 |
 |
 |
| |
RBV Exposure During weeks 1-12 of Treatment Improves SVR Rate in Canadian Pegasys EAP
|
| |
| |
Reported by Jules Levin
AASLD, Oct 27-31, 2006, Boston, MA
"Exposure to ribavirin predicts EVR and SVR in patients with HCV genotype 1 infection: analysis of the Canadian PEGASYS Expanded Access Program"
Note from Jules Levin: one of the themes emerging from this AASLD is the importance of RBV exposure. Taking appropriate ribavirin dosing significantly affects chances for SVR. We knew this before & studies at AASLD including this one confirm the importance. Several studies at AASLD showed high RBV dosing can improve outcomes.
V.G. Bain,1 S.S. Lee,2 K. Peltekian,3 E. Yoshida,4 M. Deschenes,5 M. Sherman,6 R. Bailey,7 H. Witt-Sullivan,8 R. Balshaw,9 M. Krajden10
1University of Alberta, Edmonton, AB, Canada; 2University of Calgary, Calgary, AB, Canada; 3Dalhousie University, Halifax, NS, Canada; 4Vancouver Hospital Health Sciences Centre, Vancouver, BC, Canada; 5Royal Victoria Hospital, Montreal, QC, Canada;
6University Health Network, Toronto, ON, Canada; 7Royal Alexandra Hospital, Edmonton, AB, Canada; 8Hamilton Health Sciences Corp., Hamilton, ON, Canada; 9Syreon Corporation, Vancouver, BC, Canada; 10BC Centre for Disease Control, Vancouver, BC, Canada
INTRODUCTION
The combination of pegylated interferon plus ribavirin is the treatment of choice for chronic hepatitis C.[1]
However, patients treated with combination therapy often receive less than the recommended optimal drug doses secondary to dose reductions for adverse events/laboratory abnormalities and non-adherence.
Preliminary analyses of phase III trials suggest that exposure to ribavirin is a significant predictor of sustained virologic response (SVR),[2,3] although the exact impact of reduced drug exposure on SVR is unknown.
OBJECTIVE
We examined the relationship between exposure to peginterferon alfa-2a
(40KD) (PEGASYS) and ribavirin (COPEGUS) (as continuous variables) and
the probability of early virologic response (EVR) and SVR in treatment-naive,
hepatitis C virus (HCV) genotype 1 patients enrolled in the Canadian Pegasys
Expanded Access Program (EAP).
AUTHOR CONCLUSIONS
In treatment-naive HCV genotype 1 patients, ribavirin (COPEGUS) exposure from baseline to week 12 was significantly correlated with the probability of achieving an EVR and, especially, an SVR.
Exposure to peginterferon alfa-2a (40KD) (PEGASYS) was not a statistically
significant predictor of outcomes; this may have been due to near optimal exposure in all groups.
(note from Jules Levin: previous studies have shown both pegIFN & RBV dose levels affects SVR but that RBV dosing levels appear to have more affect, particularly during the 1st 12 weeks of initiation of therapy.
METHODS
Patients
Patients eligible for the EAP had chronic hepatitis C (i.e. were HCV RNA reactive in plasma by commercial polymerase chain reaction [PCR] assay, COBAS AMPLICORa HCV Test).
This analysis was restricted to treatment-naive HCV genotype 1 patients who were assigned to receive peginterferon alfa-2a (40KD) plus ribavirin for 48 weeks.
Treatment
In the initial phase of the EAP, all patients received peginterferon alfa-2a (40KD) 180 _g/week plus ribavirin 800 mg/day. In later phases of the study, the dose of ribavirin recommended for genotype 1 patients was increased to 1000 mg/day (for patients weighing <75 kg) or 1200 mg/day (for patients weighing 375 kg).
- these rules have been used to define 'treatment groups' for the purposes of this analysis.
Hematopoietic growth factors were not used in the trial.
Outcome definitions
EVR at week 12 was defined as a >2-log10 drop in HCV RNA as determined by quantitative PCR assay (COBAS AMPLICOR HCV MONITORa Test, limit of quantitation 600 IU/mL) or undetectable HCV RNA by qualitative PCR assay (COBAS AMPLICORa HCV Test, v2.0, limit of detection 50 IU/mL).
SVR was defined as undetectable HCV RNA by qualitative PCR assay 24 weeks after treatment was stopped.
Exposure analysis
Only patients with known SVR status were included in the exposure analysis.
A ribavirin dose of 1000/1200 mg/day and a peginterferon alfa-2a (40KD) dose of 180 _g/week was defined as 'optimal' (100%), consistent with current treatment guidelines.
- in this analysis, we examined the impact of drug exposure from baseline
to week 12.
Dose adjustments for adverse events or laboratory abnormalities were recorded by investigators on case report forms and were used to calculate a continuous measure of exposure (expressed as a percentage of optimal).
The impact of baseline factors and exposure to peginterferon alfa-2a (40KD) and ribavirin was evaluated by multiple logistic regression (MLR) analyses.
Separate predictive models were developed for EVR and SVR.
- baseline factors considered for inclusion in the models were: age, bodyweight, body mass index (BMI) and baseline HCV RNA level as continuous variables; and gender, race (Caucasian vs non-Caucasian), and histologic stage as categorical variables
- drug exposure was converted to the percentage of optimal and was included in the models as a continuous variable. The possibility of an interaction between the dose of peginterferon alfa-2a (40KD) and ribavirin was also considered.
RESULTS
A total of 720 patients were included in the analysis of whom 318 (44%) and 402 (56%) were assigned to ribavirin 800 and 1000/1200 mg/day, respectively. The baseline characteristics are presented in Table 1.
Exposure to treatment and outcomes
During the first 12 weeks of treatment, the mean exposure, expressed as a percentage of the optimal dose, was 70%, 97% and 96% in patients assigned to ribavirin 800, 1000 and 1200 mg/day, respectively. Over the same period, the mean administered dose of peginterferon alfa-2a (40KD) was 94% of the planned dose.
Among patients assigned to ribavirin 800, 1000, and 1200 mg/day, an EVR
was achieved in 73%, 87% and 76%, respectively, and an SVR in 47%, 65% and 58%, respectively.
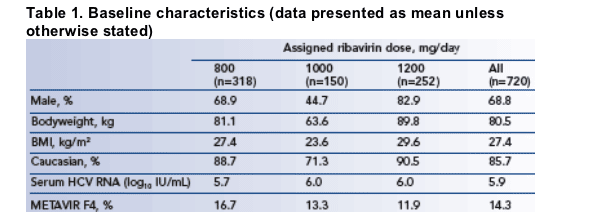
SVR by extent of exposure to ribavirin is shown in Figure 1.
- as a single predictor, ribavirin exposure from baseline to week 12 was significantly associated with observed SVR rates (chi-square test
p<0.001, Figure 1).
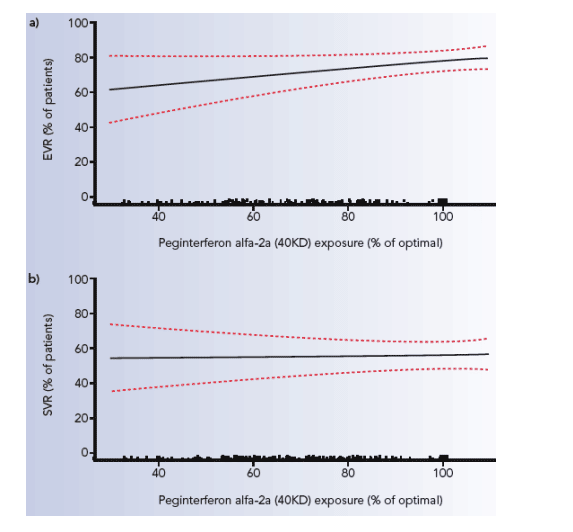
The likelihood of achieving an EVR and an SVR according to exposure to ribavirin is shown in Figure 2.
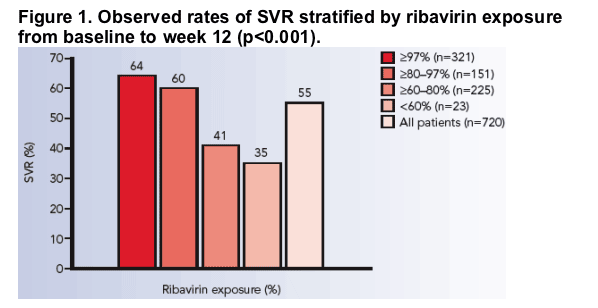
Figure 2. Exposure to ribavirin between baseline and week 12 and probability of a) EVR and b) SVR (dotted lines indicate 95% confidence intervals). The black points just above the X axis represent the distribution of exposure. The X axis extends to 120% to include two patients who received treatment for more than 48 weeks and four patients weighing <75 kg who received ribavirin 1200 mg/day.
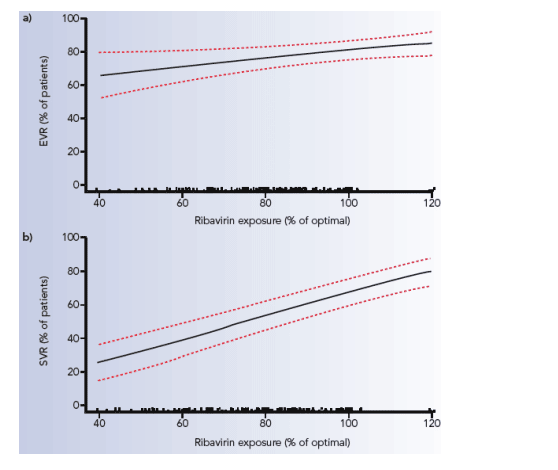
The likelihood of achieving an SVR according to ribavirin dose in mg/kg is shown in Figure 3.
The likelihood of achieving an EVR and an SVR according to exposure to
peginterferon alfa-2a is shown in Figure 4.
Figure 3. Exposure to ribavirin (mg/kg/day) between baseline and week 12 and probability of SVR (dotted lines indicate 95% confidence intervals). The black points just above the X axis represent the distribution of exposure.
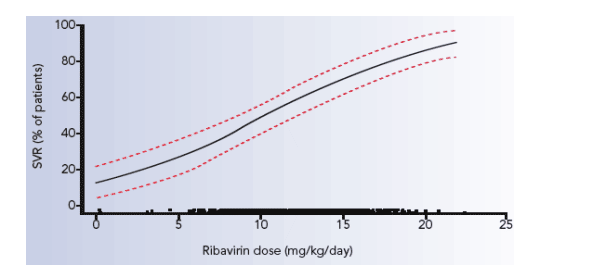
Figure 4. Exposure to peginterferon alfa-2a (40KD) between baseline and
week 12 and probability of a) EVR and b) SVR (dotted lines indicate 95%
confidence intervals). The black points just above the X axis represent the
distribution of exposure.
The importance of maintaining treatment exposure during treatment weeks 12 to 48 on outcomes is difficult to assess in a study of this type; however, in an analysis of patients who completed at least 44 weeks of treatment, the impact of ribavirin exposure from baseline to week 12 and from week 12 to end of treatment were almost equally important (data not shown).
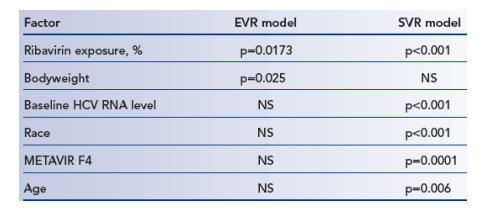
MLR analyses
Significant predictors of EVR and SVR are presented in Table 2. Exposure to peginterferon alfa-2a (40KD) was not significantly associated with SVR (p=0.84), but showed a strong trend for the prediction of EVR (p=0.051).
Table 2. Significant predictors of EVR and SVR in the MLR models.
Includes only factors that were statistically significant in at least one
of the two models. Results were similar when ribavirin exposure was
expressed as mg/kg/day
REFERENCES
1. Strader DB, Wright T, Thomas DL, et al. Diagnosis, management, and treatment of hepatitis C. Hepatology 2004; 39 (4): 1147-71
2. Lee J, Hu S, Lopez-Talavera J. Ribavirin (RBV) dose reductions in patients with HCV genotype 1 infection receiving combination treatment with peginterferon alfa-2a (40KD) (PEGASYS) plus ribavirin (COPEGUS) [abstract no. 394]. Hepatology 2004; 40 (Suppl 1): 335A
3. Reddy KR, Fried MW, Shiffman M, et al. The influence of cumulative exposure to combination peginterferon alfa-2a (40 KD) (PEGASYS) and ribavirin on sustained virological response (SVR) rates in patients with genotype 1 chronic hepatitis C. J Hepatol 2004; 40 (Suppl 1): 149 Abstract no. 505
|
| |
|
 |
 |
|
|