 |
 |
 |
| |
Tipranavir 500/100, 500/200 vs Kaletra in Naives
|
| |
| |
Reported by Jules Levin
Eighth International Congress on Drug Therapy in HIV Infection
Glasgow, UK, 12-16 November 2006
"Efficacy and safety of two doses of tipranavir/ritonavir versus lopinavir/ritonavir-based therapy in antiretroviral-naïve patients: results of BI 1182.33"
This is the poster version of the oral talk.
Poster Number PL13.4
Eighth International Congress on Drug Therapy
in HIV Infection
Glasgow, UK, 12-16 November 2006
D Cooper1, R Zajdenverg2, K Ruxrungtham3, J Scherer4, R L Chaves5
1National Centre in HIV, Sydney, Australia, 2Projeto Praça Onze - Federal University of Rio de Janiero, Rio de Janeiro, Brazil, 3Chulalongkorn Hospital, Bangkok, Thailand
4 Boehringer Ingelheim Pharmaceuticals Inc., CT, USA; 5Boehringer Ingelheim Pharma GmbH & Co KG, Biberach, Germany
AUTHOR CONCLUSIONS
The results of a post-hoc Week 48 efficacy analysis performed after all patients had completed a Week 60 visit (when they could have confirmed a first VL <50 copies/mL at Week 48) showed that the 500/200 mg BID dose of TPV/r was non-inferior to LPV/r. However, the 500/100 mg BID dose of TPV/r could no longer be considered non-inferior.
The two TPV/r doses tested were effective in treatment-naïve patients:
- Similar proportions of patients achieved a VL <50 copies/mL at Week 48 in both arms.
- Similar increases in CD4 cell counts in both arms.
There was a higher rate of patients who experienced Grade 3/4 ALT elevations in patients receiving TPV/r 500/200 mg BID compared with those taking LPV/r or TPV/r 500/100 mg BID.
The proportions of patients experiencing Grade 3/4 lipid elevations were comparable among arms.
At the doses tested in this trial, TPV/r cannot be recommended for ARV-naïve patients infected with wild type HIV-1.
Abstract
Purpose of the Study Tipranavir (TPV) is a PI with potent activity against multi-PI resistant HIV-1. TPV/r 500/200 mg bid provides superior efficacy to other PI/r in highly treatment-experienced (HTE) patients. We compared safety and efficacy of two TPV/r doses with lopinavir/ritonavir (LPV/r) in antiretroviral (ARV)-naïve patients.
Methods A dose ranging, multicentre, open-label trial randomised 562 ARV-naïve patients to 3TC/tenofovir + TPV/r 500/100 mg bid (TPV/r-100), 500/200 mg bid (TPV/r-200) or LPV/r 400/100 mg bid (LPV). Primary endpoint was confirmed viral load (VL) <50 copies/mL at Week 48 without prior ARV changes.
Summary of Results Baseline (BL) characteristics were similar. At Week 48, similar %s had confirmed VL <50 copies/mL: 65.8% (TPV/r-100), 66.7% (TPV/r-200), 69.2% (LPV) (ITT); 80.1%, 81.4%, 83.0% (OT). No differences in responses were detected regardless of BL VL. Median CD4 cell counts increased in all arms (+172, +175, +207 cells/mm3). Higher % of patients in TPV/r-200 arm had Grade 3/4 (G3/4) ALT elevations (19.6%) vs. TPV/r-100 (6.0%) and LPV/r arms (3.8%). G3/4 triglyceride rates were similar in all arms.
Conclusions The TPV/r-200 arm was closed due to higher rate of G3/4 ALT elevations. While non-inferiority was achieved with data collected up to Week 48 in all arms, TPV/r-100 was no longer non-inferior using Week 60 data from all patients due to a higher discontinuation rate based on a less favourable GI tolerability profile.
Introduction
Tipranavir (TPV) is a new generation protease inhibitor (PI) with potent activity against multi-PI resistant HIV-1 [1]. In combination with an optimized background regimen, TPV co-administered with low dose ritonavir (TPV/r) at a dose of 500/200 mg BID provided superior efficacy to comparator PI/r in highly treatment experienced (HTE) patients for at least 96 weeks [2,3]. We compared the safety and efficacy of two TPV/r doses with a standard dose of lopinavir/ritonavir (LPV/r) in antiretroviral (ARV)-naïve patients (study BI1182.33). All patients also took lamivudine (3TC) and tenofovir.
Patients
BI1182.33 was a dose ranging, multi-centre, open-label trial (Figure 1). Patients were HIV-infected, ARV naïve adults (≥18 years old) with a baseline VL ≥5,000 copies/mL and a CD4 cell count <500 cells/mm3. There were no entry restrictions with respect to very low CD4 counts. A total of 562 ARV-naïve patients were randomised to receive 3TC/tenofovir + TPV/r 500/100 mg bid (TPV/r-100), 500/200 mg bid (TPV/r-200) or LPV/r 400/100 mg bid (LPV), stratified according to CD4 cell counts of <200 or ≥200 cells/mL.
Study design
The study design of BI 1182.33 is shown in Figure 1.
Open label, randomised, international, parallel group non-inferiority trial
Primary endpoint: percentage of treatment responders (VL <50 cp/mL) at Week 48

The primary endpoint was a confirmed viral load (VL) <50 copies/mL at Week 48 without prior antiretroviral (ARV) changes.
Non-inferiority between each TPV/r group and the LPV/r group was concluded if the lower limit of the 97.5% confidence interval for the difference in response rate, adjusted for CD4 stratum, was not less than or equal to the pre-defined non-inferiority margin of 15%.
Results
Patients
Table 1 summarises the baseline characteristics of patients enrolled in BI1182.33 (Full Analysis Set [FAS]). Approximately one quarter of the patients (23.5%) were female. The median CD4 cell count was 207 cells/mm3 and the median VL was 5.03 log10 copies/mL.
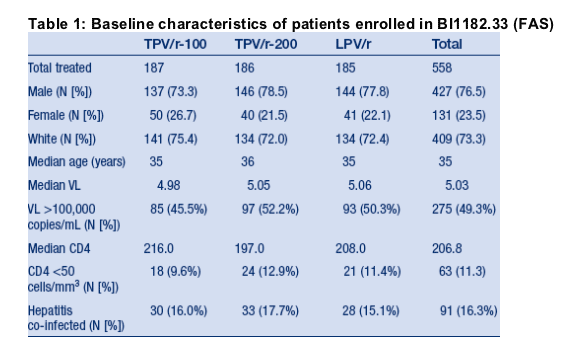
Figure 2 depicts the disposition of patients at Week 48.
Virological efficacy
At Week 48, similar proportions of patients in each study arm had confirmed
VLs <50 copies/mL (Intent to Treat analysis [ITT]) (Figure 3):
- TPV/r-100: 65.8%
- TPV/r-200: 66.7%
- LPV/r: 69.2%
The primary analysis comparing treatment response among treatment groups showed non-inferiority (CD4 stratum adjusted 97.5 C.I.s (-14.2, 7.1) and (-11.8, 9.6) for TPV/r-100 and TPV/r-200 compared to LPV/r, respectively).
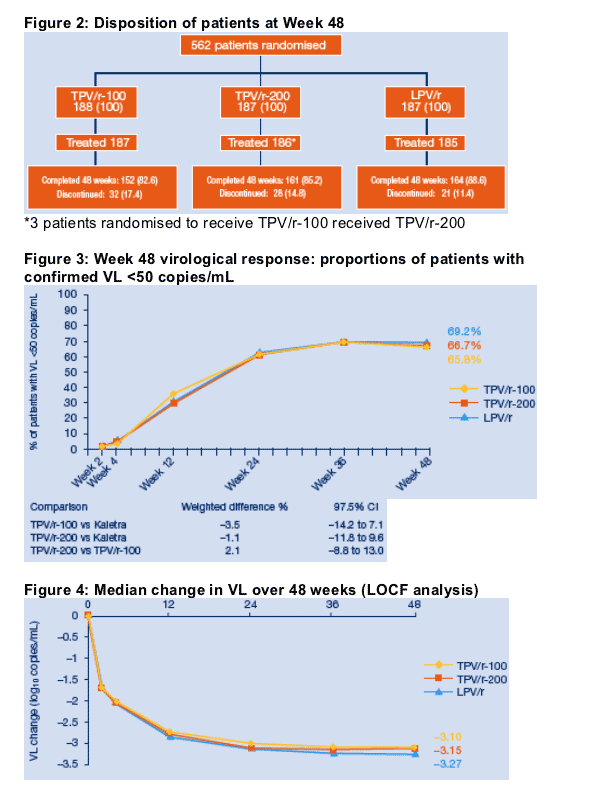
Since treatment response was defined as a confirmed VL <50 copies/mL and some patients achieved their first VL <50 copies/mL at Week 48, a post-hoc Week 48 analysis was conducted. Since Week 60 was the visit immediately following Week 48, the Week 48 post-hoc analysis included data up until Week 60 in all patients in order to allow VL confirmation to patients who had their first VL <50 copies/mL at Week 48. When Week 60 data were included for all patients in this post-hoc analysis, the lower limit of the 97.5% confidence interval for TPV/r-100 - LPV/r (-15.03, 5.8) extended below the pre-defined non-inferiority margin of 15% resulting in TPV/r-100 no longer being demonstrated to be non-inferior to LPV/r.
The median change in VL over 48 weeks (last observation carried forward [LOCF] analysis), shown in Figure 4, was comparable between treatment groups.
Immunological efficacy
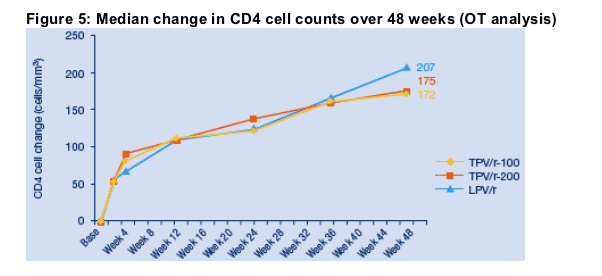
Median CD4 cell counts increased in all arms by Week 48: +172, +175, +207 cells/mm3 in the TPV/r-100, TPV/r-200 and LPV arms, respectively, (LOCF analysis) (Figure 5).
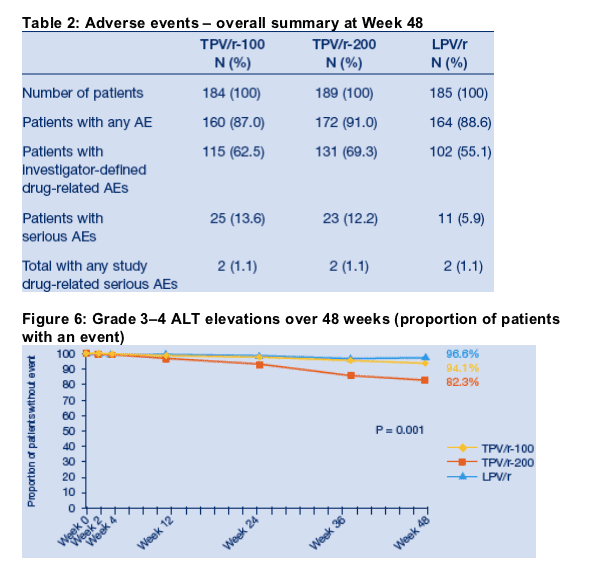
Safety
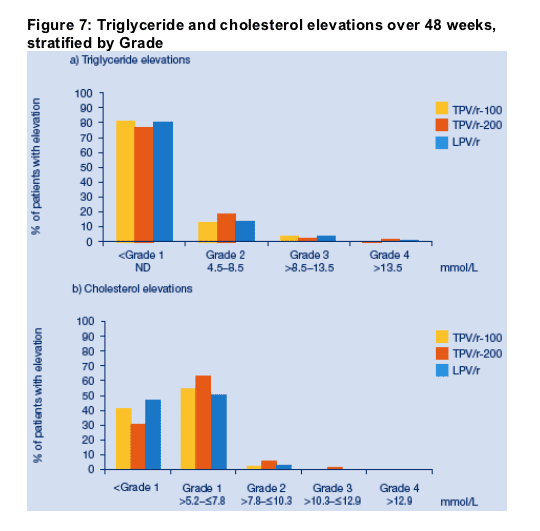
Table 2 provides an overall summary of adverse events reported by Week 48. A higher proportion of patients in the TPV/r-200 arm had Grade 3/4 ALT elevations (17.7%) than in the TPV/r-100 (5.9%) or LPV/r arms (3.4%) (Figure 6). Despite this increased rate, no clinical cases of hepatitis or hepatic failure or fatal cases due to hepatotoxicity occurred in the TPV/r-200 arm. Triglyceride and cholesterol elevations, stratified by Grade, over 48 weeks are depicted in Figure 7. The low rates of Grade 3/4 elevations in triglyceride and lipid levels were similar in all three study arms.
References
1. Larder, B., et al. Tipranavir inhibits broadly protease inhibitor-resistant HIV-1 clinical samples. AIDS, 2000. 14: p. 1943-48.
2. Farthing, C., et al. Tipranavir/r demonstrates superior and durable treatment response compared with comparator PI/r in highly treatment experienced (HTE) patients: Week 96 RESIST 1 and 2 results. In 46th Interscience Conference on Antimicrobial Agents and Chemotherapy (ICAAC). 2006. San Francisco. Abs. H-1385.
3. Hicks, C.B., et al. Durable efficacy of tipranavir-ritonavir in combination with an optimised background regimen of antiretroviral drugs for treatment-experienced HIV-1-infected patients at 48 weeks in the Randomized Evaluation of Strategic Intervention in multi-drug reSistant patients with Tipranavir (RESIST) studies: an analysis of combined data from two randomised open-label trials. Lancet, 2006. 368(9534): p. 466-75.
|
| |
|
 |
 |
|
|