 |
 |
 |
| |
Resistance Rates on the Wane in Southern Europe
|
| |
| |
8th International Congress on Drug Therapy in HIV Infection
November 12-16, 2006
Glasgow, Scotland
Mark Mascolini
Studies from Portugal, Italy, and Spain found that rates of resistant virus--and rates of virologic failure--are plunging in these southern European countries.
Perhaps the most interesting of the three reports came from Belgium's Rega Institute group, working with colleagues in Portugal, because they tracked incidence of multidrug-resistant (MDR) virus (that is, newly acquired resistance) rather than MDR prevalence [1]. As a result this analysis offers a look at how rates of MDR virus may, or may not, change as simpler, more potent regimens replace the standards of yesteryear. In fact, Jurgen Vercauteren and coworkers charted steep drops in MDR incidence since 2001.
The study relied on resistance records from 2373 patients seen at 22 hospitals across Portugal. Defining MDR as susceptibility to one or fewer antiretrovirals (excluding enfuvirtide, tipranavir, and darunavir) according to the Rega resistance algorithm, Vercauteren counted new MDR cases over 12-month periods beginning in 2001 and ending in 2006. As the total number of genotypes analyzed remained fairly constant, incidence of MDR genotypes fell consistently in every period analyzed (Table).
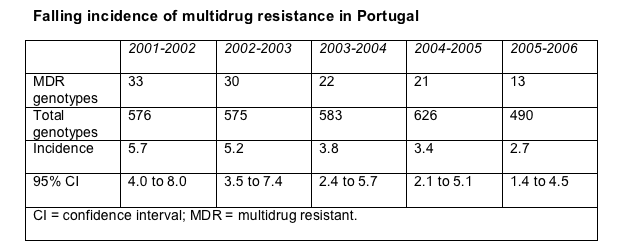
Univariate analysis determined an odds ratio (OR) of 0.82 for MDR virus through the past 5 years (95% confidence interval [CI] 0.72 to 0.94, P = 0.004), meaning that the risk of MDR virus dropped 18% in each consecutive period analyzed. Multivariate analysis found an OR of 0.80 (95% CI 0.69 to 0.93, P = 0.003) for period-to-period risk of MDR virus. In contrast, every year on antiretroviral therapy raised the risk of MDR virus 16% (OR 1.16, 95% CI 1.09 to 1.23, P < 0.001), and having an indeterminate date of first antiretroviral therapy raised the risk of MDR virus 7.88 times (95% CI 4.6 to 13.48, P < 0.001).
Vercauteren proposed that waning incidence of MDR virus in Portugal reflects the increasing efficacy of antiretroviral therapy. Although people living today with MDR HIV certainly need new antiretrovirals that stifle resistant virus, the Rega researchers proposed that future design of antiretrovirals can focus more on toxicity, tolerability, and ease of use.
A resistance prevalence study covering a similar span, 1999 through 2005, logged drops in resistance to the three first antiretroviral classes, along with a declining proportion of patients with a viral load above 1000 copies [2]. Simona Di Giambenedetto and colleagues at Catholic University in Rome figured resistance prevalence among all patients tested for mutations and calculated resistance prevalence for the entire treated population in their clinic. That population rose from 494 people in 1999 to 1429 in 2005. Yet over those years the proportion of people with a viral load topping 1000 copies--generally taken as the lower cutoff for standard genotypic resistance testing--dwindled from 59.5% to 11.3%.
Prevalence of resistance to any nucleoside, nonnucleoside, or protease inhibitor dropped significantly in both the tested population (P < 0.001 for all classes) and the total treated population (P < 0.001 for all classes). Rates of "pathway 1" thymidine analog mutations (TAMs) 41L, 210W, and 215Y all fell significantly during the study period, while rates of two "pathway 2" TAMs (67N and 219E/Q) also fell significantly, and rates of 3TC/FTC-induced 184V fell nearly significantly (P = 0.08). The ddI/abacavir-related 74V mutation became more common from 1999 through 2005, but not significantly so. And rates of the 65R mutation, which may be elicited by tenofovir, bounced up and down.
Di Giambenedetto noted that these resistance rate shifts paralleled replacement of unboosted PIs with boosted PIs in their clinic, declining use of the thymidine analogs AZT and d4T, and a more recent drop in nonnucleoside use.
A single-center study at Ramon y Cajal Hospital in Madrid confirmed a consistently falling proportion of patients with a viral load of at least 1000 copies [3]. Among people seen at the HIV clinic during the first third of 2000 through the first third of 2006, rates of those qualifying for genotyping with a 1000-plus load fell from 14.4% (174 of 1211), to 10.7% (129 of 1201), 11.1% (131 of 1183), 10.2% (113 of 1113), 7.8% (107 of 1375), 4.6% (63 of 1376), and 3.4% (41 of 1204) (P = 0.000). Among patients with a first therapeutic failure seen in those 4-month periods, rates of people qualifying for resistance testing plunged from 100% (174 of 174) to 61.2% (79 of 129), 57.2% (75 of 131), 56.5% (64 of 113), 52.3% (56 of 107), 38.1% (24 of 63), and 43.9% (18 of 41) (P = 0.000).
References
1. Vercauteren J, Theys K, Debruyne M, et al. Declining incidence of multi-drug-resistant HIV-1 over time (2001-2006): which new drugs do we really need? 8th International Congress on Drug Therapy in HIV Infection, November 12-16, 2006, Glasgow. Abstract PL5.5.
2. Di Giambenedetto S, Colafigli M, Pinnetti C, et al. Reduction in drug resistance prevalence in treatment-experienced patients: varied estimates according to employed methodologies in a clinical cohort study. 8th International Congress on Drug Therapy in HIV Infection, November 12-16, 2006, Glasgow. Abstract 214.
3. Hernandez B, Perez-Elias MJ, Gutierrez C, et al. Decline in the number of patients susceptible for resistance testing: 2000-2006. 8th International Congress on Drug Therapy in HIV Infection, November 12-16, 2006, Glasgow. Abstract 212.
|
| |
|
 |
 |
|
|