 |
 |
 |
| |
PegIntron/RBV Effective for >65 Year Olds
|
| |
| |
Reported by Jules Levin
AASLD, Oct 27-31, 2006, Boston, MA
"Pegylated Interferon alfa-2b + Ribavirin Is Equally Efficacious and Well Tolerated in Patients >65 Years Old in Comparison to Other Age Groups: Subanalysis of a Randomized, Controlled Study (WIN-R Trial)"
S. L. Flamm,1 I. M. Jacobson,2 R. S. Brown,3 B. Freilich,4 N. Afdhal,5 P. Kwo,6 J. Santoro,7 S. Becker,8 A. Wakil,9 D. Pound,10 J. Harvey,11 L. H. Griffel,11 C. A. Brass11
1Northwestern University, Chicago, Illinois; 2Weill Medical College of Cornell University, New York, New York; 3Columbia University College of Medicine, New York, New York; 4Baptist Medical Center, Kansas City, Missouri;
5Beth Israel Deaconness Medical Center, Boston, Massachusetts; 6Indiana University School of Medicine, Indianapolis, Indiana; 7Atlantic Gastroenterology, Egg Harbor Township, New Jersey; 8Austin Gastroenterology, PA, Austin, Texas;
9California Pacific Medical Center, San Francisco, California; 10Indianapolis Gastroenterology, RSCH, Indianapolis, Indiana; 11Schering-Plough Research Institute, Kenilworth, New Jersey
AUTHOR CONCLUSIONS
Patients 18 to 25 years of age had the highest SVR rate (57%) among the 6 age groups.
The SVR rate in patients older than 65 years (46%) was similar to the SVR rates observed in younger age groups (41%-57%).
Although elderly patients tended to experience more AEs, the rate of SAEs was similar in all groups, and the incidence of treatment discontinuation in elderly patients was similar to or less than that of younger patients.
Data from this subanalysis of the WIN-R trial strongly suggest that patients should not be denied antiviral therapy based on age alone.
Abstract*
Background: Peg interferon (IFN) + ribavirin are the standard of care for chronic HCV. There is reluctance to administer anti-viral medications to older populations due to a fear of side effects and possible decreased efficacy. Limited data is
available on pts. age >65 due to ineligibility for clinical trials. The role of age in determining response to IFN-based antiviral therapy for chronic HCV has not been clearly defined.
Aim: To determine if age is an independent predictor of sustained virological response (SVR) or medication tolerability within a randomized, controlled clinical trial of treatment-naive pts. with HCV. Patient age grps studied: 18-25 yrs., n=69;
26-35 yrs., n=350; 36-45 yrs., n=1866; 46-55 yrs., n=2200; 56-65 yrs., n=368; and >65 yrs., n=55.
Methods: A retrospective review of the multi-center WIN-R trial database was undertaken. Pts. were randomized to receive PEG IFN 2b (1.5 g/kg/wk) + either ribavirin 800 mg/d or ribavirin 800mg-1400 mg/d based on body wt. Pts. with HCV GT1/4 received 48 wks of therapy while pts with HCV GT 2/3 were randomized to 24 or 48 wks of therapy. 4913 pts. received at least 1 dose of medication and are included in this analysis. Although pts. age >65 yrs. were ineligible, 55 such pts. Were enrolled as protocol exceptions. Logistic regression analyses of SVR comparing two age categories were performed. The potential influence of demographic variables on SVR was evaluated using the chi square test (two-way frequency table).
Results:
The overall SVR was 44%.
SVR rates for the grps were:
18-25 yrs. = 57%,
26-35 yrs. = 41% (p=0.02 vs 18-25),
36-45 yrs. = 44% (p=0.03 vs 18-25),
46-55 yrs. = 42%,
56-65 yrs. = 40% (p=0.01 vs 18-25) and for
>65 yrs. SVR = 45%.
There was no difference in SVR in any other pt. age grps including pts. age >65 yrs.
There were no differences in SAEs between all age groups. Pts. age 26-35 yrs., 36-45 yrs. and 46-55 yrs. had fewer AEs than those in the 56-65 yrs. and the
>65 yrs. age groups.
Treatment discontinuations were significantly higher among the 26-35 yrs. group when compared to two groups but there was no difference in the treatment discontinuations in the >65 yrs. group compared with any other.
Conclusions:
1) Adults age 18-25 yrs. had significantly increased chances of obtaining SVR than most other age groups.
2) Pts. >65 had similar SVR to all other age groups.
3) Although there were more AEs in the older age groups, the rate of SAEs was the same and treatment drop-outs were the same or less than the younger age groups.
4) Pts. should not be denied anti-viral therapy based upon age alone.
*Poster presents data updated since abstract submission.
The Weight-Based Dosing of Peginterferon alfa-2b and Ribavirin (WIN-R) study was a prospective, multicenter, community and academic-based, open-label, investigator-initiated, US clinical trial that evaluated the efficacy and safety of PEG-IFN alfa-2b plus flat-dose or weight-based ribavirin in treatment-naive patients with chronic hepatitis C.
- Overall SVR rates were 44% in the weight-based ribavirin arm and 41% in the flat-dose ribavirin arm (P = .020).3
- SVR rates in patients with chronic hepatitis C caused by hepatitis C virus (HCV) genotype 1 (G1) were 34% and 29%, respectively (P =.004).3
- Weight-based ribavirin dosing was associated with higher rates of dose reduction for anemia but not with higher rates of discontinuation.3
Study Design
- Eligible patients had chronic hepatitis C and were treatment naive.
- Patients in the WIN-R trial were randomly assigned to 1 of 2 treatment regimens (Figure 1):
- PEG-IFN alfa-2b 1.5 _g/kg/wk plus ribavirin 800 mg/d (fixed dosing)
- PEG-IFN alfa-2b 1.5 _g/kg/wk plus ribavirin 800 mg/d to 1400 mg/d (weight-based dosing)
- Patients with HCV G1 or G4 were treated for 48 weeks.
- Patients with HCV G2 or G3 were treated for 24 weeks or 48 weeks.

Statistical Analyses
- Logistic regression analysis was used for all pair-wise combinations of SVR, serious adverse events (SAEs), AEs, and withdrawal rates (_ = 0.05; uncorrected for multiple comparisons).
- Baseline measurements were calculated using a simple arithmetic mean.
Results
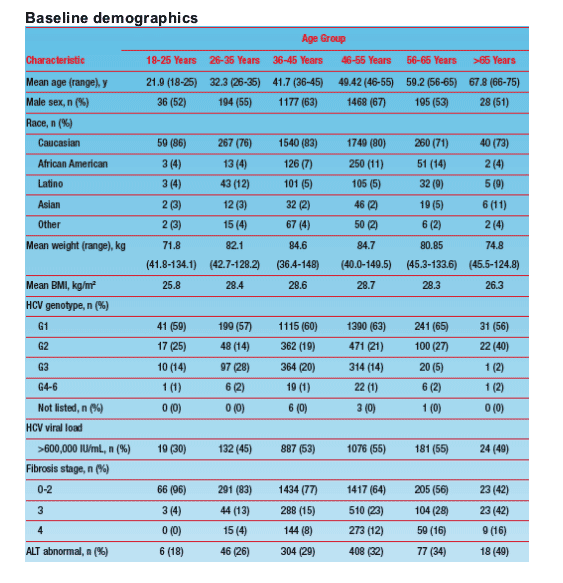
Baseline Characteristics and Patient Disposition
- Of 5519 patients enrolled in the WIN-R trial, 4913 patients received at least 1 dose of study medication and were included in the primary safety analysis (Figure 1).
- For 5 patients, no data were available for age; therefore, this subanalysis was based on 4908 patients. Although patients older than 65 were ineligible to participate in the WIN-R trial, 55 patients were enrolled as protocol exceptions.
- Baseline host and viral characteristics were comparable between treatment arms; however, there was a trend toward greater incidence of advanced fibrosis (F3-F4) and alanine transaminase (ALT) abnormalities with increasing age (Table 1).
18-25 yrs: n=69
26-35 yrs: n=350
36-45 yrs: n=1866
46-55 yrs: n=2200
56-65 yrs: n=368
>65 yrs: n=55
Efficacy Analysis
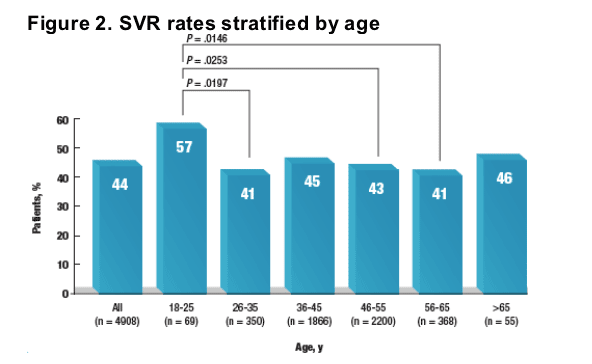
Overall SVR rate was 44%.
When stratified by age, SVR rates ranged from 41% to 57% (Figure 2).
The SVR rate in the 18- to 25-year-old age group (57%) was significantly greater than SVR rates in the 26- to 35-year-old group (41%), the 46- to 55-year-old group (43%), and the 56- to 65-year-old group (41%) (P < .05 for all comparisons).
The SVR rate observed in elderly patients (older than 65 years; 46%) was comparable to that of the other age groups.
Safety and Tolerability
Adverse Events
Patients 56 to 65 years of age had significantly more AEs than patients in the 26- to 35-year-old group, the 36- to 45-year old group, and the 46- to 55-year-old group (P < .05 for all comparisons).
Patients older than 65 years had significantly more AEs than patients in the 26- to 35-year-old group and the 36- to 45-year old group (P < .05 for both comparisons).
No statistically significant differences in AEs were noted in the other age comparisons.
No difference was observed in the number of SAEs among age groups.
Discontinuations
Significantly more patients discontinued treatment in the 26- to 35-year-old group than in the 36- to 45-year-old and the 46- to 55-year-old groups (P < .05 for both comparisons).
The incidence of treatment discontinuation in the elderly group (older than 65 years) was similar to that of the other age groups.
|
| |
|
 |
 |
|
|