 |
 |
 |
| |
Fosamprenavir boosted with a single 100 mg capsule of ritonavir as part of a once daily first line regimen in naive patients
|
| |
| |
Reported by Jules Levin
Glasgow, Nov 2006
S De Wit, B Poll, C Necsoi, N Clumeck
Saint-Pierre University, Division of Infectious Diseases, Brussels, Belgium
Several studies have recently reported results from the study of Fosamprenavir 1400 mg once daily boosted by only 100 mg of ritonavir. Here are links to reviews of studies:
The recommended dos schedule for Fosamprenavir in naive patients is 1400 mg FAPV + 200 mg Ritonavir (RTV). Administration of FAPV 1400 mg QD + RTV 100mg QD has been shown to result in equivalent plasma APVCmax and
AVCo-t compared to the standard regimen. Although APV C trough concentrations were 38% lower, it remained 6-fold above the historical mean APV protein binding (90%) adjusted IC50 for wild-type HIV. (Ruane et al. Plasma APV PK and Safety Following Coadministration of FAPV with a Reduced RTV Dose [100mg] Once-Daily [COL10053], 44th ICAAC Wash DC 2004)
This study evaluated the efficacy and safety of a once daily combination of FAPV boosted with a single RTV capsule + tenofovir (TDF) + Lamivudine (3TC) (or Emtricitabine (FTC)) as first line therapy.
This is a prospective, open-label, single arm study of 93 naive patients treated with FAPV 1400 mg + RTV 100 mg + TDF + 3TC )or FTC), as a single dose combination. Virological and immunological outcomes, toxicity, therapy withdrawal and impact on lipid profile were evaluated at weeks 24 and 48.
PATIENT CHARACTERISTICS, n=93 (56 males, 37 females)
Ethnicity-
African: 54%
Caucasian: 39%
Others: 7%
Acquisition of HIV-
Heterosexual: 66%
Homosexual: 24%
Other: 10%
Median baseline viral load: 87,600 c/ml
[IQR (Q1-Q3): 27,350 tp 100,000 c/ml]
Median baseline CD4: 154
[IQR (Q1-Q3): 63 to 259]
RESULTS
At week 24, 83% of the patients were below 50 c/ml and 92% were below 400 c/ml. Median VL decrease was 4.8 log. Median CD4 was 290 cells with a median increase of 127 cells.
34 patients have reached week 48: 83% were below 50 c/ml and 94% below 400 c/ml. Median VL decrease was 4.9 log. Median CD4 was 348 with a median increase of 199 cells.
So far 7 of 93 patients stopped therapy: 2 switched to a 2 pills daily regimen; 5 stopped for toxicity: 4 digestive intolerance, 1 rash.
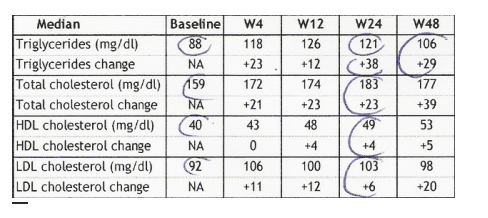
LIPID PROFILE
Median triglycerides were 88 mg/dl at baseline, and 121 at week 24 and 106 at week 48.
Median total cholesterol was 159 mg/dl at baseline, and 183 at week 24 and 177 at week 48.
HDL-cholesterol was 40 mg/dl at baseline, 48 mg/dl at week 12, and 53 mg/dl at week 48.
LDL-cholesterol was 92 mg/dl at baseline, 103 at week 24, and 98 mg/dl at week 48.
|
| |
|
 |
 |
|
|