| |
Non-Invasive Fibrosis Tests Limited
|
| |
| |
"Non-invasive evaluation of hepatic fibrosis: don't count your chickens before they're hatched"
COMMENTARY
Liver fibrosis
Gut March 2006;55:310-312
M Pinzani
Dipartimento di Medicina Interna, Universita degli Studi di Firenze, Viale G B Morgagni, 85, 50134 Firenze, Italy; m.pinzani@dmi.unifi.it
Following the commentary are the two studies upon which the commentary is based.
There is an increasing desire for non-invasive tests to assess both the stage of liver fibrosis and the rate of progression of fibrogenic chronic liver diseases and so reduce the need for repeated liver biopsies. However, higher quality non-invasive diagnostic procedures are still needed.... all of the proposed non-invasive methodologies perform much better in detecting the presence of advanced fibrosis and cirrhosis (that is, METAVIR F3-F4) while they are insufficient in detecting non-significant or low-moderate fibrosis (that is, F0-F2). Therefore, their clinical utility in the decision to treat process is limited. For example, in the case of a patient with chronic hepatitis C virus (HCV) infection, normal liver tests, and exclusion of cirrhosis obtained by one of the non-invasive methods, liver biopsy may still be necessary for treatment decisions.....
Cost pressures and recognition of the limitations of liver biopsy have led to an increasing desire for non-invasive tests to assess both the stage of liver fibrosis and, more importantly, the rate of progression of fibrogenic chronic liver diseases (CLD). In addition, the increase in knowledge of the mechanisms regulating the fibrogenic process in CLD and the consequent knowledge of its dynamic features (that is, scarring as the net result of extracellular matrix deposition and degradation) have led to the need for diagnostic tools with higher prognostic value and flexibility for longitudinal follow up.
There are two main groups of non-invasive methodologies for the evaluation of hepatic fibrosis and its progression. The first group, defined "serum markers", is aimed at predicting fibrosis stage and, possibly, other prognostic information, using parameters measurable in serum. The second group includes methodologies derived from elaboration of parameters obtainable with the current liver imaging techniques (ultrasound, computed tomography (CT) scan, magnetic resonance) or to the innovative use of principles of physics (that is, transient elastography).
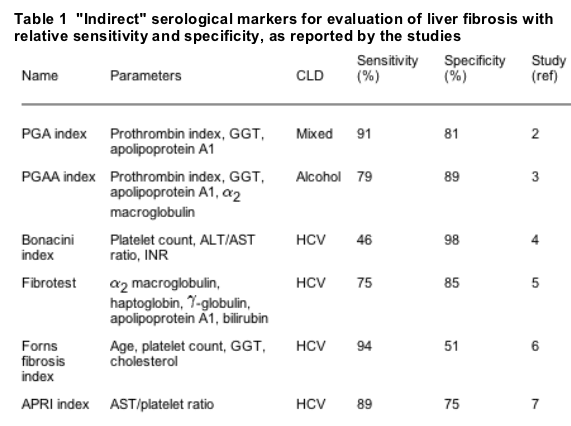
Among serum markers we can distinguish indirect and direct markers.1 Indirect markers are based on single or algorhythmic elaboration of commonly observed alterations in liver function that do not necessarily reflect extracellular matrix metabolism (that is, platelet count, aspartate aminotransferase (AST), and total cholesterol) (table 1). Assessment through indirect markers is in general easy to obtain as it is based on routine laboratory tests or tests easily available in a hospital general laboratory. The best known tests based on indirect markers include: the PGA (prothrombin index, -glutamyl-transpeptidase (GGT), apolipoprotein)2 and the PGAA (same as PGA plus 2 macroglobulin)3; Bonacini index (platelet count, alanine aminotransferase (ALT)/AST ratio, international normalised ratio),4 Fibrotest (2 macroglobulin, haptoglobin, gamma globulin, apolipoprotein, bilirubin),5 Forns fibrosis index (age, platelet count, GGT, cholesterol),6 and APRI index (AST/platelet ratio).7
Direct markers reflect actual extracellular matrix turnover within the liver: total amount of matrix, matrix deposition, matrix removal. They are based on knowledge of the fine cellular and molecular mechanisms of fibrogenesis and they have evolved accordingly in the past decade. This approach, which is an attempt to translate the basic mechanisms into clinically useful applications, is however not simple. Indeed, the requirements for the ideal direct serum markers of liver fibrosis (that is, liver specificity, high sensitivity for fibrogenesis/fibrolysis, known half life and excretion route, easily measurable and inexpensive) are rather difficult to match and an extensive evaluation of their potential is still under investigation. Indeed, the diagnostic performance of these markers and their ability to assess the severity and progression of liver fibrosis has been disappointing, although some individual assays have been shown to be promising in detecting cirrhosis8,9 in alcoholic liver disease (hyaluronic acid),10 or milder fibrosis in non-alcoholic fatty liver disease.11 Other markers have been reported to reflect changes in liver histology attributable to antiviral therapy.12,13
An alternative approach has recently been provided by the European Liver Fibrosis study, an international multicentre cohort study including more than 1000 patients with a wide range of CLD, in which the relationship between serum levels of nine surrogate markers of liver fibrosis, fibrosis/fibrolysis, and fibrosis stage in liver biopsies scored by three expert pathologists, working together or independently, was evaluated.14 Different algorithms, obtained by discriminant analysis of a test set of samples, were then evaluated for their ability to distinguish between individual histology scores. Similar performance characteristics were found with algorithms that incorporated hyaluronic acid, collagen IV, collagen VI, laminin, aminoterminal peptide of procollagen III, tissue inhibitor of metalloproteinase 1 (TIMP-1), and matrix metalloproteinase 2 in varying combinations. Overall, analysing the fibrosis score versus the algorithm discriminant score allowed clear differentiation between "mild" fibrosis (F1-F2) and "significant" fibrosis (F3-F4). The results of this promising approach are now the subject of internal and external validation in larger cohorts of patients with different CLD before they can be proposed as validated tools for extensive clinical use. Finally, attempts have been made to verify the diagnostic efficacy of combinations of direct and indirect serum markers. In a recent study, an algorithm including hyaluronic acid, TIMP-1, and 2 macroglobulin was shown to be effective in differentiating moderate/severe fibrosis (F2-F4) from those with no/mild fibrosis (F0-F1), although accurate delineation between stages was not possible.15
It is predictable that technological advancements in liver imaging will allow reliable and non- invasive assessment of hepatic tissue structure in the future. This possibility would allow elimination of the actual limitations related to sampling error and the need to rely on serum markers. At present, CT and MR can indicate the presence of cirrhosis with high specificity but with very low sensitivity. In addition, these approaches do not allow any delineation of fibrotic stage.16-18 The value of Doppler-ultrasonography (US) is still unclear. Some authors report that compensated cirrhosis could be diagnosed in 90% of cases using some Doppler-US parameters and, above all, the hepatic vein spectrum.19 Other studies report a weak correlation between Doppler-US parameters and portal hypertension.20 Recently published data indicate that US allows monitoring with a sufficient degree of accuracy the transition from advanced fibrosis to cirrhosis by employing three parameters: liver surface nodularity, caudate lobe hypertrophy, and pattern of hepatic venous blood flow.21
In addition to imaging techniques, a non-invasive medical device based on transient elastography (FibroScan) has been proposed for measurement of liver stiffness, considered as a direct consequence of the fibrotic evolution of CLD. The results of two recent studies performed in cohorts of patients with chronic hepatitis C suggest that this system has potential in detecting significant fibrosis (that is, >F2) or cirrhosis.22,23
Several considerations emerge from an overall critical analysis of the proposed methodologies. Firstly, the diagnostic efficacy of non-invasive methods is evaluated in terms of sensitivity (that is, the proportion of truly diseased persons, as measured by the gold standard, who are identified as diseased by the test under study), specificity (that is, the proportion of truly non-diseased persons, as measured by the gold standard, who are so identified by the diagnostic test under study), and negative and positive predictive values (that is, the probability that a person with a positive test is a true positive, or that a person with a negative test truly does not have the disease). Predictive values of a diagnostic test are determined by the sensitivity and specificity of the test, and by the prevalence of the condition for which the test is used. In addition, receiver operating characteristic and area under the receiver operating characteristic curves, commonly employed for graphic illustration of the diagnostic efficacy of a test, basically show the trade off between sensitivity and specificity (any increase in sensitivity will be accompanied by a decrease in specificity). It is therefore clear that the reliability of the test depends heavily on the gold standard and hence on the correct interpretation of liver histology in terms of grading and staging. As a paradox, the trustworthiness of non-invasive markers relies on the diagnostic procedure (that is, liver biopsy) they are supposed to replace because it is imperfect apart from being invasive. Furthermore, even assuming that the stage of fibrosis was obtained by analysing liver biopsies matching the current guidelines24 and that intra- and interobserver variability was maximally reduced (that is not the case in the large majority of studies), qualitative descriptors of stage of fibrosis (that is, for METAVIR F0>F4) are generally employed in the test as quantitative measures. In other words, a score F4 is used as double F2. This is, however, incorrect as fibrosis progression is not a linear process and the rate of progression may be subject to acceleration and deceleration that cannot be standardised. There are in addition several other possible problems related to interpretation of these results.1,25
Finally, it should be stressed that all of the proposed non-invasive methodologies perform much better in detecting the presence of advanced fibrosis and cirrhosis (that is, METAVIR F3-F4) while they are insufficient in detecting non-significant or low-moderate fibrosis (that is, F0-F2). Therefore, their clinical utility in the decision to treat process is limited. For example, in the case of a patient with chronic hepatitis C virus (HCV) infection, normal liver tests, and exclusion of cirrhosis obtained by one of the non-invasive methods, liver biopsy may still be necessary for treatment decisions.
The development of non-invasive methodologies implies that these tools should complement and enrich the information derived from liver histopathology, particularly in the long term dynamic assessment of patients with CLD. However, since the beginning of their history, most if not all non-invasive methods have been referred to as "surrogate" markers, thus implying that they could be cheap and/or fast substitutes for liver biopsy. New systems are continuously proposed and, to date, studies detailing new methodologies have found easy access to top specialised journals. Consequently, it is not rare to find that they are directly used by clinicians for routine use without an appropriate phase of internal and, more importantly, external validation. In the clinical context in which execution of liver biopsy has traditionally been a key requirement for diagnosis and for deciding whether or not to start a treatment, the use of a non-invasive system rapidly becomes a dogma, sometimes with definite ethical and commercial biases. This has led to a scenario where the feelings of hepatologists towards non-invasive evaluation of hepatic fibrosis ranges from confused to very critical.
Luckily, as in the example provided by the paper by Macias and colleagues26 published in the current issue of Gut (see page 409), clinical investigators start wondering how these systems could fit into their everyday routine and how efficient they are in particular subsets of patients. In this paper, the aim was set at a very simple level-that is, detect advanced fibrosis in HCV-human immunodeficiency virus (HIV) coinfection by employing two methods with internal and external validation (Forns index, APRI, and Bonacini index). The results of the study suggest that only a very limited percentage of these patients would benefit from exclusion of liver biopsy. The authors offer a number of possible explanations to justify the low performance of these validated methodologies in coinfected patients: faster fibrosis progression, younger age of patients, concomitant antiretroviral treatment, etc. However, the conclusion is that application of these methods in groups of patients with special features should be even more cautious and further evaluation is needed. This is also strongly suggested by another recent and extensive evaluation of the performance of "simple" non-invasive tests for the detection of advanced fibrosis/cirrhosis in HCV related CLD performed by other authors.27
In another paper published in this issue of Gut, Foucher and colleagues28 report on the diagnostic efficacy of liver stiffness measurement for the detection of cirrhosis in patients with different types of CLD (see page 403). The results of the study indicate that detection of different cut off values of liver stiffness by transient elastography allows allocation of cirrhotic patients into different groups based on clinical manifestations. Although this method requires further evaluation, its clinical utility in the initial clinical workup of cirrhotic patients appears relevant. In this group of patients, transient elastography, together with US, could represent a fast screening tool for prioritising further diagnostic evaluation and, if this is confirmed in longitudinal follow up studies, for allocating cirrhotic patients to a specific class of risk. However, it would be inadequate to suggest that this method could replace a complete diagnostic evaluation of this group of patients.
In conclusion, we are in indeed in a period of transition that hopefully will lead to non-invasive diagnostic procedures of higher quality. These procedures will reduce the need for repeated liver biopsies and will be employed in the dynamic longitudinal follow up of patients with CLD. In any case, before we count our chicken we should know them better.
Prediction of liver fibrosis in human immunodeficiency virus/hepatitis C virus coinfected patients by simple non-invasive indexes
Gut 2006;55:409-414
J Macias1, J A Giron-Gonzalez2, M Gonzalez-Serrano3, D Merino4, P Cano5, J A Mira1, A Arizcorreta-Yarza2, J Ruiz-Morales3, J M Lomas-Cabeza4, J A Garcia-Garcia1, J E Corzo1 and J A Pineda1
1 Hospital Universitario de Valme, Seville, Spain
2 Hospital Universitario Puerta del Mar, Cadiz, Spain
3 Hospital Universitario Virgen de la Victoria, Malaga, Spain
4 Hospital Juan Ramon Jimenez, Huelva, Spain
5 Hospital Universitario Reina Sofia, Cordoba, Spain
ABSTRACT
Background: Liver biopsy is an invasive technique with associated major complications. There is no information on the validity of five non-invasive indexes based on routinely available parameters, estimated and validated in hepatitis C virus (HCV) monoinfected patients, in human immunodeficiency virus (HIV)/HCV coinfected patients.
Aim: To validate these predictive models of liver fibrosis in HIV/HCV coinfected patients.
Patients: A total of 357 (90%) of 398 patients from five hospitals were investigated, who underwent liver biopsy and who had complete data to validate all of the models considered.
Methods: The predictive accuracy of the indexes was tested by measuring areas under the receiver operating characteristic curves. Diagnostic accuracy was calculated by estimating sensitivity, specificity, and positive (PPV) and negative (NPV) predictive values.
Results: The models performed better when liver biopsies >15 mm were used as reference. In this setting, the Forns and Wai indexes, models aimed at discriminating significant fibrosis, showed PPV of 94% and 87%, respectively. Using these models, 27-34% of patients could benefit from exclusion of liver biopsy. If both models were applied sequentially, 41% of liver biopsies could be spared. The indexes aimed at predicting cirrhosis achieved NPV of up to 100%. However, they showed very low PPV.
Conclusions: The diagnostic accuracy of these models was lower in HIV/HCV coinfected patients than in the validation studies performed in HCV monoinfected patients. However, simple fibrosis tests may render liver biopsy unnecessary in deciding anti-HCV treatment in over one third of patients with HIV infection and chronic hepatitis C.
INTRODUCTION
Human immunodeficiency virus (HIV) and hepatitis C virus (HCV) dual infection is highly prevalent among intravenous drug users as a result of shared transmission routes.1 In addition, chronic hepatitis C seems to follow an accelerated course in HIV infection.2 Thus liver failure is increasingly affecting HIV/HCV coinfected patients, as their AIDS free survival is being prolonged.3 For these reasons, HCV infection should be treated in this setting. However, the available treatment for HCV infection is far from optimal. Indeed, HIV/HCV coinfected patients show even worse responses to pegylated interferon plus rivabirin than HCV monoinfected patients.4 In this regard, different strategies have been proposed and evaluated to improve the selection of patients to receive therapy. A rational screening is to perform a liver biopsy and reserve treatment for those with more advanced stages of liver fibrosis. We have shown that this approach would spare up to 40% of coinfected patients from anti-HCV therapy.5
Liver biopsy is an invasive technique. Although infrequent, there are major complications associated with liver biopsy.6 Mild adverse events are more frequent, such as pain, that occurs in more than 30% of biopsied patients.6 Moreover, the procedure is costly7 and can be limited by sampling error as only 1/50 000 of the organ is sampled. Hence some authors have validated models to predict the severity of liver fibrosis by non-invasive means. Some rely on routine laboratory tests, easily available in clinical practice.8,9,10,11,12,13,14 There are only two reported models which have focused on non-invasive diagnosis of liver fibrosis among HIV/HCV coinfected patients.15,16 However, none of the models has been validated by independent authors in this population. In addition, the usefulness of these indexes may be curtailed because some of the predictive markers, such as 2 macroglobulin, haptoglobin, or apolipoprotein A115 and hyaluronic acid16 are not routinely used in clinical practice.
Our aim was to validate five predictive models of liver fibrosis comprising readily available laboratory data, previously constructed and validated in HCV monoinfected patients,8,9,10,11,12,13,14 in HIV/HCV coinfected patients.
DISCUSSION
In this study, we attempted to validate predictive models of liver fibrosis previously estimated in HCV monoinfected patients. We selected models based on data easily available, which had been subject to internal or external validation. The diagnostic accuracy of these models was lower in HIV/HCV coinfected patients than in validation studies performed in HCV monoinfected patients. However, simple fibrosis tests may render liver biopsy unnecessary for deciding therapy against HCV in over one third of patients with HIV infection and chronic hepatitis C, as significant liver fibrosis may be predicted in such patients.
Liver biopsy is an invasive technique with associated morbidity and mortality6 and has a significant cost.7 Because of this, others have attempted to find accurate non-invasive markers of liver fibrosis in chronic hepatitis C. However, only two studies evaluated a models to predict liver fibrosis in HIV infected patients with chronic hepatitis C, but they were based on laboratory parameters not routinely performed which limits their clinical applicability.15,16 In addition, a cumbersome determination, only publicly available very recently, is involved in the calculation of the Fibrotest. This model appears to identify correctly an increased number of HIV/HCV coinfected patients with and without significant hepatic fibrosis, potentially sparing half of the patients from liver biopsy.15 This index has been subject to external validation by independent authors in only one study in patients with chronic hepatitis C without HIV infection.19 Unfortunately, the diagnostic yield of the test was not reproduced by these authors. A novel model, also based on non-routinely used laboratory parameters, has recently been elaborated in HIV/HCV coinfected patients.16 However, this index was not validated in a separate group of patients by the authors. In addition, the performance of the model did not improve previous simpler indexes.
In the present study, the models aimed at discerning significant from non-significant fibrosis reliably predicted the presence of substantial fibrosis. Thus the model of Forns and colleagues8 predicted the presence of significant fibrosis with 96% certainty, and only 4% of patients with a score >6.9 showed non-significant fibrosis. Similarly, the APRI predicted the presence of significant fibrosis with 91% certainty, and misclassified 9% of patients with a score >1.5 who showed F0 to F1 stage fibrosis on liver biopsy. Hence 27-34% of patients would benefit from exclusion from liver biopsy as a tool for deciding anti-HCV therapy. This represents one third of patients potentially excluded from liver biopsy compared with half of patients prevented from liver biopsy in the original studies.8,9 If patients with indeterminate results with the APRI are screened with the Forns model,8 40% of patients could be spared liver biopsy by combining both models, as treatment for HCV could be indicated in these cases.
The APRI has recently been validated in patients with chronic hepatitis C with HIV infection.16 Among HIV coinfected patients, AUROC was 0.71. Liver biopsy size >10 mm was required by the authors in this study. This poor result is in agreement with our findings as the APRI had an AUROC value of 0.73 for liver biopsies 10 mm in our study. We found that with larger liver biopsies as reference, this and other indexes performed better. In this regard, another recent study validated the APRI in patients with chronic hepatitis C without HIV infection.20 At least six portal tracts were required and mean length of the biopsy core was 19 mm. The AUROC of the APRI was 0.80, which is in agreement with our results for larger liver biopsies. Thus the potential variability of liver biopsy, whose diagnostic performance is critically affected by sample size, probably influenced the diagnostic yield of the indexes of fibrosis found in previous studies.
The APRI, the Bonacini model,10 and platelet count showed high levels of certainty in predicting the absence of cirrhosis. This may be reassuring for patients and physicians but is of little clinical use. Thus patients classified as not having cirrhosis still need a liver biopsy for treatment decisions. In contrast, these models did not confidently predict the presence of cirrhosis, as the PPV was low. These disappointing results are in agreement with a recent survey on patients without HIV infection.20
Liver biopsy was used as a reference for the diagnosis of fibrosis. However, the accuracy of liver biopsy for assessing fibrosis is limited by observer and sampling variability. Several studies have assessed interobserver variability in the evaluation of fibrosis. These surveys concluded that reproducibility in staging fibrosis in chronic hepatitis C is good, whatever the scoring system.21 In the present study, interobserver variability was also low. In contrast, sampling variability is more difficult to control. We evaluated the performance of the indexes evaluated in this study for different liver biopsy sizes. We observed that increasing the liver biopsy size from 10 mm to 15 mm improved the diagnostic yield but further increases did not provide better yields. This is in accordance with a previous survey that analysed discordant results between liver biopsy and markers of fibrosis.22 In this study, patients categorised as staging errors on liver biopsy showed smaller biopsy size. However, larger biopsies, >15 mm or >25 mm, were similarly frequent in patients with and without discordant results.
The patients included in this study may not be fully representative of the HIV/HCV population. Only patients who adhered to clinical visits and antiretroviral therapy were selected for liver biopsy. In addition, patients were usually scheduled to undergo liver biopsy only if they had been abstinent from alcohol and other drugs, and HIV infection was stable and under control. Thus there was a possible bias towards patients with less advanced HIV infection and less concomitant alcohol related liver disease. However, the indexes evaluated in this study would probably have performed worse in patients with these associated problems.
The presence of HIV infection changes the course of chronic hepatitis C. Thus coinfected patients show accelerated evolution of chronic hepatitis C, most probably related to immunosuppression.2 These patients are exposed to antiretroviral drugs that are associated with elevations in transaminases, bilirubin, -glutamyl-transpeptidase, and cholesterol, all of which can distort the results of some indexes. Moreover, antiretroviral therapy may alter the course of liver fibrosis in HCV infection.23,24 However, analysis of the study population stratified by CD4+ cell counts and undetectable HIV RNA achieved with antiretroviral therapy did not show changes in the performance of the indexes. Indeed, inclusion of HIV related variables in the validation of the Fibrotest in HIV/HCV coinfected patients did not improve the diagnostic yield of the model.16
In conclusion, therapy for HCV may be decided without liver biopsy evaluation of fibrosis in over one third of HIV infected patients with chronic hepatitis C using simple indexes. Absence of cirrhosis, but not its presence, and significant liver fibrosis can be predicted with certainty in most patients. However, these results clearly need improvement. Hence achieving a non-invasive tool, readily available at the bedside, to predict liver fibrosis in the setting of HIV/HCV coinfection, still requires further investigation.
METHODS
Patients
This retrospective cross sectional study included 398 consecutive patients with HIV/HCV coinfection who were admitted to five hospitals in southern Spain for liver biopsy, from January 1991 to January 2005. Liver biopsies were taken mainly with the aim of establishing the prognosis and indicating therapy for chronic hepatitis C. Eligible patients were those coinfected with HIV and HCV who had undergone liver biopsy, regardless of levels of transaminases. Exclusion criteria included positive hepatitis B surface antigen, other causes of liver disease (autoimmune, tumoral, biliary, or vascular associated liver disease) and prior anti-HCV therapy. Clinical, biochemical, and haematological data within one month of liver biopsy were collected from databases that abstracted patient records.
For each patient a case report form was completed. It included the main demographics, and clinical, laboratory, and virological data at the time of liver biopsy. Recorded demographics included age, sex, risk category, and history of alcohol intake. Data regarding both HIV and HCV infections were recorded, including plasma HIV-RNA, CD4+ T cell counts at the time of liver biopsy, and antiretroviral therapy. Regarding HCV infection, data recorded included genotype, viral load, both at the time of liver biopsy, and date of infection. The latter was estimated as the first year of needle exchange in intravenous drug users. The date of HCV infection was considered as unknown for subjects infected through sexual contact or an undefined source.
This study was approved by each local ethics committee. All patients gave written informed consent for liver biopsy.
Predictive indexes of fibrosis
Among the indexes based on routinely available laboratory tests, we selected those with internal8,9 or external validation.10-14 These indexes were elaborated with the aim of discriminating significant fibrosis, F2 to F4 stages, and/or cirrhosis in HIV seronegative patients:
Indexes aimed at discriminating significant fibrosis
Forns and colleagues8 and Wai and colleagues9 validated their results in a separate group of patients. The index by Forns and colleagues8 is calculated by applying the following regression equation:
7.811-3.131 ln (platelet count (109/l)) + 0.781 ln (-glutamyl-transpeptidase (UI/l)) + 3.467 ln (age (y)) - 0.014 (cholesterol (mg/dl)).
In the estimation of the model by Forns and colleagues,8 the authors excluded drinkers of more than 30 g/day of alcohol and the predominant HCV genotype was 1. The high prevalence of genotype 3 in HIV/HCV coinfected patients3 and its influence on cholesterol levels17 could have affected the accuracy of the index in our study. Because of this, we also analysed a subgroup of patients with alcohol intake <50 g/day and without genotype 3 to validate this model. The index by Wai and colleagues,9 known as the AST platelet ratio index (APRI), is calculated by dividing the aspartate aminotransferase (AST) level (UI/l), expressed as the number of times above the upper limit of normal (ULN), by platelet count (109/l):
AST (/ULN) x100/platelet count (109/l)
Alcohol drinkers were not excluded from elaboration of the index. The cut off points validated by these authors and positive (PPV) and negative (NPV) predictive values of the indexes are shown in table 1.
Indexes aimed at discriminating cirrhosis
APRI was also aimed at predicting cirrhosis.9 The index by Bonacini and colleagues10 has been recently validated by Saadeh and colleagues13 This index was calculated by assigning arbitrary scores to three laboratory parameters and summing them with a possible value of 0 to 11. The laboratory parameters were scored as follows:
1. platelet count (109/l): >340 = 0; 280-340 = 1; 220-270 = 2; 160-219 = 3; 100-159 = 4; 40-99 = 5; <40 = 6.
2. ALT/AST ratio: >1.7 = 0; 1.2-1.7 = 1; 0.6-1.19 = 2; <0.6 = 3.
3. international normalised ratio: <1.1 = 0; 1.1-1.4 = 1; >1.4 = 2.
Alcohol drinkers were not excluded from validation of this index. AST/ALT ratio and platelet count were assessed as indexes in numerous previous surveys.11,12,14 Alcohol drinkers were not excluded in some of the studies that validated AST/ALT ratio11 or platelet count.12 The cut off points validated by these authors and PPV and NPV of the indexes are shown in table 1.
Laboratory methods
Blood determinations
HCV infection was diagnosed when serum specific antibodies were identified by enzyme immunoassay and a recombinant immunoblot assay before 1996. Since December 1996, diagnosis of HCV infection was made when a positive EIA-3 was found and serum viral RNA was detected by either qualitative or quantitative polymerase chain reaction. HCV genotype was determined by line probe assay.
Histological evaluation
Specimens were immediately placed in buffered formalin. After 24 hours of fixation they were embedded in paraffin using routine methods. Histological evaluation was made on sections stained with haematoxylin-eosin and Masson's trichrome. A single pathologist, who was not aware of the clinical data of the patients, evaluated all of the stained sections at each centre. Liver fibrosis was scored following the Knodell histological activity index modified by Scheuer.18 A minimum liver biopsy length of 10 mm was required. Reproducibility of liver fibrosis staging was assessed by blinded re-evaluation by a single pathologist of 50% of the liver biopsies from each centre randomly selected.
Statistical methods
Continuous variables were expressed as median (Q1-Q3) and categorical variables as numbers (percentage). Continuous variables were compared using the Student's t test or the Mann-Whitney U test when appropriate. Categorical variables were compared using the 2 test with Yates' correction or Fisher's test where appropriate.
The predictive accuracy of the indexes was tested by measuring the areas under the receiver operating characteristic curves (AUROC). The cut off points evaluated were those previously validated for each index in HIV uninfected patients. Diagnostic accuracy was calculated by sensitivity, specificity, PPV, and NPV. Significant fibrosis (stages 2-4) or cirrhosis (stage 4) was considered as the disease depending on the index. Performance of the indexes was also assessed using ROC curves in different subpopulations of patients, classified according to the size of their liver biopsies. Agreement between pathologists from different centres and the central pathologist was assessed by the kappa test.
Statistical analysis was carried out using the SPSS 11 statistical software package (SPSS, Chicago, Illinois, USA).
RESULTS
Characteristics of the patients
A total of 357 (90%) of 398 patients had complete data for validation of all of the models. The main characteristics of the study patients by date of liver biopsy are summarised in table 2
Liver biopsy was carried out in 321 (90%) patients after 1997. HCV genotype was 1 in 189 (53%), 3 in 87 (24%), 4 in 46 (13%), and not available in 36 (10%) patients. Median CD4+ cell counts by the time of liver biopsy were 494 (336-653) cells/ml. Median nadir CD4+ cell counts were 255 (136-396) cells/ml. A total of 189 (53%) patients showed undetectable HIV viral load achieved with highly active antiretroviral therapy (HAART) by the date of liver biopsy: 221 (62%) patients received protease inhibitor based HAART before liver biopsy, 57 (16%) were treated with nevirapine based antiretroviral regimens, and 71 (20%) were prescribed efavirenz based HAART. Good agreement was found between each centre's pathologist and the central pathologist in scoring significant fibrosis (kappa scores 0.76-0.80) and cirrhosis (kappa scores 0.87-0.93).
Predictive models of fibrosis applied to HIV infected patients with chronic hepatitis
Models aimed at predicting significant fibrosis
AUROC for the models of Forns and colleagues8 and APRI9 by biopsy length are shown in table 3. Both models performed better for biopsy size 15 mm. Further increases in biopsy length did not improve AUROC. Because of this, 263 (74%) of 357 patients, in which liver biopsy length was at least 15 mm, were selected to validate these indexes. Characteristics of these patients are shown in table 2.
For the model of Forns and colleagues,8 applying the lower cut off level (<4.2), 42 (38%) of 110 patients without significant fibrosis were correctly identified (table 4).
The presence of significant fibrosis could not be excluded with certainty, as 33 (44%) of 75 patients with a score <4.2 had significant fibrosis (NPV 56%). Applying the higher cut off level (>6.9), 66 (43%) of 153 patients with significant fibrosis were correctly identified (table 4). Sixty six (94%) of 70 patients with a score >6.9 showed significant fibrosis. Two of the four falsely classified patients showed F0 and two showed F1 stage at liver biopsy. In the study group, 106 patients reported alcohol intake <50 g/day and harboured genotype non-3. We also applied the model by Forns and colleagues8 to these patients. AUROC was 0.77 (0.65-0.83). Diagnostic accuracy for this analysis was:
1. low cut off (<4.2): sensitivity 79%, specificity 48%, PPV 63%, and NPV 67%;
2. high cut off (>6.9): sensitivity 41%, specificity 98%, PPV 96%, and NPV 60%.
Twenty three (96%) of 24 patients with a score >6.9 showed significant fibrosis.
Using the APRI, for patients with a score <0.5, 36 (33%) of 110 without significant fibrosis would be correctly classified (table 4). Among the 48 patients with a score <0.5, 12 (25%) showed significant fibrosis (75% NPV). Seven showed F2, four patients showed F3, and one patient F4 stage on liver biopsy. For patients with a score >1.5, 78 (51%) of 153 with significant fibrosis were correctly classified (table 4). Seventy eight (89%) of 88 patients with a score >1.5 showed significant fibrosis. Seven of 11 misclassified patients showed F1 and three showed F0 stage on liver biopsy.
A total of 175 patients showed a score <1.5 in the APRI. These patients with indeterminate results were screened with the Forns and colleagues8 index, and 21 (12%) showed a Forns score >6.9. Two patients were misclassified. Thus the diagnostic accuracy of the index of Forns and colleagues8 applied to APRI indeterminate results (score <1.5) was: sensitivity 25%, specificity 98%, PPV 91%, and NPV 64%. Combining both indexes, 109 (41%) patients could be spared from liver biopsy.
The diagnostic accuracy of the indexes was not affected by HIV related variables. Patients with and without undetectable HIV RNA at the time of liver biopsy had an AUROC of 0.77 (0.70-0.82) and 0.75 (0.70-0.80) for the Forns model and 0.80 (0.75-0.84) and 0.79 (0.73-0.82) for the APRI, respectively. Patients with CD4+ cell counts 500 and >500 at the time of liver biopsy had an AUROC of 0.77 (0.72-0.84) and 0.76 (0.71-0.82) for the Forns model and 0.79 (0.74-0.83) and 0.79 (0.75-0.84) for the APRI, respectively.
Models aimed at predicting cirrhosis
AUROC values for these models are shown in table 3. The models performed better for biopsy size 15 mm. Further increases in biopsy length did not improve AUROC. Because of this, patients with a liver biopsy length of at least 15 mm were selected to validate these indexes.
For APRI, 126 (93%) of 135 patients with a score <1 did not have cirrhosis (table 5). Nine (23%) of 40 patients with cirrhosis were classified falsely. For patients with a score >2, 21 (46%) of 46 had cirrhosis and 25 (11%) of 223 without cirrhosis were identified falsely. PPV for both cut off points was low (table 5).
For the Bonacini model,10 all 34 patients with a score below the low cut off did not show cirrhosis (table 5). Twenty (29%) of 68 patients with score above the high cut off had cirrhosis, and 48 (16%) of 297 without cirrhosis were incorrectly identified. PPV for both cut off points was low (table 5).
The AST/ALT ratio was not accurate in predicting the absence or presence of cirrhosis (table 6). Platelet count, using a cut off of 150 109/l, allowed prediction of the absence of cirrhosis with 92% certainty. The presence of cirrhosis was predicted with 33% certainty (table 6).
Diagnosis of cirrhosis by transient elastography (FibroScan): a prospective study
Gut 2006;55:403-408
J Foucher1, E Chanteloup1, J Vergniol1, L Castera1, B Le Bail2, X Adhoute1, J Bertet1, P Couzigou1 and V de Ledinghen3
1 Service d'Hepato-Gastroenterologie, Centre d'Investigation de la Fibrose hepatique, Hopital Haut Leveque, CHU Bordeaux, Pessac, France
2 INSERM E362 IFR 66, Universite Victor Segalen, Bordeaux, France, and Laboratoire d'Anatomie-Pathologique, Hopital Pellegrin, CHU Bordeaux, Bordeaux, France
3 Service d'Hepato-Gastroenterologie, Centre d'Investigation de la Fibrose hepatique, Hopital Haut Leveque, CHU Bordeaux, Pessac, France, and INSERM E362 IFR 66, Universite Victor Segalen, Bordeaux, France
ABSTRACT
Background: Transient elastography (FibroScan) is a new, non-invasive, rapid, and reproducible method allowing evaluation of liver fibrosis by measurement of liver stiffness. In cirrhotic patients, liver stiffness measurements range from 12.5 to 75.5 kPa. However, the clinical relevance of these values is unknown. The aim of this prospective study was to evaluate the accuracy of liver stiffness measurement for the detection of cirrhosis in patients with chronic liver disease.
Methods: A total of 711 patients with chronic liver disease were studied. Aetiologies of chronic liver diseases were hepatitis C virus or hepatitis B virus infection, alcohol, non-alcoholic steatohepatitis, other, or a combination of the above aetiologies. Liver fibrosis was evaluated according to the METAVIR score.
Results: Stiffness was significantly correlated with fibrosis stage (r = 0.73, p<0.0001). Areas under the receiver operating characteristic curve (95% confidence interval) were 0.80 (0.75-0.84) for patients with significant fibrosis (F>2), 0.90 (0.86-0.93) for patients with severe fibrosis (F3), and 0.96 (0.94-0.98) for patients with cirrhosis. Using a cut off value of 17.6 kPa, patients with cirrhosis were detected with a positive predictive value and a negative predictive value (NPV) of 90%. Liver stiffness was significantly correlated with clinical, biological, and morphological parameters of liver disease. With an NPV >90%, the cut off values for the presence of oesophageal varices stage 2/3, cirrhosis Child-Pugh B or C, past history of ascites, hepatocellular carcinoma, and oesophageal bleeding were 27.5, 37.5, 49.1, 53.7, and 62.7 kPa, respectively.
Conclusion: Transient elastography is a promising non-invasive method for detection of cirrhosis in patients with chronic liver disease. Its use for the follow up and management of these patients could be of great interest and should be evaluated further.
INTRODUCTION
Progressive hepatic fibrosis with the development of cirrhosis is a feature of almost all chronic liver diseases. Approximately 10-20% of patients with chronic hepatitis C virus infection have cirrhosis at first clinical presentation, and as many 20-30% of those who do not have cirrhosis will eventually develop this condition and its complications within one or more decades.1-3 These complications are liver failure, ascites, variceal bleeding, portal-systemic encephalopathy, and hepatocellular carcinoma.3
Liver biopsy is currently considered the gold standard for assessing hepatic fibrosis. However, it is an invasive and painful procedure,4 with rare but potential life threatening complications,5 limiting its acceptance and repetition in usually asymptomatic patients. In addition, the accuracy of liver biopsy in assessing fibrosis may be questioned because of sampling error and interobserver variability, which may lead to understaging of cirrhosis.6-9 Thus there is a need to develop and validate non-invasive tests that can accurately reflect the full spectrum of hepatic fibrosis, cirrhosis, and its severity in liver diseases.
Transient elastography (FibroScan; Echosens, Paris, France) is a novel, rapid, and non-invasive technique which measures liver stiffness.10 Briefly, this system is equipped with a probe consisting of an ultrasonic transducer mounted on the axis of a vibrator. A vibration of mild amplitude and low frequency is transmitted from the vibrator to the tissue by the transducer itself. This vibration induces an elastic shear wave which propagates through the tissue. In the meantime, pulse-echo ultrasonic acquisitions are performed to follow the propagation of the shear wave and measure its velocity, which is directly related to tissue stiffness (or elastic modulus). The harder the tissue, the faster the shear wave propagates. Recent reports have shown that liver stiffness measurement using FibroScan allowed accurate prediction of hepatic fibrosis in patients with chronic hepatitis C virus infection.11-14 In patients with chronic hepatitis C, we have shown that liver stiffness measurements ranged from 2.4 to 75 kPa, with a median value of 7.4 kPa.14 Based on the stiffness measurement distribution according to fibrosis stage and receiver operating characteristic (ROC) curves, we found that the cut off value for cirrhosis was 12.5 kPa. However, the clinical relevance of theses values (from 12.5 to 75 kPa) in cirrhotic patients is unknown.
The aim of this prospective study was to assess the accuracy of transient elastography for the detection of cirrhosis in clinical practice in a large cohort of consecutive patients with chronic liver disease.
DISCUSSION
The results of the present study conducted prospectively in a large cohort of patients with chronic liver disease showed that transient elastography is an efficient technique for the diagnosis of cirrhosis and its severity. With a cut off value of 17.6 kPa, negative and positive predictive values for the diagnosis of cirrhosis were 92% and 91%, respectively. We established the cut off value for complications of cirrhosis, with a negative predictive value of more than 90%. These cut off values were 27.5 kPa for the presence of oesophageal varices stage 2/3, 37.5 kPa for cirrhosis Child BC, 49.1 kPa for a past history of ascites, 53.7 kPa for hepatocellular carcinoma, and 62.7 kPa for oesophageal bleeding. In clinical practice, such results could be of major relevance for follow up of patients with severe fibrosis or cirrhosis.
In the present study, cut off values for the diagnosis of fibrosis were slightly different from those previously published.13,14 In published studies, for a diagnosis of fibrosis F>2, cut off values ranged from 7.1 to 8.8 kPa.13,14 In our study, this cut off was 7.2 kPa. For the diagnosis of fibrosis F>3, cut off values ranged from 9.5 to 9.6 kPa.13,14 In our study, this cut off was 12.5 kPa. Finally, for the diagnosis of cirrhosis, cut off values ranged from 12.5 to 14.6 kPa.13,14 In our study, the cut off for diagnosis of cirrhosis was 17.6 kPa. These differences could be due to the study population which comprised patients with chronic liver diseases of various aetiologies. In the two previous studies, only patients with chronic HCV infection were included. Secondly, in the two previously published studies, cut off values were chosen to maximise the sum of sensitivity and specificity whereas in this study we chose cut off values to have a positive predictive value of more than 90%, which favours specificity.
Cirrhotic patients with alcoholic liver disease had higher liver stiffness values than cirrhotic patients with chronic hepatitis C. Indeed, patients with chronic hepatitis C are diagnosed at an early stage than alcoholic patients, with a histological diagnosis without clinical complications of cirrhosis. In contrast, patients with alcoholic disease are diagnosed later, when clinical complications of cirrhosis occur.
To our knowledge, this study is also the first to compare liver stiffness measurement and fibrosis stage assessed on liver biopsies in a large population of patients with chronic liver disease of various aetiologies. The diagnostic performances for F>F2, F>F3, and F = F4 obtained in these different aetiological populations were similar to those obtained in previously published studies conducted only in HCV patients.13,14 In our study, the proportion of patients with advanced fibrosis stages (F>3) was higher than in the general population so diagnosis performance might be lower in the general population. However, these results indicate that liver stiffness measurement could be used to evaluate liver fibrosis in chronic liver diseases, whatever the aetiology. A liver biopsy was not performed in all patients. However, no statistically significant difference was observed between the characteristics of patients with or without liver biopsy.
Needle liver biopsy has been used as the "gold standard" for assessment of liver fibrosis. Usually, the diagnosis of cirrhosis is based on a biopsy specimen that only represents 1/50 000 of the total liver mass.5 Furthermore, inter- and intraobserver discrepancies of 10% to 20% in assessing hepatic fibrosis have been reported, which may lead to understaging of cirrhosis.7,15 Therefore, while liver biopsy remains the "gold standard", both the clinician and researcher should view the results with some reservations and should interpret the findings in the broader clinical context. Although the risks of liver biopsy can be reduced by operator experience and using ultrasound guidance, several known risks of obtaining the tissue, such as pain, bleeding, pneumothorax, haemothorax, bile peritonitis, haemobilia, puncture of the kidney and intestine, infections, anxiety, and even death, do not seem to be entirely avoidable.4,5,16,17
Liver stiffness measurement using FibroScan is reproducible and independent of the operator12 and explores a volume of liver parenchyma which can be approximated to a cylinder of 1 cm in diameter and 4 cm in length. This volume is 100 times larger than the biopsy specimen volume and is thus much more representative of the entire hepatic parenchyma. Correlation of liver stiffness and fibrosis stage is not affected by steatosis or activity grade.13
A variety of indirect markers of cirrhosis have been evaluated, including variables such as the AST/ALT ratio,16,18,19 platelet count,20 prothrombin index,21 APRI,22 and Fibrosure.23 All of these methods have been evaluated in HCV patients. Our study showed that liver stiffness measurement using transient elastography is a new non-invasive method for the diagnosis of cirrhosis in either HCV infected patients or in other patients with chronic liver diseases. With FibroScan, AUROC for the diagnosis of cirrhosis was 0.96. None of the other non-invasive methods provide such an accurate value.
Ultrasonographic or radiological evaluation of the liver to assess fibrosis has been limited to identification of individuals with cirrhosis and its complications. In a study of 243 patients, the diagnosis of cirrhosis could be made by ultrasound with an accuracy of 82-88%.24 However, significant interobserver variability and inability to gather all of the required measurements, due to technical problems, limit the value of ultrasonography. Using pulsed Doppler ultrasonography, Chawla and colleagues showed that there was a significant decrease in portal flow velocity in patients with Child's C cirrhosis compared with controls and patients with Child's A and Child's B cirrhosis.25 With liver stiffness measurements using FibroScan, there is no intra- or interobserver variability and the technique is reproducible.12 With only five successful acquisitions and a 30% success rate, the result of liver stiffness measurements correlated with fibrosis stage.
Cirrhosis places the patient at risk of clinical complications, such as portal hypertension, and variceal rupture is the second cause of death in cirrhosis, justifying early screening for oesophageal varices. The usual means of diagnosing oesophageal varices is upper gastrointestinal endoscopy. However, endoscopy can be considered invasive due to the technique and level of discomfort. Non-invasive methods for diagnosis need to be developed. In a study of cirrhotic patients, diagnosis accuracy of oesophageal varices was 72% with two variables: platelet count and prothrombin index.20 In another study, the positive predictive value of platelet count for the presence of oesophageal varices was 67%.26 In our study, 20 patients did not undergo endoscopy because of refusal or because an endoscopy had been performed 1-2 years before the study. With a negative predictive value of 95%, a liver stiffness measurement of >27.5 kPa was associated with the presence of oesophageal varices stage 2 or 3, independently of the cause of cirrhosis. Thus a prospective study in clinical practice should be done evaluating the relevance of oesophageal varices screening in patients with liver stiffness values of more than 27.5 kPa.
Maharaj et al, by performing three transcutaneous biopsies in the same patients using different entry points, reported that, in proven cirrhotic patients, a histopathological feature of cirrhosis was present in all three biopsy specimens in only 50% of patients.6 Similarly, Abdi and colleagues27 performed several post mortem biopsies and showed that the diagnosis of cirrhosis could be obtained from one biopsy specimen in only 16 of 20 cases. According to Bedossa et al, sampling variation in liver fibrosis is a significant limitation in the assessment of fibrosis with liver biopsy.8 Thus some cirrhotic patients are misclassified as F3 patients. To determine the usefulness of liver stiffness measurements with FibroScan in clinical practice, to include all patients with cirrhosis, even real cirrhotic patients with an incorrect F3 fibrosis at liver biopsy examination, we evaluated the diagnostic accuracy of liver stiffness for complications of cirrhosis in F3F4 patients.
In patients with hepatocellular carcinoma, ultrasonography was performed before FibroScan in order to evaluate liver stiffness in a part of the liver without hepatocellular carcinoma. Therefore, tumour stiffness did not influence the results of FibroScan. In this study, only a small number of patients had hepatocellular carcinoma. Thus the role of FibroScan in assessing the risk of hepatocellular carcinoma needs further investigations. At last, in clinical practice (need for surgery, medical treatments, etc) the risk of cirrhosis decompensation could be excluded in patients with liver stiffness measurements <27 kPa (cut off value for Child A stage).
In conclusion, liver stiffness measurement is a good method for the diagnosis of fibrosis and cirrhosis, irrespective of the cause of liver disease. Values in cirrhotic patients ranged from 17.6 to 75.4 kPa. Liver stiffness measurement may be accurate for assessing the severity of cirrhosis. However, a longitudinal cohort study needs to be performed to predict the complications of cirrhosis using FibroScan so that screening for complications of cirrhosis, and close follow up, could be performed.
RESULTS
Patients
A total of 758 patients were enrolled. Forty seven patients (6.2%) were excluded because of unsuccessful liver stiffness measurement, mostly due to overweight (31 patients had less than five valid measurements and 16 had a success rate lower than 30%). Thus 711 patients were analysed. Their characteristics at the time of the FibroScan examination are summarised in table 1. They were 403 males, with a mean age of 52 (13) years. Aetiologies of chronic liver diseases were: HCV (n = 398) or HBV infection (n = 43), alcoholic liver disease (n = 89), HCV infection and alcoholic liver disease (n = 26), HCV and human immunodeficiency virus infection (n = 24), non-alcoholic steatohepatitis (n = 26), haemochromatosis (n = 17), cholestatic liver disease (n = 13), and other (n = 75).
A total of 354 patients (49.8%) had undergone liver biopsy at the time of the liver stiffness measurement. Fibrosis stage distribution was as follows: 111 patients (31.4%) had no or mild fibrosis (F0F1), 99 patients (28.0%) had moderate fibrosis (F2), 49 patients (13.8%) had severe fibrosis (F3), and 95 patients (26.8%) had cirrhosis (F4). Median biopsy length was 16.5 mm.
For cirrhotic patients, Child-Pugh score was A in 70 (73.7%) cases, B in 15 cases (15.8%), and C in 10 (10.5%) cases.
Relationship between liver stiffness and histological parameters
Liver stiffness measurements ranged from 2.4 to 75.4 kPa (median 6.8). Mean number of measurements per patient was 12.6 (4.1) (range 7-33). A total of 696 of 711 patients had 10 valid measurements. The other patients had 5-9 valid measurements. Therefore, the success rate was 84.7 (18.3)% (range 30-100%).
Figure 1 shows box plots of liver stiffness for each fibrosis stage. For patients with severe fibrosis (F3 and F4), median liver stiffness was 18.7 kPa (range 3.3-75.4). For patients with cirrhosis, median liver stiffness was 31.1 kPa (range 5.5-75.4). Liver stiffness was significantly different between patients according to their fibrosis stage (p<0.001) and significantly correlated with fibrosis stage (r = 0.73, p<0.0001).
Figure 2 shows the diagnostic value (ROC curves) of liver stiffness measurement for different degrees of fibrosis: moderate fibrosis or more (F>F2), severe fibrosis or more (F>F3), and cirrhosis (F = 4). Corresponding AUROC values (95% confidence interval) were 0.80 (0.75-0.84) for F>F2, 0.90 (0.86-0.93) for F>F3, and 0.96 (0.94-0.98) for F = 4. Based on the stiffness measurement distribution according to fibrosis stage and ROC curves, the best discriminant cut off levels were determined (positive predictive value of at least 90%). These cut off levels were 7.2 kPa for moderate fibrosis or more, 12.5 kPa for severe fibrosis or more, and 17.6 kPa for cirrhosis (table 2). Twenty of 95 cirrhotic patients (21%) had liver stiffness measurements less than 17.6 kPa. For patients with viral and alcohol related cirrhosis, median liver stiffness values were 23 kPa and 52.4 kPa, respectively (p<0.001).
Correlation between elastography measurement and parameters of severity of cirrhosis
For all patients as well as for the subgroup of F3F4 patients, as indicated in table 3, liver stiffness was significantly (p<0.05) correlated with clinical parameters (past history of bleeding varices, hepatocellular carcinoma, or ascites), biological parameters (platelet count, prothrombin time, factor V, albumin, and total bilirubin) and morphological parameters (oesophageal varices stage 2/3, ultrasonographic splenomegaly). In cirrhotic patients, liver stiffness was significantly (p<0.0001) correlated with Child-Pugh score (r = 0.517).
Diagnosis accuracy of liver stiffness measurement in clinical practice
For patients with severe fibrosis or more (F>F3), AUROC values for the performances of the liver stiffness measurements in detecting complications of cirrhosis are given in table 4. With a negative predictive value >90%, the cut off for the presence of oesophageal varices stage 2/3 was 27.5 kPa, for cirrhosis Child BC 37.5 kPa, for a past history of ascites 49.1 kPa, for hepatocellular carcinoma 53.7 kPa, and for oesophageal bleeding 62.7 kPa (fig 3). According to these negative predictive values, the usefulness of the liver stiffness measurements with FibroScan in clinical practice is indicated in fig 4.
PATIENTS AND METHODS
Patients
Between June 2003 and September 2004, all consecutive patients with chronic liver diseases seen at the Hepatology Unit of Haut-Leveque Hospital (University Hospital of Bordeaux, Pessac, France) were prospectively included. Determination of the aetiology of chronic liver disease was made using standard diagnostic criteria. Hepatitis C virus (HCV) or hepatitis B virus (HBV) was diagnosed by serological detection of hepatitis C antibodies (with positive serum HCV-RNA by polymerase chain reaction) and hepatitis B surface antigen, respectively. Alcoholic liver disease was diagnosed in those with consumption of at least 40 g of alcohol daily for five years or more. All other diseases were diagnosed as usual. The study protocol conformed to the ethical guidelines of the 1975 Declaration of Helsinki. Patients were enrolled after written informed consent was obtained. All patients consented to the study.
Characteristics of patients
For all patients, the following parameters were determined at the time of the liver stiffness measurement. Clinical parameters included weight, height, past history of ascites or bleeding varices, and hepatocellular carcinoma. Biological parameters included aspartate aminotransferase (AST), alanine aminotransferase (ALT), -glutamyl-transpeptidase, total bilirubin, platelet count, prothrombin time, factor V, albumin, and fetoprotein. Morphological parameters included oesophageal varices (after upper gastrointestinal endoscopy) and ultrasonographic splenomegaly. As ascites is a physical limitation to the technique because elastic waves do not propagate through liquids, patients with ascites were excluded.
Liver stiffness measurement
Details of the technical background and examination procedure have been previously described.11,12 Measurements were performed on the right lobe of the liver through intercostal spaces on patients lying in the dorsal decubitus position with the right arm in maximal abduction. The tip of the probe transducer was covered with coupling gel and placed on the skin, between the rib bones at the level of the right lobe of the liver. The operator, assisted by an ultrasonic time-motion image, located a liver portion of at least 6 cm thick free of large vascular structures. Once the measurement area had been located, the operator pressed the probe button to start an acquisition. Measurement depth was between 25 mm and 65 mm below the skin surface. Measurements which did not had a correct vibration shape or a correct follow up of the vibration propagation were automatically rejected by the software. Up to 10 successful measurements were performed on each patient. Success rate was calculated as the ratio of the number of successful measurements over the total number of acquisitions. The results are expressed in kilopascal (kPa). Median value of the successful measurements was kept as representative of liver stiffness. The whole examination duration was less than five minutes. Only liver stiffness measurements obtained with at least five successful measurements and a success rate of at least 30% were considered reliable.
Liver histology and quantification of liver fibrosis
For patients who had liver biopsy at the time of the liver stiffness measurement (usual indications for liver biopsy), liver biopsy was fixed in formalin and paraffin embedded. Liver stiffness measurements were performed just before liver biopsy. All biopsy specimens were analysed independently by an experienced pathologist blinded to the clinical data and the results of the liver stiffness measurements.
Liver biopsies that contained less than 10 portal tracts (except for cirrhosis) were excluded from the histological analysis. The length of each liver biopsy specimen was also established (in millimetres). Fibrosis was staged according to the METAVIR scoring system as follows: no or mild fibrosis (no fibrosis or portal fibrosis without septa, F0F1), moderate fibrosis (portal fibrosis and few septa, F2), severe fibrosis (numerous septa without cirrhosis, F3), and cirrhosis F4.15
Statistical analysis
The Kruskal-Wallis non-parametric analysis of variance was used to compare liver stiffness among the different METAVIR fibrosis stages. For fibrosis and complications of cirrhosis, the diagnostic performance of liver stiffness measurement was assessed using ROC curves. A subject was assessed as positive or negative according to whether the non-invasive marker value was greater than, less than, or equal to a given cut off value. Associated with any cut off value was the probability of a true positive (sensitivity) and the probability of a true negative (specificity). The ROC curve is a plot of sensitivity versus 1-specificity for all possible cut off values. The most commonly used index of accuracy is area under the ROC curve (AUROC), values close to 1.0 indicating high diagnostic accuracy. ROC curves were thus constructed for detection of patients with METAVIR fibrosis stage 2 or more (F>2), METAVIR fibrosis stage of 3 or more (F>3), and cirrhosis (F = 4).
Spearman coefficients of correlation and their associated probability (p) were used to evaluate the relationship between parameters. As some data were missing for some patients, the number of patients (n) included in the calculation of each correlation coefficient was specified.
Optimal cut off values for liver stiffness were chosen to optimise the predictive value according to the diagnostic question. Statistical analyses were performed with NCSS 2004 software (Statistical Systems, Kayville, Utah, USA).
|
|
| |
| |
|
|
|