 |
 |
 |
| |
16 vs 24 weeks Pegasys/RBV for Genotype 2 HCV; Taiwan Study: Week 4 Response & Ribavirin Level/Dose Predicts SVR
|
| |
| |
Reported by Jules Levin
AASLD, Oct 31, 2006, Boston, MA
A randomized, controlled, open-label study of peginterferon alfa-2a (40KD) (PEGASYS) plus ribavirin (COPEGUS) for 16 vs. 24 weeks in patients with genotype 2 hepatitis C infection
M. Yu1, 2; C. Dai2, 3; J. Huang4; N. Hou4; L. Lee1; M. Hsieh1; C. Chiu5; Z. Lin1, 2; S. Chen1, 2; M. Hsieh1, 2; L. Wang1, 2; W. Chang1, 2; W. Chuang1, 2
1. Faculty of Internal Medicine, College of Medicine, Kaohsiung Medical University, Kaohsiung, Taiwan.
2. Department of Internal Medicine, Kaohsiung Medical University, Chung-Ho Memorial Hospital, Kaohsiung, Taiwan.
3. Department of Internal Medicine, Kaohsiung Medical University, Hsiao-Kang Hospital, Kaohsiung, Taiwan.
4. Department of Internal Medicine, Foo Yin Hospital, Pintung, Taiwan.
5. Department of Internal Medicine, Paochien Hospital, Pintung, Taiwan.
Dr Yu presented this interesting study data at the final oral session at AASLD.
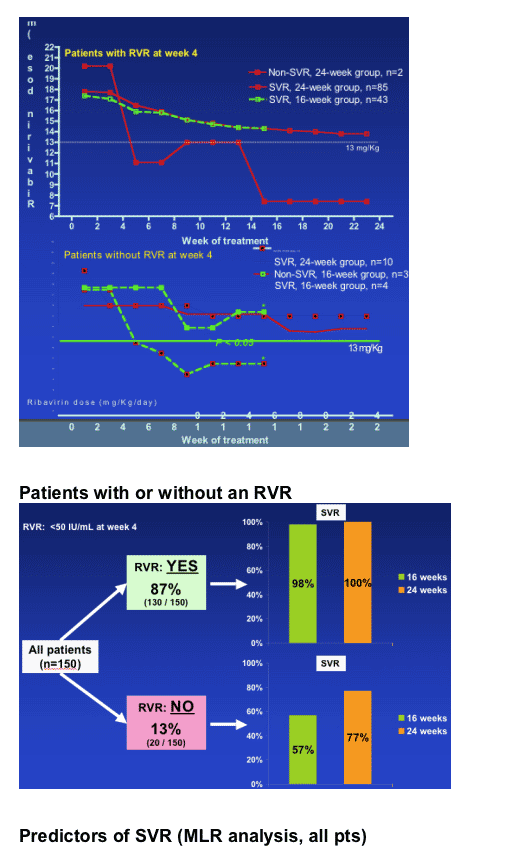
Note from Jules Levin: the findings from this study reinforce what we already knew from previous research findings, that ribavirin dosing matters; a reduction in ribavirin dosing early in the course of therapy affects SVR outcome. And in this study it appears that ribavirin dosing between weeks 4 and 16 in the 16 week group affected outcome. But in the graph below there appears to be a suggestion that RBV dosing prior to week 4, between weeks 2 & 4 affect RVR. And the study finding is that RVR, rapid viral response at week 4 was the significant factor in achieving SVR for the 16 week group.
Author Conclusions:
In Taiwanese genotype 2 patients, 16 weeks' PegINFa-2a plus RBV at a weight-based dose of 1000/1200 mg/day with an RVR at week 4:
-- As effective as a 24-week regimen
-- Comparable incidence of AEs
-- Lower cost
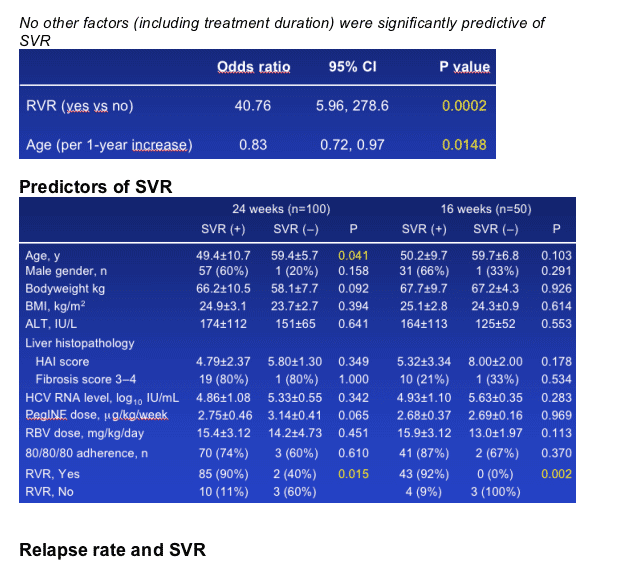
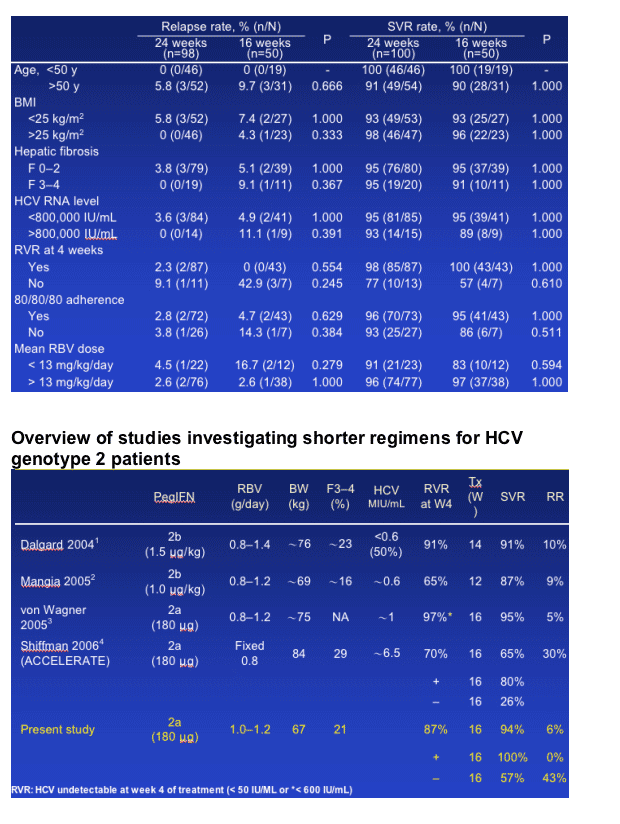
Relapse rate is comparable between the 16- and 24-week groups for patients with an RVR at week 4, but it is more than 4 times with the 16-week treatment in patients without an RVR at week 4.
A higher dose of RBV may be required when shortening duration of therapy so that efficacy is not compromised.
Study Report
The recommended treatment for patients with HCV genotype 2 infection is a pegylated interferon plus ribavirin for 24 weeks. However, SVR rates of >80% with peginterferon alfa-2a (40KD) plus ribavirin in genotype 2/3 patients have prompted interest in whether even shorter treatment duration can yield similarly high SVR rates. Therefore, we determined whether the efficacy of 16 weeks' therapy was comparable to the standard 24-week treatment course in HCV genotype 2 patients.
In a controlled, multicenter, open-label study in Taiwan, 150 treatment-naïve patients with HCV genotype 2 infection were randomized (1:2) to 16 weeks (n=50) or 24 weeks (n=100) peginterferon alfa-2a (40KD) 180 _g/week plus ribavirin 1000/1200 mg/day, with a follow-up period of 24 weeks. The primary endpoint was sustained virological response (SVR; undetectable HCV RNA [<50 IU/mL] after 24 weeks' untreated follow-up). SVR was also determined in patients with and without a rapid virological response (RVR; undetectable HCV RNA after 4 weeks).
RESULTS
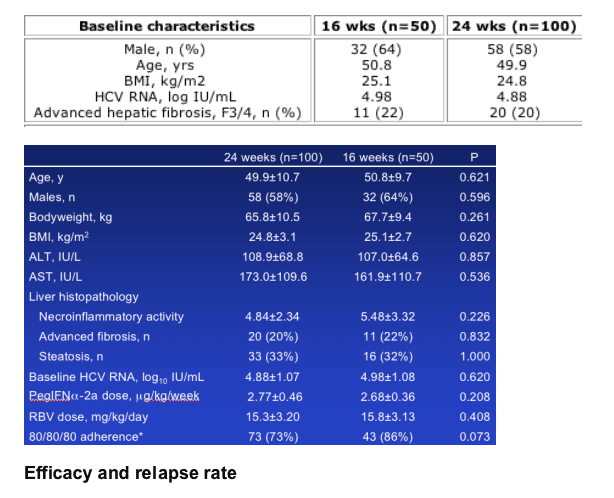
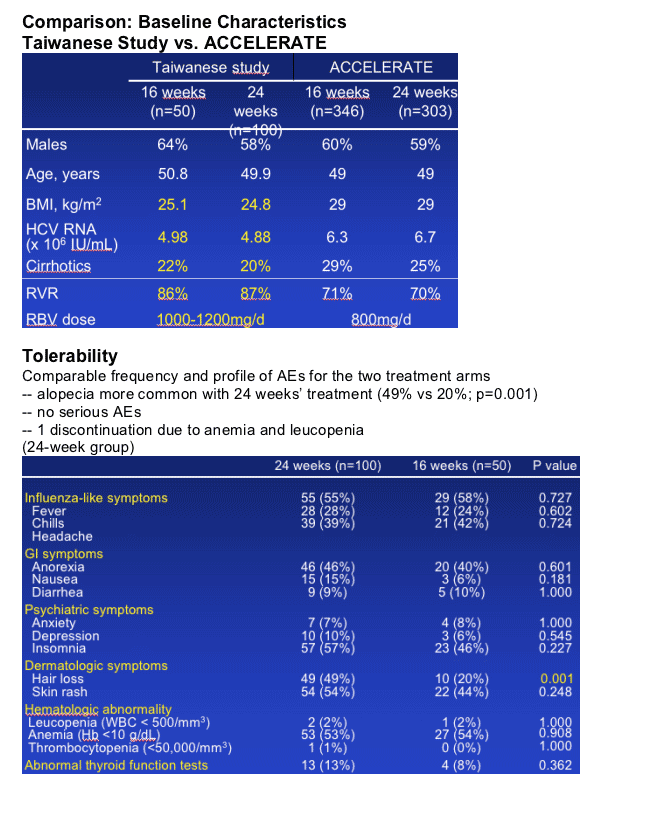
Baseline characteristics were similar in the two groups (Table).
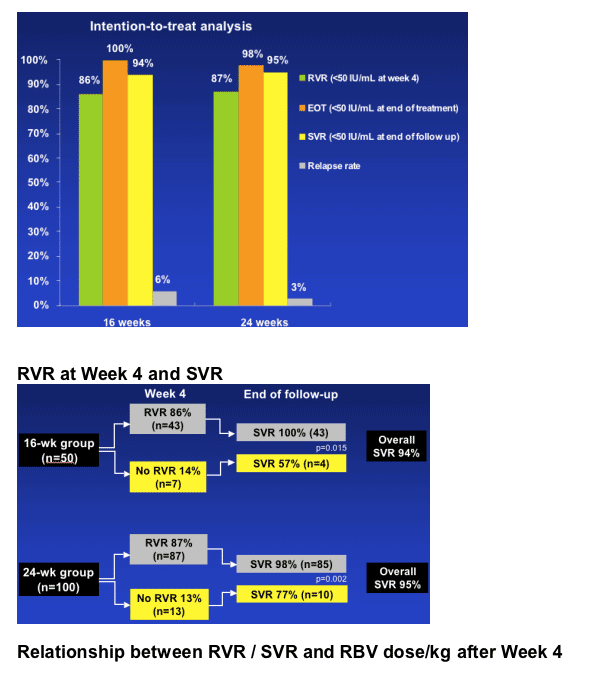
Overall, an SVR was achieved in 94% and 95% of patients receiving 16 and 24 weeks' treatment (Table).
Patients with an RVR had a significantly higher SVR rate than patients without an RVR in both the 16- and 24-week treatment arms.
For patients without RVR, the mean dose of ribavirin from week 5 to 16 of treatment was significantly lower in non-responders than in responders (11.3 ± 2.5 vs. 16.1 ± 0.8 mg/Kg/day; p=0.034 [Mann-Whitney test]). (note from jules Levin: it appears from the graph below that RBV dose decrease before week 4 may play a role in not achieving RVR).
Multivariate logistic regression analysis in all patients showed that an RVR and age were independently associated with an SVR. Both treatment arms were equally well tolerated.
The incidence of alopecia was significantly higher in the 24-week group (49%) than in the 16-week group (20%, p=0.001).
The authors concluded, In this study, high SVR rates (>94%) were seen with both 16 and 24 weeks' of peginterferon alfa-2a (40KD) plus ribavirin 1000/1200 mg/day in genotype 2 patients from Taiwan. This probably reflects the absence of negative prognostic factors in this patient population and the rapid rate with which patients responded (>86% RVR rate in both arms). We were unable to detect a difference in SVR rates with 16 and 24 weeks' treatment among the 150 patients included in our trial.
|
| |
|
 |
 |
|
|