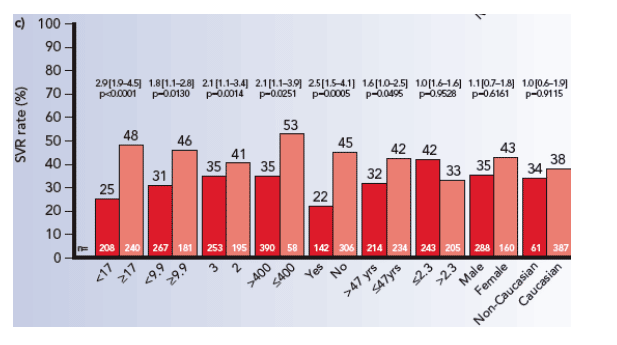
Since factors considered predictive of an SVR differed between patients with and without an RVR (ALT quotient was not predictive of an SVR among patients with an RVR, and gender was not considered predictive among patients without an RVR) in order to compare results across these subgroups of patients, all factors were included in the multiple logistic regression models (Figure 1).
Peginterferon alfa-2a (40KD), ribavirin exposure, baseline HCV RNA level, and histologic status remained strongly influenced by the likelihood of achieving an SVR regardless of the RVR status.
Effect of cumulative dose of peginterferon alfa-2a (40KD) plus ribavirin on SVR
Analysis of SVR rates by increasing cumulative exposures of both peginterferon alfa-2a (40KD) and ribavirin are shown in Figure 2.
SVR rates were highest in the group of patients who received the greatest cumulative exposure of both peginterferon alfa-2a (40KD) and ribavirin (87%; Figure 2a).
- the SVR rate was 97% in RVR patients who received the highest combined exposures.
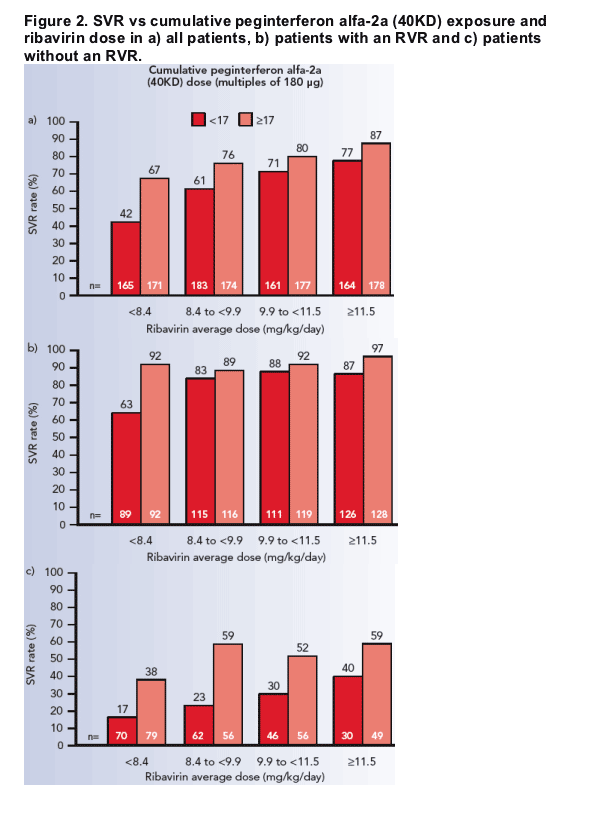
SVR rates were consistently higher in patients who received ≥17 doses of peginterferon alfa-2a (40KD), compared with patients who received <17 doses, over the range of ribavirin exposure (Figure 2a):
- in patients in the lowest quartile of ribavirin exposure (ribavirin dose <8.4 mg/kg/day), a 25% improvement in the rate of SVR was achieved in those patients who received ≥17 doses of peginterferon alfa-2a (40KD), compared with those who received <17 doses
- a 10% improvement in the rate of SVR was achieved in patients in the highest quartile of ribavirin exposure (≥11.5 mg/kg/day) who received ≥17 doses of peginterferon alfa-2a (40KD), compared with those who received <17 doses.
In patients with an RVR, ribavirin exposure had minimal impact on SVR in those who received ≥17 doses of peginterferon alfa-2a (40KD) (Figure 2b).
- SVR ranged from 89% to 97% for mean daily ribavirin <8.4 mg/kg to ≥11.5 mg/kg
- however, peginterferon alfa-2a (40KD) exposure remained important even among RVR patients receiving mean daily ribavirin ≥11.5 mg/kg. SVR was 10% higher (97% vs 87%) between those who received ≥17 vs <17 doses of peginterferon alfa-2a (40KD).
In patients without an RVR, SVR rates were consistently higher in patients who received ≥17 doses of peginterferon alfa-2a (40KD), compared with patients who received <17 doses, over the range of ribavirin exposure (Figure 2c).
- ribavirin exposure also had an effect on SVR in patients who received <17 and ≥17 doses of peginterferon alfa-2a (40KD) (Figure 2c).
METHODS & STUDY DESIGN
This analysis included all patients (from both treatment arms) who received at least one dose of either study drug and for whom complete baseline information was available.
Patients with an end-of-treatment response but who failed to return at the end of the follow-up for a final assessment of their SVR were excluded from this analysis.
The primary efficacy end-point of ACCELERATE was SVR, defined as undetectable serum HCV RNA (HCV RNA <50 IU/mL; COBAS AMPLICOR HCV Test, v2.0) at 24 weeks after the end of the treatment period.
Statistical analyses
Stepwise, backward, and multiple linear logistic regression analyses were used to examine the impact of cumulative exposure of peginterferon alfa-2a (40KD) and cumulative exposure of ribavirin, along with demographic and baseline disease characteristics, on the probability of achieving an SVR.
- a probability of 0.2 was used for both entering a variable into the model and keeping a variable in the model during the stepwise and backward selection processes.
For each patient, the cumulative exposure of peginterferon alfa-2a (40KD) was expressed as the number of standard 180 _g doses. Cumulative exposure of ribavirin was expressed as the mean daily dose (mg/kg) during treatment.
-- since patients who discontinued peginterferon alfa-2a (40KD) prematurely would simultaneously discontinue ribavirin, the duration of ribavirin treatment was not considered in the analyses as it was connected to cumulative peginterferon alfa-2a (40KD) dose.
Demographic and baseline disease factors including age (years), gender (male vs female), ethnicity (Caucasian vs non-Caucasian), HCV genotype (type 3 vs 2), pretreatment ALT quotient, pretreatment HCV RNA titer (IU/mL, log10 transformed), and pretreatment cirrhosis status (cirrhotic vs non-cirrhotic) were also examined.
- since bodyweight was used to derive the mean daily ribavirin dose, weight was not considered in the analyses.
Age, ALT quotient, pretreatment HCV RNA, cumulative peginterferon alfa-2a (40KD) dose, and mean ribavirin daily dose were considered continuous in the models.
- however, for ease of interpretation, medians were used to dichotomize each of these variables in the multiple logistic regression models
- mean ribavirin daily dose was also examined in categories defined by quartiles.
Since stepwise, backward, and multiple logistic regression analyses showed very similar results, only results from the multiple logistic regression analyses are shown.
Since, during the stepwise variable selection process, RVR was found to be the most important predictor for SVR (RVR had a chi-square statistic >350 vs chi-square statistic <80 with all other variables examined), further analyses were repeated separately among patients with an RVR and without an RVR.
- analyses were performed to examine the importance of drug exposures in the
presence or absence of an RVR.
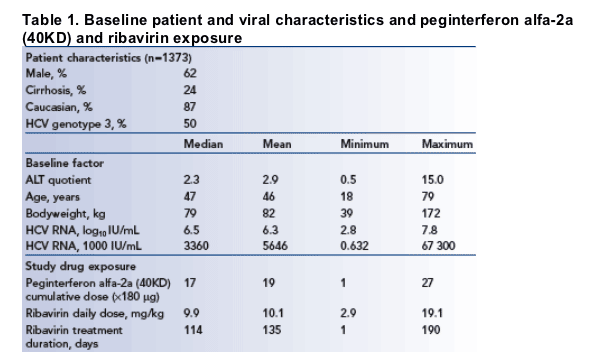
A total of 1373 patients were included in the analyses. Baseline patient and viral
characteristics and exposure levels of peginterferon alfa-2a (40KD) and ribavirin are summarized in Table 1.
|
| |
|
 |
 |
|
|