 |
 |
 |
| |
Antiviral Efficacy and Emergent PI Substitutions in Treatment-Naive Subjects Receiving Atazanavir +/- Ritonavir-Based HAART in Subtype B and Non-Type B HIV-1 Infection
|
| |
| |
Reported by Jules Levin
16th Intl HIV Drug Resistance Workshop, June 12-16, 2007, Barbados
Donnie McGrath, Victoria Wirtz, Rong Yang, Marina Mathew, Anna Rightmire, Steven Schnittman for the BMS AI424089 Study Group
Research and Development, Bristol-Myers Squibb Company, Wallingford, CT, USA
AUTHOR CONCLUSIONS
Both ATV300/RTV- and ATV400-based HAART had a potent antiviral effect against the 2 major subtype populations prevalent in the HIV pandemic today, subtypes B and C
Virologic failure rates were low on both regimens, but virologic failure was observed in fewer subjects on ATV300/RTV
Among subjects with known HIV subtype, phenotypic resistance to ATV was observed only in a single subject with subtype B who received ATV400
No phenotypic resistance developed to any other PI
IAS-defined major PI substitutions (I50L and N88N/S) were only observed on ATV400 and only in patients with subtype B infection
Conclusions regarding virologic response in subjects infected with viral subtypes other than B and C cannot be made due to the limited numbers of subjects infected with these subtypes in this study
INTRODUCTION
Atazanavir (ATV) is a potent, generally well-tolerated once-daily HIV-1 protease inhibitor (PI) studied in combination therapy and boosted with ritonavir (RTV)1-3
HIV-1 infection is characterized by genetic diversity wherein distinct viral subtypes (and circulating recombinant forms [CRFs]) are expanding in different geographical regions of the world4
In vitro analyses indicate that HIV-1 subtypes A, B, C, D, F, G, and J and CRFs AE and AG are susceptible to ATV5
Early ATV clinical studies were conducted primarily in patients with subtype B HIV-1 viruses. However, the majority of worldwide HIV infections involve non-subtype B HIV-1 and there are limited data on the clinical efficacy and emergent substitutions in treatment-naive patients on ATV-based HAART with non-subtype B infection
The BMS 089 study was a randomized, open-label, multicenter, 96-week study
designed to compare the efficacy and safety of ATV 300 mg/RTV 100 mg with that of ATV 400 mg in HIV treatment-naive subjects
In a 48-week analysis of this study, virologic response rates were 86%; 85% (ATV300/RTV; ATV400) and 75%; 70% (ATV300/RTV; ATV400), for the < 400 and < 50 copies/mL TLOVR analyses, respectively (non-inferiority, delta -10%,
ATV300/RTV - ATV400)3
This current analysis examines the emergence of PI substitutions and antiviral
efficacy at 48 weeks in subjects treated with ATV who have varied HIV-1 subtypes
METHODS
In the 089 study, treatment regimens included:
--ATV 300 mg once a day (QD) + RTV 100 mg QD + lamivudine (3TC) 300 mg QD + stavudine (d4T) extended-release (not commercially available) 100 mg QD
--ATV 400 mg QD + 3TC 300 mg QD + d4T extended-release 100 mg QD
HIV genomic sequencing was conducted using the GeneSeq HIV at Monogram Biosciences, Inc. (South San Francisco, California). The subtypes were derived from the sequence data
For subjects with HIV RNA subtype results, demographics were summarized at
baseline, and analysis of response rates based on Virologic Response - Observed Cases (VR-OC), virologic suppression, and immunologic response were summarized at baseline and each scheduled visit through week 48 by treatment regimen and by subtype
Genotypic and phenotypic data on subjects with virologic failure through 48 weeks were analyzed
RESULTS
Demographics and Baseline Characteristics
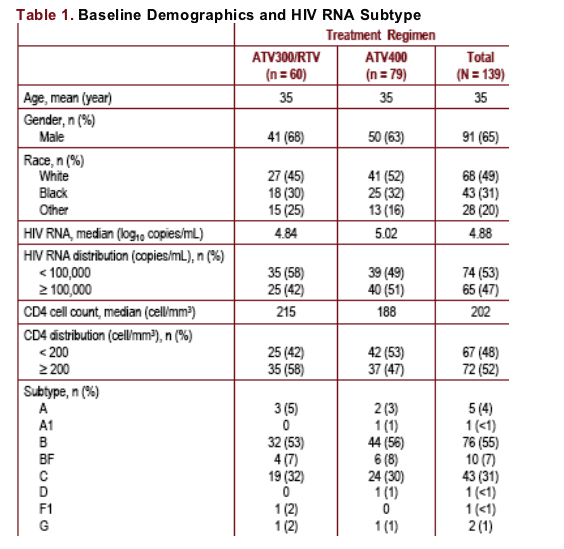
Of the 200 patients randomized, 139 subject samples were available for subtype analysis (ATV300/RTV 60 subjects; ATV400 79 subjects). Baseline demographics and subtype characteristics are shown in Table 1
Subjects (N = 139) were well matched between treatment groups and baseline characteristics were comparable (Table 1); baseline characteristics were similar to those of all subjects in the primary study cohort (N = 200)
The baseline median HIV RNA for the 2 largest subtype groups, B and C, were
4.94 log10 copies/mL and 4.82 log10 copies/mL, respectively
--These are consistent with the median baseline HIV RNA of 4.88 log10 copies/mL observed in the overall study
Virologic Response by Subtype Group
At week 48, VR-OC rates for the groups with the largest sample sizes, subtypes
B and C, were consistent with the overall rates for the study for both HIV RNA
< 400 copies/mL (both regimens 93%)3 and < 50 copies/mL (ATV300/RTV 87%; ATV400 76%)3 (Table 2)

Virologic Suppression
Mean changes from baseline in HIV RNA for subtypes B and C at week 48 were
also consistent with the overall study rates (ATV300/RTV -3.12 log10 copies/mL; ATV400 -3.10 log10 copies/mL) (Table 3)

Immunologic Response
CD4 mean changes from baseline through week 48 for subtypes B and C were consistent with those of the primary study cohort (ATV300/RTV 189 cells/mm3; ATV400 224 cells/mm3)3 (Table 4)

Emergent PI Mutations in Patients with Virologic Failure at Week 48
Overall, virologic failure was reported in 13 subjects (ATV300/RTV 3 subjects;
ATV400 10 subjects)
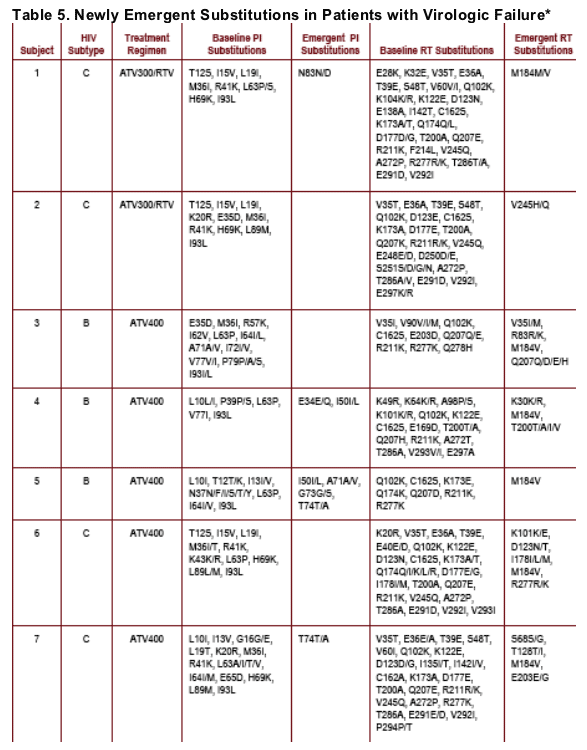
Baseline and on-study genotypic data were available for 10 subjects with known HIV subtypes who had protocol-defined virologic failure by week 48; 2 ATV300/RTV subjects and 8 ATV400 subjects; 6 with B virus and 4 with C virus
Newly emergent substitutions
--All subjects had baseline PI substitutions (Table 5)
--The ATV-specific substitution of I50L emerged in 3 subjects, all in the
ATV400 arm; in 2 it was present as a mixed population with wild-type virus
--The isolate with the pure I50L substitution also had newly emergent M36M/V, A71V, N88N/S substitutions
--In the 2 containing a mixture of 150I/L isolates, few newly emergent substitutions clearly associated with PI resistance were present
(A71A/V, G73G/S)
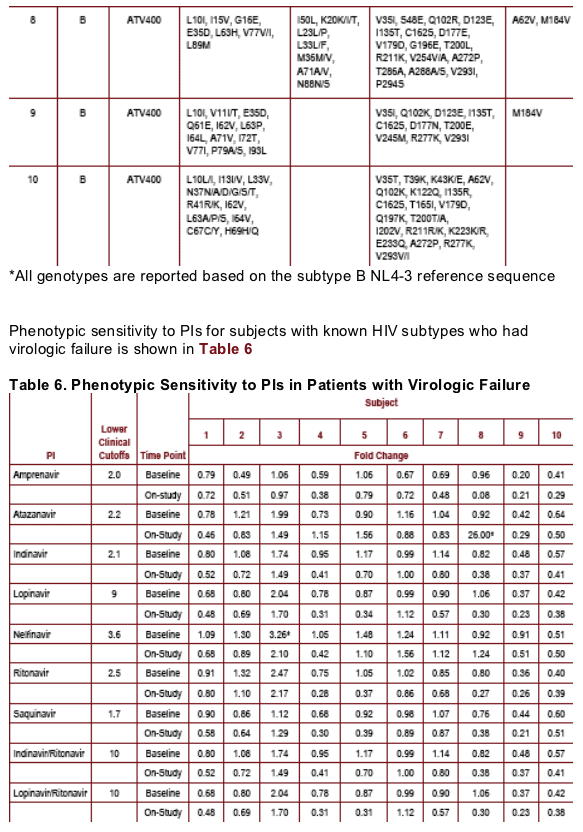
--Phenotypic resistance to ATV (fold change = 26) was only observed in the
isolate that had the pure I50L substitution (Table 6)
--No phenotypic resistance developed to any other PI
--No IAS-defined major PI mutation was observed through 48 weeks on ATV300/RTV
REFERENCES
1. Becker S. Expert Rev Anti Infect Ther. 2003;1:403-413.
2. Sanne I et al. J Acquir Immune Defic Syndr. 2003;32:18-29.
3. Malan N et al. Presented at: 13th CROI, Denver, Colorado, February 5-8, 2006. Abstract 107LB.
4. Spira S et al. J Antimicrob Chemother. 2003;51:229-40.
5. Reyataz [package insert]. Bristol-Myers Squibb Company; 2006.
|
| |
|
 |
 |
|
|