 |
 |
 |
| |
TMC125, Darunavir, and Predicting Rescue Failure:
-- TMC125 runs into a resistance wall
-- Once- vs twice-daily enfuvirtide, and predictors of failure
-- Susceptibility to--and drug interactions with--darunavir
-- Trizivir plus tenofovir after toxicity or failure (and nuke preexisting
resistance & predicting failure)
-- Drug breaks with loads above 400 up risk of later rebound
|
| |
| |
Glasgow Meeting: Part 5
8th International Congress on Drug Therapy in HIV Infection
November 12-16, 2006, Glasgow
Mark Mascolini
(note from Jules Levin: the results of the TMC125 study reported below and the others to follow show that for patients with significant resistance to ART drugs the selection process of putting together a new HAART regimen should be done carefully. For patients with very extensive resistance within class or to multiple classes of HIV drugs, bear in mind there are four new drugs recently made available in 3 classes of ART drugs (Merck integrase inhibitor- new class, TMC114 is a PI, TMC125 is a NNRTI, and Maraviroc is a CCR5 inhibitor- new class). In addition in the last 2 years two new drugs became available: tipranavir is a PI and Fuzeon, a fusion inhibitor. When putting together a new combination the results from the study below and the others in this Glasgow Report #5 show the if you have extensive resistance to a class of drugs or more than 1 class, in this case NNRTIs and/or NRTIs, you must consider this in putting together a new HAART regimen. So for example, if you have extensive PI & nuke resistance perhaps you need new drugs from two new classes to bolster adding a new PI, so a regimen might include both the Merck integrase and Maraviroc; or consider a new PI plus TMC125 plus Maraviroc and integrase. And as well you might still have to consider Fuzeon. The point is make sure not to underestimate how much resistance the patient has, don't undertreat!!!)
Among studies of rescue and salvage strategies unveiled in Glasgow, a trial of the nonnucleoside (NNRTI) TMC125 in people who had tried only one drug from this class arched the most eyebrows. Scrutiny of the results shows, though, how unsurprising they are--and how much they have to say about current use of efavirenz, nevirapine, and nucleoside reverse transcriptase inhibitors (NRTIs). Two bands of HIV docs offered intriguing--if small--looks at predicting failure of enfuvirtide plus tipranavir or Trizivir plus tenofovir.
TMC125 runs into a resistance wall
TMC125, the much-touted investigational NNRTI from Tibotec, appeared to stumble in a trial that enrolled people with only one NNRTI failure on their record [1]. But why an NNRTI expected to stifle NNRTI-resistant virus did poorly in this study proved much less shocking than the bare headline: Many study participants had virus already laden with mutations making HIV resistant to both NNRTIs and NRTIs, and many of them recycled NRTIs they had tried earlier. When Tibotec's Brian Woodfall wound up his slide talk, the message seemed clear: TMC125 may indeed have a sturdier barrier to resistance than efavirenz or nevirapine, and it may rein in virus bearing (only) the fearsome K103N mutation. But it's not an anti-resistance elixir that will stifle virus already sporting a string of reverse transcriptase inhibitor mutations.
Woodfall and colleagues in South Africa, Thailand, Argentina, Brazil, Mexico, Russia, Spain, and the UK randomized 57 people to an investigator-selected protease inhibitor (PI) plus two NRTIs and 59 people to 800 mg of TMC125 twice daily plus two NRTIs. (A new TMC125 formulation achieves the same drug exposure as that dose with 200 mg twice daily.) Everyone had NNRTI experience and at least one NNRTI-bred mutation, but no one had tried a PI. Almost all patients randomized to PIs took a ritonavir-boosted PI, 61% getting lopinavir/ritonavir and 32% atazanavir/ritonavir. Researchers picked NRTIs based on resistance test results.
About 80% of study recruits had at least one NRTI mutation, and 51% in the TMC125 arm had two or more NRTI mutations. Among people randomized to TMC125, 37% recycled at least one NRTI and 9% recycled two. NNRTI mutations were even more prevalent when people signed up for this trial: 69.5% in the TMC125 group had two or more NNRTI mutations when the study began, as did 72% in the control arm. Even so, median fold change in susceptibility to TMC125 measured only 2.0 at study entry, compared with 88.0 for nevirapine and 129.8 for efavirenz.
During the first 4 weeks of treatment, median viral loads dropped about 1.3 log copies in the TMC125 arm and 1.5 log in the PI control arm. But loads kept tumbling in the PI group through week 16, whereas the median load rebounded after week 4 in the TMC125 group. Eying these diverging response curves, the trial's independent review board shut down the TMC125 arm.
People with three or more thymidine analog mutations (TAMs) and/or M184V when they started TMC125 got almost no response from their salvage regimen (Table 1). People with two or three TAMs saw their viral load fall for 4 weeks, then rebound almost as abruptly as it dropped. Study participants with as few as one NNRTI mutation when they began TMC125 also endured a rebound after 8 weeks, but that finding does not necessarily mean this new NNRTI cannot control virus with a single NNRTI mutation: TMC125 may have slowed viral replication, but without backup from other drugs in the regimen, it cannot salvage a person with multidrug-resistant virus. In fact a multivariate analysis with statistical adjustment for level of NNRTI and NRTI resistance when salvage began determined that two critical NNRTI mutations by themselves--K103N or Y181C--did not predict response to TMC125.

Grade 3 or 4 side effects proved rare in this brief trial--affecting 10% taking TMC125 and 12% taking a PI. Compared with people assigned to the PI arm, those randomized to TMC125 had fewer lipid or bilirubin leaps and fewer gastrointestinal side effects (3% versus 25%). Central nervous system and psychiatric problems proved equivalent in the two treatment arms. Rash flared in 1 in 10 people taking TMC125, but there were no grade 3 or 4 rashes, and rash incidence did not differ between men and women.
Woodfall and colleagues stressed the tenet that people in whom an NNRTI regimen is failing should stop the nonnucleoside immediately to avoid pileups of NNRTI mutations and thus lower the chance of later responding to TMC125.
Tibotec's Thomas Kakuda presented a thorough review of what's known so far about drug interactions with TMC125 [2]. Click on the link at Reference 2 for details.
Once- vs twice-daily enfuvirtide, and predictors of failure
Enfuvirtide remains the only licensed antiretroviral requiring injection, and having to poke oneself twice a day doesn't make this fusion inhibitor any more popular. To see if two shots at the same time would control HIV as well as two shots in the morning and the evening, US and Canadian clinicians randomized 64 treatment-experienced people to inject 180 mg of enfuvirtide once daily or the standard 90 mg twice daily when starting additional salvage drugs [3]. People in the once-daily group gave themselves two separate injections at different body sites, one right after the other.
Study participants began their enfuvirtide-based salvage regimen with a median CD4 count of 115 (range 2 to 662) and a median viral load of 4.9 log (just under 100,000 copies). About one quarter also started tipranavir in their salvage regimen, and 87% already had an AIDS diagnosis. Twelve people dropped out of the once-daily arm and 10 quit the twice-daily arm before week 48, mostly for reasons unrelated to side effects.
Week 48 intent-to-treat analyses showed that similar proportions in the two study arms reached a viral load below 400 copies, but the twice-daily arm did better in getting their loads under 50 copies and in cutting their loads at least 10-fold (1 log) (Table 2). Twelve of 30 people (40%) in the once-daily arm and 11 of 31 (36%) in the twice-daily arm had a virologic failure, defined as confirmed failure to push the viral load down a half-log at or after week 4. CD4 gains did not differ significantly between the once-daily and twice-daily groups.
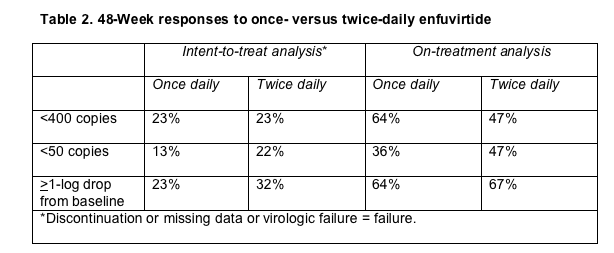
After 48 weeks of treatment 28 of 30 people (93%) in the once-daily group versus 25 of 31 (81%) in the twice-daily group reported at least 85% adherence to enfuvirtide (measured by 4-day recall); that difference fell short of statistical significance (P = 0.14). Twenty-four of 30 people (80%) in the once-daily group versus 18 of 31 (58%) in the twice-daily group reported enfuvirtide adherence of 95% or better, but that difference also fell just shy of statistical significance (P = 0.06). These differences in adherence did not correlate with virologic suppression at the 400-copy level.
Among people who reported at least one injection site reaction, injection site ecchymoses (purple patches), induration (skin hardening), and erythema (redness) were moderately less severe with twice-daily dosing. One person quit the once-daily group because of injection site reactions, while none quit the twice-daily arm for that reason.
Results of this small pilot trial must be judged equivocal because the authors note that the study did not have enough statistical power to demonstrate noninferiority between once- and twice-daily dosing. Injecting two loads of enfuvirtide at the same time may be just as burdensome to some people as injecting the drug at two different times of day. The needle-free Biojector system, not yet available in the US or Europe, proved more acceptable than standard needle injection in a Canadian study [4] and may make two shots at the same time more tolerable.
Having two or more mutations from a set of only six mutations at five protease positions predicted lack of virologic response to salvage regimens including enfuvirtide and ritonavir-boosted tipranavir [5]. But this response predictor rests on analysis of only 24 triple-class-experienced people starting enfuvirtide salvage, so it must be validated in larger groups of patients.
Rodger MacArthur (Wayne State University, Detroit) focused on 24 people with a median pre-enfuvirtide viral load above 100,000 copies and an average CD4 count of 113. That group comprised 13 "responders," including 10 who met the full-response definition (reaching a viral load under 400 copies on enfuvirtide/tipranavir) and 3 who met the partial-response definition (at least a 10-fold drop in viral load). No single mutation predicted lack of virologic response, but two or more from a protease mutation subset--L33F, I47V, I54A/M, V82T, and I84V--had a 100% positive predictive value and a 68% negative predictive value for lack of response (P = 0.006) (Table 3).

Responders had a higher presalvage CD4 count (mean 151, range 6 to 360) than did nonresponders (mean 69, range 2 to 324), a bigger CD4 gain with enfuvirtide/tipranavir (mean 131, range 29 to 353) than nonresponders (mean -10, range -159 to 41), and a lower presalvage viral load (median 60,000 copies, range 1500 to >100,000) than nonresponders (median >100,000, range 15,000 to >100,000).
Susceptibility to--and drug interactions with--darunavir
Mutations conferring resistance to darunavir (TMC114) proved relatively sparse in a UK cohort of 885 PI-experienced people, reported Clive Loveday of the ICVC Charitable Trust, Buckinghamshire [6]. Those findings suggest this new PI will help a fair portion of PI-experienced people in Britain, where darunavir stands likely to be licensed in 2007.
Loveday searched for protease mutations correlating with resistance in the three POWER trials, dividing them into high-impact mutations (V32I, L33F, I47V, I54L, and L89V) and low-impact mutations (V11I, I50V, I54M, G73S, L76V, and I84V) based on whether they arose in 10% or more virologic failures. Most viral samples (60%) came from people currently taking a failing PI regimen, 21% from people taking a non-PI regimen, and 19% from people taking no antiretrovirals; 44% of samples came from people with HIV-1 subtypes other than B.
Only 14% of the cohort--126 people--had one or more darunavir-related mutations, including 83% currently taking a failing PI, 11% off treatment, and 6% taking a non-PI regimen. Of the 126 people with darunavir mutations, 15 (12%) had two such mutations and 13 (10%) had three or more such mutations. Only 2 people had five darunavir-related mutations, including four high-impact mutations. No one in the cohort had the L89V high-impact mutation.
David Back from the University of Liverpool offered a detailed review of drug interaction studies involving darunavir [7], which has already been posted on the NATAP site (click on link at reference 7).
Tibotec researchers found that darunavir cuts concentrations of the antidepressants sertraline and paroxetine and advised clinical monitoring and--if necessary--dose adjustment when these drugs are prescribed with darunavir (click on link at reference 8). The same researchers also recommended clinical monitoring for people on methadone maintenance therapy when starting darunavir, although they found no need for up-front dose adjustments (click on link at reference 9).
Trizivir plus tenofovir after toxicity or failure
An all-nucleoside/nucleotide regimen combining tenofovir with Trizivir (fixed-dose AZT, 3TC, and abacavir) often proved suppressive in people with an incomplete response to another all-nucleoside regimen, including just Trizivir [10]. Brenda Dauer and colleagues at J.W. Goethe University in Frankfurt found that low but detectable viral loads after 24 weeks of Trizivir/tenofovir continued to drop in many people until they reached sub-50 territory. But figuring whether to continue this regimen past 24 weeks in incomplete responders could be tricky, because new NRTI mutations did emerge in people who ultimately endured virologic failure with Trizivir/tenofovir.
This retrospective, single-center analysis of treatment-experienced people included 61 who switched to Trizivir/tenofovir because of toxicity and 53 who switched from a nonsuppressive triple-NRTI regimen (including 35 who switched from nonsuppressive Trizivir). The group had taken antiretrovirals for a median of 6.8 years and had tried a median of seven antiretrovirals. They had a median viral load of 406 copies (range <50 to 1 million copies) and a median CD4 count of 283 (range 2 to 1304). Median follow-up measured 80 weeks (range 3 to 254 weeks), and 78 of 122 people (64%) took Trizivir/tenofovir for at least 48 weeks.
Fifty-four people (88.5%) who switched to Trizivir/tenofovir because of toxicity improved or maintained their viral load; 41 people (77%) switching away from any all-NRTI regimen maintained or improved suppression; and 24 people (69%) switching from a nonsuppressive Trizivir-only regimen reached a load below 400 copies. Overall, 80 of 122 people (66%) pushed their load under 400 copies. Unsurprising clinical predictors of a sub-400 response were a baseline load below 5000 copies and a baseline CD4 count at or above 200 (P < 0.001 for both).
The median 24-week viral load of 72 copies continued to fall into undetectability in the next 24 weeks, and median CD4 count kept climbing after week 24. Among 42 virologic failures (34%), only three failures occurred after treatment week 24. Among 16 people whose virus could be genotyped after Trizivir/tenofovir failure, 9 had a new M41L mutation either alone or with other thymidine analog mutations (TAMs). The baseline mutation combination of M41L, L210W, and T215Y/F proved the only significant predictor of virologic failure with Trizivir/tenofovir (P = 0.03).
Among 85 people with genotypic results before starting Trizivir/tenofovir, 47 (55%) had three or more primary NRTI mutations (range 3 to 10). Nonetheless, the median viral load of this group fell from 12,000 before Trizivir/tenofovir to 758 with the quadruple regimen. But Dauer and colleagues could not sift out genotypic predictors of response in this group. The Frankfurt team cautioned that, despite the generally good results with this regimen, their retrospective data do not support Trizivir/tenofovir alone as a salvage strategy.
Clinicians from four Berlin clinics confirmed the baleful predictive power of L210W among NRTI-experienced people switching to Trizivir/tenofovir [11]. Their analysis of 89 people making this switch also yielded a mutation score that foretold a sub-50-copy response in their patients but needs confirmation in larger groups.
The 89 people seen by Stefan Dupke and Berlin colleagues had a median 72 months of antiretroviral experience with a median of four NRTIs, one NNRTI, and one PI before opting for Trizivir/tenofovir. They had a median CD4 count of 384 but poorly controlled viral replication, with a median load of 72,296 copies. After 3 months of Trizivir/tenofovir, 52 of 85 people (61%) had a sub-50 viral load, after 6 months 45 of 75 (60%) were under the 50-copy threshold, and after 12 months 24 of 41 (59%) had an undetectable load.
Analysis of pre-Trizivir/tenofovir TAMs plus M184V singled out L210W as the strongest predictor of failure at month 3. Only 1 of 40 sub-50-copy 3-month responders (3%) carried L210W, compared with 15 of 27 nonresponders (56%). Weighting the impact of TAMs and M184V on 3-month response in their patients, Dupke and coworkers devised a scoring system with a negative predictive value of 94% and a positive predictive value of 78% for 3-month response to Trizivir/tenofovir (Table 4).
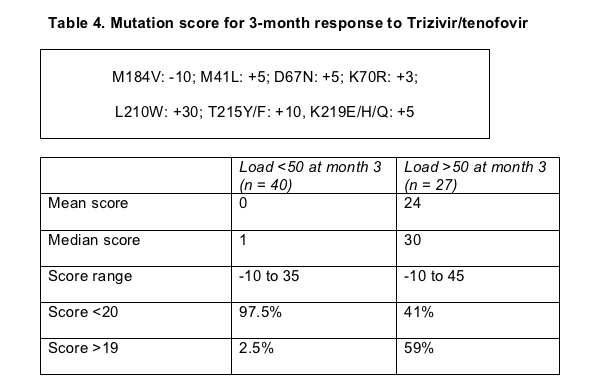
Whether this score proves helpful in predicting prospects for Trizivir/tenofovir in NRTI-experienced patients awaits further analysis in more people.
Drug breaks with loads above 400 up risk of later rebound
Interrupting antiretroviral therapy when the viral load exceeds 400 copies boosts the risk of viral rebound if the interrupter resumes therapy and reaches a load under 50 copies [12]. Three or more treatment breaks make later rebound risk more likely than one or two breaks--even after statistical adjustment for confounding factors. Those conclusions emerged from a study of 12,997 people in the UK CHIC cohort who trimmed their viral load below 50 copies with antiretroviral therapy. From that group, 2246 (17.3%) had taken at least one treatment break, including 2047 who took one or two breaks and 199 who took three or more.
With London colleagues Loveleen Bansi from the Royal Free and University College Medical School undertook the study because earlier research showed that many people who suspend antiretroviral therapy can control HIV when they resume treatment. But no one had analyzed whether such people risk rebound after suppression.
Defining viral rebound as two consecutive loads above 400 copies or one load above 400 copies followed by a regimen change, Bansi counted 2202 rebounds in 31,060 person-years of follow-up among people who never took a drug holiday for a rate of 7.1 rebounds per 100 person-years. Among people who took one or two holidays, she tallied 704 rebounds in 5799 person-years for a rebound rate of 12.1 per 100 person-years. And among people who took three or more holidays, she chalked up 106 rebounds in 472 person-years for a rate of 22.5 per 100 person-years.
Statistical analysis adjusted for whether viral load was detectable at treatment interruption determined that people with an undetectable load at interruption did not have a higher risk of later rebound than people who never suspended therapy. But people who took one or two breaks with a detectable load had a 34% higher rebound risk than noninterrupters, and people who took three or more breaks had an 89% higher risk. Every additional failed regimen on a person's chart also independently raised the risk of rebound. These analyses factored in viral load when antiretroviral therapy began, time since beginning antiretroviral therapy, and the interaction between time with an undetectable load and the number of regimens that failed.
With senior investigator Andrew Phillips, Bansi proposed that rebound rates may rise with a higher number of treatment breaks because resistant virus may emerge during drug holidays with a detectable viral load. Unmeasured variables, such as worse adherence in people who tend to suspend treatment, may also contribute to the inflated rebound risk. The researchers suggest "patients should be made aware that there is a potential risk associated with treatment interruption even if viral suppression is achieved" when restarting treatment after the drug break.
Mark Mascolini writes about HIV infection (markmascolini@earthlink.net).
References
1. Woodfall B, Vingerhoets J, Peeters M, et al. Impact of NNRTI and NRTI resistance on the response to the regimen of TMC125 plus two NRTIs in study TMC125-C227. 8th International Congress on Drug Therapy in HIV Infection. November 12-16, 2006. Glasgow. Abstract PL5.6.
2. Kakuda T. TMC125 in combination with other medications: summary of drug-drug interaction studies. 8th International Congress on Drug Therapy in HIV Infection. November 12-16, 2006. Glasgow. Abstract PL5.2 (http://www.natap.org/2006/Glasgow/Glasgow_21.htm).
3. Woodfall B, Vingerhoets J, Peeters M, et al. Impact of NNRTI and NRTI resistance on the response to the regimen of TMC125 plus two NRTIs in study TMC125-C227. 8th International Congress on Drug Therapy in HIV Infection. November 12-16, 2006. Glasgow. Abstract PL5.6.
4. Harris M, Joy R, Larsen V, et al. Enfuvirtide plasma levels and injection site reactions using a needle-free gas-powered injection system (Biojector). AIDS. 2006;20:719-723.
5. MacArthur RD, Richmond D, Crane LR. Lack of virologic response to the combination of tipranavir/ritonavir + enfuvirtide in treatment-experienced HIV-infected persons with multi-drug resistant virus can be predicted by the presence of two or more of six mutations from five positions in protease. 8th International Congress on Drug Therapy in HIV Infection. November 12-16, 2006. Glasgow. Abstract P205.
6. Loveday C, MacRae E. Susceptibility of a protease inhibitor treatment-experienced UK clinical cohort to TMC114. 8th International Congress on Drug Therapy in HIV Infection. November 12-16, 2006. Glasgow. Abstract PL2.2.
7. Back D. Use of TMC114 in combination with other drugs: guidance from pharmacokinetic studies. 8th International Congress on Drug Therapy in HIV Infection. November 12-16, 2006. Glasgow. Abstract PL5.2 (http://www.natap.org/2006/Glasgow/Glasgow_24.htm).
8. Sekar V, De Paepe E, De Marez T, et al. Pharmacokinetic interaction between TMC114, a new protease inhibitor, and the selective serotonin re-uptake inhibitors, paroxetine and sertraline. 8th International Congress on Drug Therapy in HIV Infection. November 12-16, 2006. Glasgow. Abstract P295 (http://www.natap.org/2006/Glasgow/Glasgow_45.htm).
9. Sekar V, El Malt M, De Paepe E, et al. Pharmacokinetic interaction between TMC114, a new protease inhibitor, and methadone. 8th International Congress on Drug Therapy in HIV Infection. November 12-16, 2006. Glasgow. Abstract P294 (http://www.natap.org/2006/Glasgow/Glasgow_53.htm).
10. Dauer B, Khaykin P, P Gute P, et al. Response to mono-class nucleoside regimen of tenofovir DF + Trizivir in antiretroviral-experienced patients: 48 week results and predictors of response. 8th International Congress on Drug Therapy in HIV Infection. November 12-16, 2006. Glasgow. Abstract P27.
11. Dupke S, Berg T, Hintsche B, et al. Impact of different TAMs on outcome of a HAART regimen with ABC+3TC+AZT (Trizivir) and TDF. 8th International Congress on Drug Therapy in HIV Infection. November 12-16, 2006. Glasgow. Abstract P226.
12. Bansi L, Benzie A, Sabin CA, Phillips AN. Are treatment interruptions associated with higher viral rebound rates in patients with viral suppression? 8th International Congress on Drug Therapy in HIV Infection, November 12-16, 2006, Glasgow. Abstract PL8.3.
|
| |
|
 |
 |
|
|