 |
 |
 |
| |
Optimal Kaletra Adherence During 1st Few Weeks on HAART Key To Achieving Viral Suppression
|
| |
| |
Reported by Jules Levin
8th Intl HIV Congress, Glasgow, UK, Nov 2006
"Relationship Between Time to Viral Load Suppression and
First Month Adherence to Prescribed Antiretroviral Therapy"
RA Rode1, BC Vrijens2, AC Lange2, KR Niemi1, KJ Wikstrom1, RS Heuser1, TJ Podsadecki1
1Abbott Laboratories, Abbott Park, IL, USA; 2AARDEX Ltd., Zug, Switzerland
Strict adherence to prescribed antiretroviral therapy is an important predictor of success in the treatment of HIV-1 infection. At initiation of antiretroviral therapy, the degree of adherence needed to achieve plasma HIV-1 RNA (viral load, VL) suppression might differ from that needed to maintain suppression. In this study, we investigate the relationship between the time to achieve VL suppression and early adherence to antiretroviral therapy.
AUTHOR CONCLUSIONS
-- The lower the baseline viral load, the sooner subjects achieve VL suppression on a LPV/r-based three-drug regimen. As well, the higher the CD4 count at baseline the sooner patients achieved viral load suppression. We know that the sooner you achieve viral load suppression the morelikely you are to maintain undetectable viral load.
-- Treatment-naïve HIV-1 infected patients who achieve optimal adherence during the first few weeks of HAART are more likely to achieve VL suppression and to do so more quickly than those who do not.
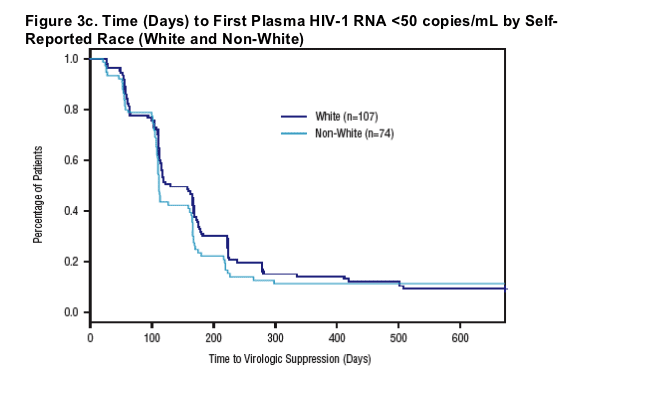
-- The relationships between race and VL suppression warrant further evaluation.
RESULTS
Lower baseline CD4+ T-cell count (p=0.021) and higher baseline VL (p<0.001) were associated with longer time to VL suppression. Overall, race (white vs. non-white) was not significantly associated with time to VL suppression (p=0.26); however, further stratification revealed that black subjects (n=56) had significantly shorter time to VL suppression compared to white subjects (p=0.046; data not shown). Gender and age were not significantly associated with time to VL suppression (p>/=0.068).
In a multivariate model adjusted for all relevant covariates, each 10% increase in adherence during the first month after initiation of antiretroviral therapy was associated with a 24% increase in the likelihood of VL suppression (HR=1.24 [95% CI: 1.09-1.40], see Table 2).
Also, a 1.0 log10 copies/mL increase in VL at baseline was associated with a 44% decrease in the likelihood of VL suppression (HR=0.56 [95% CI: 0.47-0.67]). LPV/r treatment arm (QD or BID), gender, race (white vs. non-white), age and baseline CD4+ T-cell count were tested, but not included in the final model. When race was further stratified, black subjects were more likely than white subjects to achieve VL suppression (HR=1.58 [95% CI: 1.12-2.23]).
METHODS
M02-418 was a randomized, open-label, multicenter, parallel arm study comparing the safety, tolerability, antiviral efficacy and pharmacokinetics of lopinavir/ritonavir (LPV/r) soft-gelatin capsules, administered QD 800/200 mg
(n=115) or BID 400/100 mg (n=75), with 300 mg tenofovir disoproxil fumarate (TDF) and 200 mg emtricitabine (FTC) (both QD) in antiretroviral-naïve, HIV-1 infected subjects. For the purpose of this study, subjects were considered antiretroviral-naïve if they had received less than 7 days of prior antiretroviral therapy. In addition, subjects were required to have plasma HIV-1 RNA >1,000 copies/mL at screening; however, there was no CD4+ T-cell count restriction.
Patient safety was assessed throughout the conduct of this study. Blood collection for evaluation of virology (plasma HIV-1 RNA), immunology (CD4+ T-cell count), clinical chemistry and hematology parameters was conducted at
baseline, weeks 4, 8, 16 and 24, every 8 weeks thereafter until week 48, and every 12 weeks thereafter until week 96. Time to suppression of VL is defined as the time in days between initiation of the antiretroviral regimen and the first
visit with plasma VL <50 copies/mL. Treatment compliance was assessed through the use of MEMS® monitors, which electronically recorded and stored LPV/r bottle openings for subjects enrolled in this study. Adherence was estimated between baseline and first visit by correct dosing compliance (COD).
COD: The percentage of days with the correct number of doses taken
COD is calculated as:
number of days with number of openings as prescribed X 100
number of monitored days
If the read-out from the MEMS monitor for a BID regimen shows 43 openings, but there were exactly 2 openings on only 20 of the 25 monitored days, the percentage of days with the correct number of doses taken (or COD) is
80% [100 X (20/25)].
This statistic captures some measure of the closeness to "correct compliance," but gives no information concerning the timing of dose intake and does not distinguish between days of over-dosing and days of under-dosing.
A Cox proportional hazards model was used to assess the association between time to VL suppression and adherence. Associations with baseline covariates (LPV/r treatment arm [QD vs. BID], gender, race [white vs. nonwhite], age, CD4+ T-cell count and VL) were also investigated using simple (univariate) and multiple (multivariate) stepwise regression. Only those factors determined to be statistically significant at the 0.05 level in the multiple stepwise regression were included in the "final" regression model.
RESULTS
The study population has relatively advanced HIV disease, as approximately 47% of subjects had baseline CD4+ T-cell count below 200 cells/mm3 and 40% had baseline plasma HIV-1 RNA above 100,000 copies/mL.
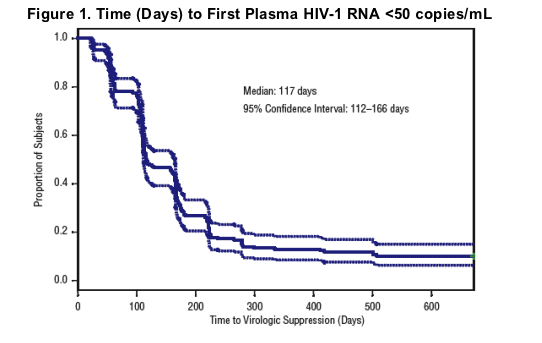
VL suppression was achieved at least once during the trial among 163 of these subjects (90%, see Figure 1). Median (95% CI) time to VL suppression was 117 (112-166) days, with no difference between LPV/r QD and BID regimens (p=0.385).
Better adherence to prescribed regimens during the first month was significantly associated with time to VL suppression (p=0.004, see Figure 2).
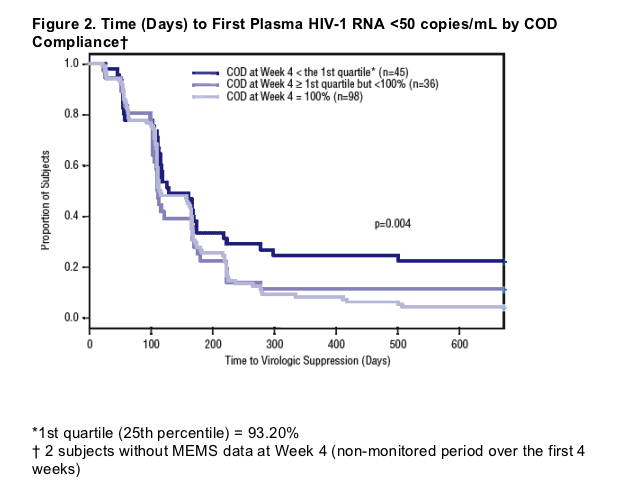
Once again, the relationship between adherence (COD) and time to VL suppression was similar, regardless of whether subjects received LPV/r QD or BID (p=0.940).
Time to VL suppression was stratified by baseline demographic characteristics (see Figures 3a-3c).
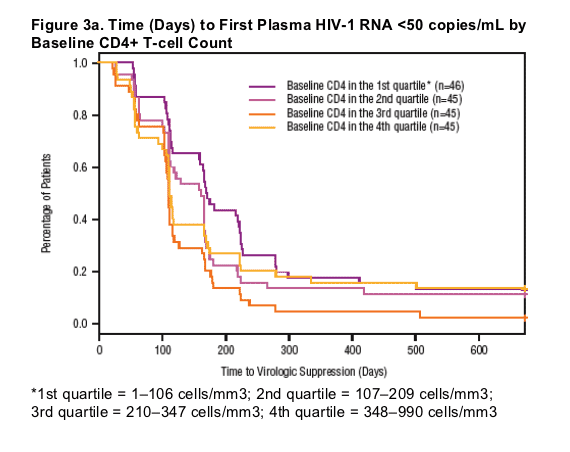
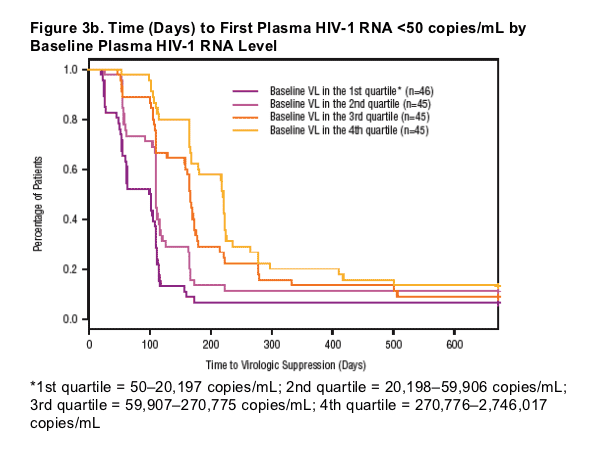
|
| |
|
 |
 |
|
|