 |
 |
 |
| |
In Vitro Susceptibility of HBV Polymerase Encoding Mutations Acquired During Adefovir Dipivoxil Therapy to Other Anti-HBV Agents
|
| |
| |
Reported by Jules Levin
AASLD, Oct 27-31, 2006, Boston, MA
X Qi, S Xiong, H Yang, M Miller, and W Delaney IV*
Gilead Sciences, Inc., Foster City, CA
Long-term nucleos/tide therapy is limited by the emergence of resistance.
Defining resistance profiles of drugs is essential for optimal patient management
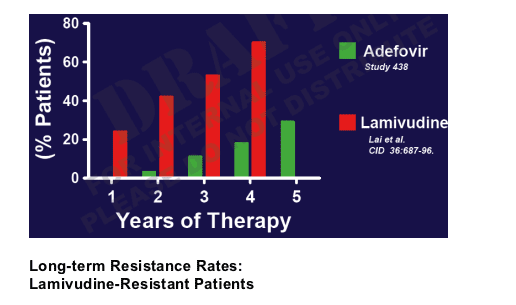
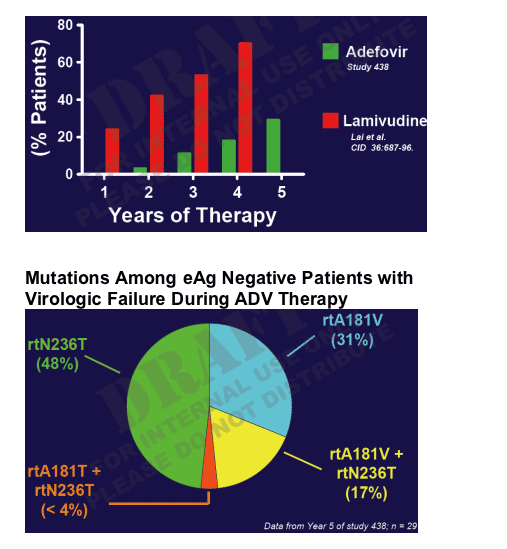
Long-term Genotypic Resistance Rates:
Treatment Naive Patients
In this table adefovir resistance development rates are about 0 in year 1, 3% after 2 yrs, 10% after 3 yrs, 20% after 4 yrs, and 30% after 5 yrs. Lamivudine rates are about 22% after 1 year, 42% after 2 yrs, 55% after 3 yrs, and 70% after 4 yrs.
OBJECTIVES
To quantify in vitro levels of resistance for adefovir-associated mutations
To quantify in vitro cross-resistance of adefovir-associated mutations to other anti-HBV agents
METHODS
Generated stable cell lines expressing five patterns of adefovir-associated mutations
- rtN236T
- rtA181V
- rtA181V + rtN236T
- rtA181T + rtN236T
- rtA181T
Used previously validated qPCR assay to test drug susceptibility
- Yang et al. Antiviral Therapy 2005 10:625
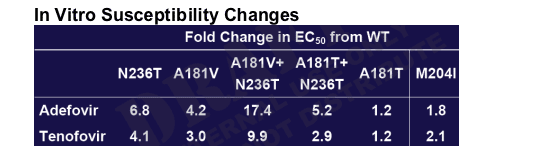
In Vitro Susceptibility Changes
These are in vitro data and are informative but in patients entecavir has demonstrated significant antiviral efficacy against adefovir resistance in Phase III study. As well, tenofovir has efficacy against adefovir resistance.
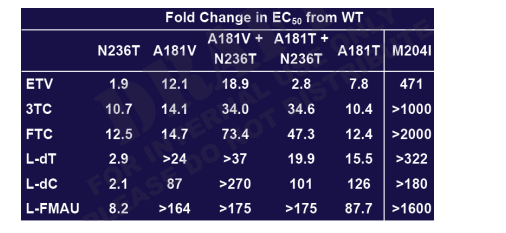
In Vitro ADV Cross-resistance Summary (I)
- A181T is not resistant to adefovir or tenofovir
- Tenofovir is more active than adefovir against ADV-associated mutants Entecavir and tenofovir are likely active clinically against ADV-associated mutations
- Villeneuve et al. Hepatology 2005 42:588A.
- Fung et al. J Hepatol 2005 43:937-43.
- Ratziu et al. Comp Hepatol 2006 5:1
A181V/T shows reduced susceptibility to all L-nucleosides
- lamivudine, emtricitabine may retain sufficient activity for clinical efficacy
- clevudine, telbivudine, torcitabine lose substantial activity
- Further clinical studies necessary for establishing in vitro cutoffs
|
| |
|
 |
 |
|
|