 |
 |
 |
| |
Genotypic relationships between tipranavir and darunavir resistance in protease inhibitor experienced patients
|
| |
| |
Reported by Jules Levin
16th Intl HIV Drug Resistance Workshop, Barbados, June 12-16, 2007
DB Hall1, J Scherer1, J Schapiro2, CAB Boucher3, J Baxter4, V M Kohlbrenner1, D Mayers1*
1Boehringer Ingelheim Pharmaceuticals, Ridgefield, CT, USA; * No longer with Boehringer Ingelheim; 2National Hemophilia Center, Israel; 3Department of Virology, University Medical Center Utrecht, the Netherlands;
4Cooper Hospital/UMDNJ-Robert Wood Johnson Medial School, New Jersey, USA
AUTHOR CONCLUSIONS
The correlation between fold change by virtual phenotype of tipranavir and darunavir for protease inhibitor experienced patients failing a protease inhibitor based regimen is modest.
PI-associated mutations 10F, 48V, 50V, and 54L are associated with susceptibility to TPV and reduced susceptibility to DRV. These mutations are selected against during TPV therapy, leading to increased susceptibility to DRV.
PI-associated mutations 82L, 82T and 84V are not associated with reduced susceptibility to DRV when emergent during TPV therapy.
Tipranavir before darunavir increases susceptibility to darunavir for the circulating virus population. The impact of archived mutations on durable response to
subsequent treatment with darunavir is unknown and can only be determined by clinical investigation.
Abstract
Purpose of the Study: Both Tipranavir (TPV) and Darunavir (DRV) are indicated for protease inhibitor experienced patients. In the RESIST (TPV) trials all patients had a screening genotype and selected virologic rebound TPV patients had on-treatment specimens sequenced. Genotype and virtual phenotype were analyzed to determine genotypic relationship to TPV and DRV resistance and to explore the impact of selected protease gene mutations on susceptibility to TPV and DRV.
Methods: Baseline isolates for 1468 RESIST patients and post-rebound isolates from 50 TPV-treated patients were sequenced and analyzed for Virtual Phenotype predicted IC50 fold change (FC, Version 4.0.0.1). For evaluation of
cross-resistance, the clinical cut-offs of 1.2 and 5.4 for TPV and 3.4 and 96.9 for DRV were used. From the entire TPV project, baseline and on-treatment genotypes were available and were analyzed for 278 patients.
Summary of Results:
48V, 50V, and 54L are associated with resistance to DRV but not to TPV. 82S and 82T are associated with resistance to TPV but not to DRV. 30N was associated with susceptibility to both drugs.
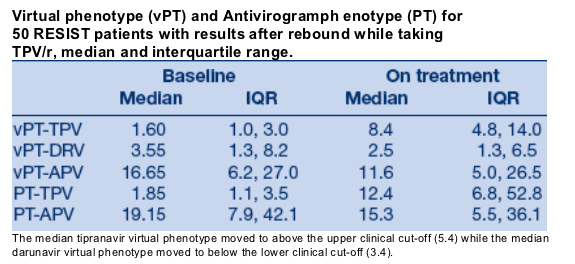
In RESIST patients with baseline and on-treatment results, baseline isolates had median FC for TPV and DRV of 1.6 and 3.55 respectively.
After TPV treatment medians were 8.4 and 2.5 respectively.
For the 80% (n=40) of on-treatment isolates with TPV above the upper clinical cut-off, 55% were below the DRV lower clinical cut-off.
Loss of 48MAV, 50V, 54ML, or 76V and emergence of 34D were associated with declining DRV FC. Selection for 82T/L was not associated with increased DRV FC.
In the larger TPV development program patient group (n=278) the 82T/L mutation was the primary emergent mutation and there was a net loss of mutations at 48, 50, and 76.
Conclusions: PI-associated mutations at 48V, 50V, and 54L are associated with susceptibility to TPV and reduced susceptibility to DRV. These mutations are selected against during TPV therapy, leading to increased susceptibility to DRV. The impact of archived mutations is unknown. Selection for mutations associated with TPV resistance (82L/T) is not associated with development of resistance to DRV.
Methods
Baseline isolates from 1468 RESIST patients were sequenced and analyzed for Virtual Phenotype (Version 4.0.0.1) predicted IC50 fold change (FC). For evaluation of cross-resistance, the clinical cut-offs of 1.2 and 5.4 for TPV and 3.4 and 96.9 for DRV were used.
50 patients enrolled in RESIST and treated with TPV/r were identified on the basis of rebound in plasma HIV RNA and had paired baseline and rebound genotyping performed. Both viral isolates were analyzed for Virtual Phenotype
drug sensitivity to all licensed PIs, in particular TPV and DRV using the proprietary VIRCO genotypic analysis. Emergent mutations were explored.
278 patients treated with TPV/r were identified on the basis of rebound in plasma HIV RNA and had paired baseline and rebound genotyping performed. The paired genotypes were analyzed to determine which mutations were selected for
and which were selected against.
RESULTS Section 1
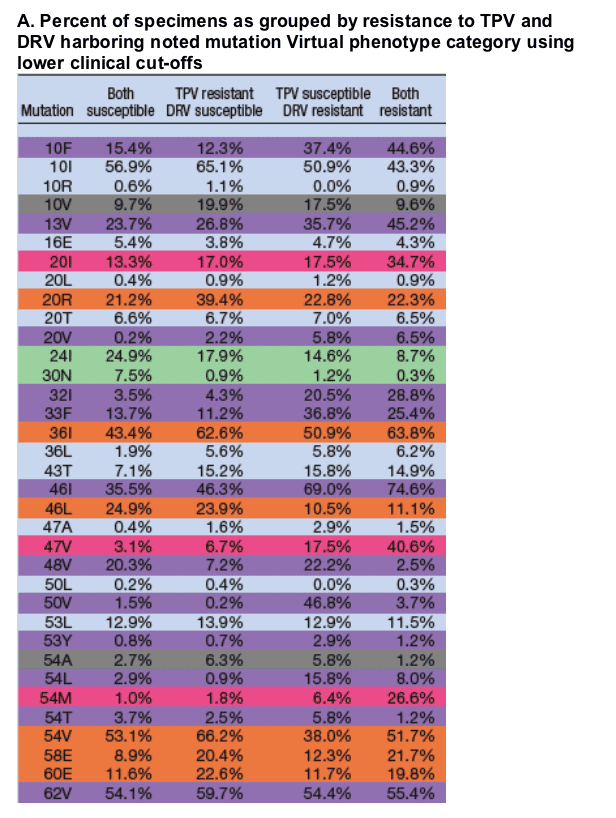
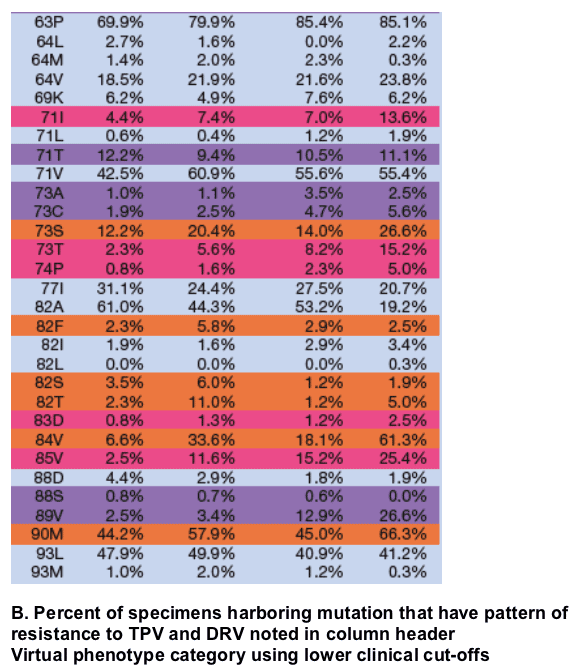
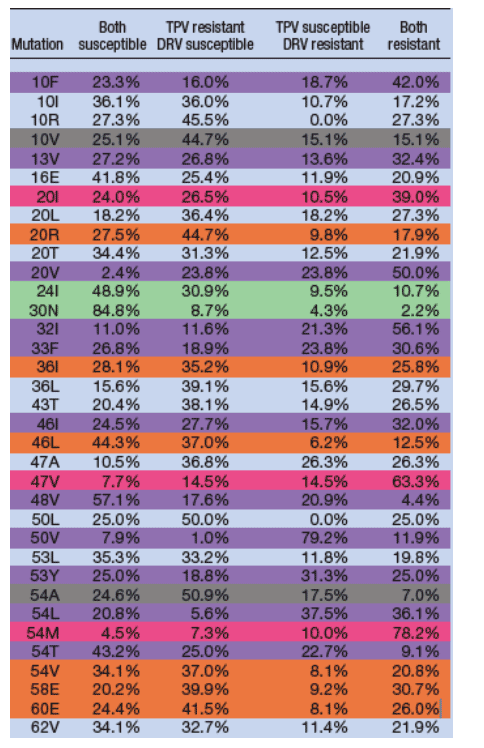
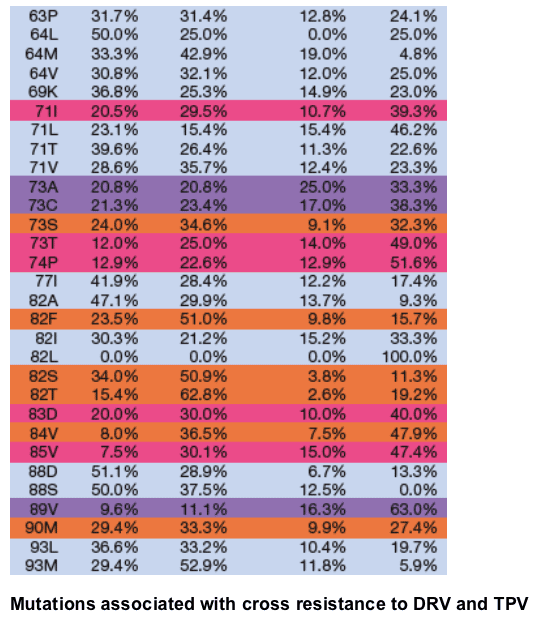
Using the lower clinical cut-offs, genotypes were reviewed to see which protease inhibitor resistance associated mutations were associated with cross resistance and which were associated with resistance to one drug and not the other.
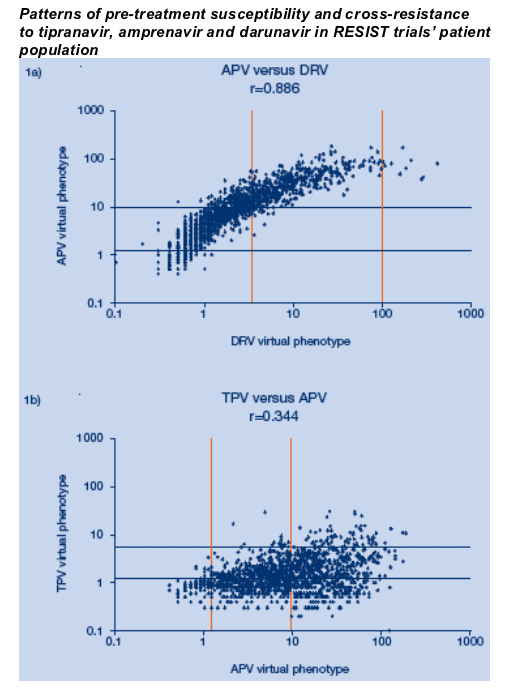
Key to table A: 44.2% for 90M under "Both susceptible" means that 44.2% of specimens below the lower clinical cut-off for both TPV and DRV had a 90M mutation by population sequencing.
Key to table B: 29.4% for 90M under "Both susceptible" means that 29.4% of specimens with a 90M mutation were below the lower clinical cut-off for both TPV and DRV.

Primarily cross resistant: For mutations 54M, 73T, 74P, or 85V nearly half or more than half of specimens were resistant to both TPV and DRV.
Primarily TPV resistant: More than half of specimens with mutations 82F, 82S, or 82T were resistant to TPV and susceptible to DRV.
Primarily DRV resistant: Almost 80% of specimens with 50V mutation were resistant to DRV and susceptible toTPV. More than half of specimens with mutations 20V, 32I, or 89V were resistant to both TPV and DRV.
Primarily cross susceptible: Almost 85% of specimens with the 30N mutations were susceptible to both TPV and DRV.
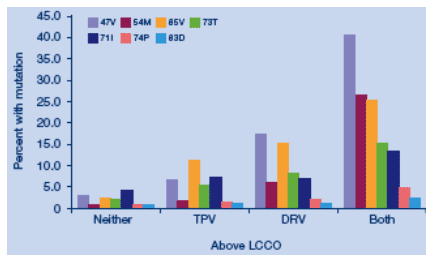
Percent of isolates with mutations in each category of resistance to tipranavir and darunavir for mutations associated with cross resistance
Mutations at 47V, 54M, 85V and 73T are most prevalent in isolates resistant to both protease inhibitors.
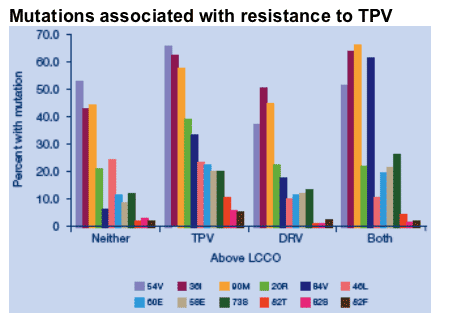
Percent of isolates with mutations in each category of resistance to tipranavir and darunavir for mutations associated with tipranavir resistance and darunavir susceptibility
Mutations 54V, 36I, 90M, 60E, 58E, and 73S are ubiquitous, but most prevalent when tipranavir vPT is above the LCCO, whether darunavir is above or below the LCCO.
Mutations 20R, 82T and 82S are only at increased prevalence in isolates resistant to tipranavir and susceptible to darunavir.
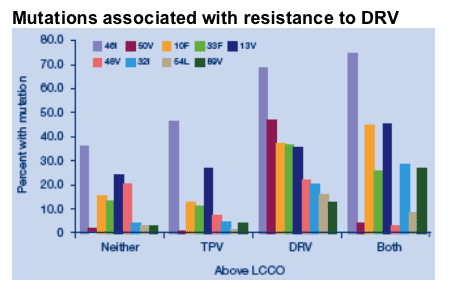
Percent of isolates with mutations in each category of resistance
to tipranavir and darunavir for mutations associated with
darunavir resistance and tipranavir susceptibility
Mutations at 46I, 10F, 13V, 32I, 33F and 89V are prevalent in both cross resistant isolates and darunavir resistant/tipranavir susceptible isolates.
Mutations at 50V and 54L are prevalent only in darunavir resistant/ tipranavir susceptible isolates.
Mutations associated with reduced susceptibility to DRV or TPV,
or associated with susceptibility to both
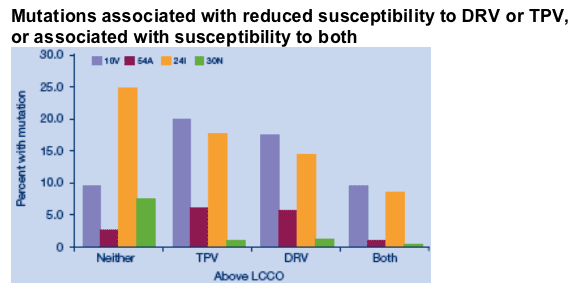
Percent of isolates with mutations in each category of resistance
to tipranavir and darunavir for mutations associated with not being cross resistant
Results section 2
Susceptibility to tipranavir and darunavir in tipranavir treated RESIST patients with baseline and on-treatment genotypic sequences

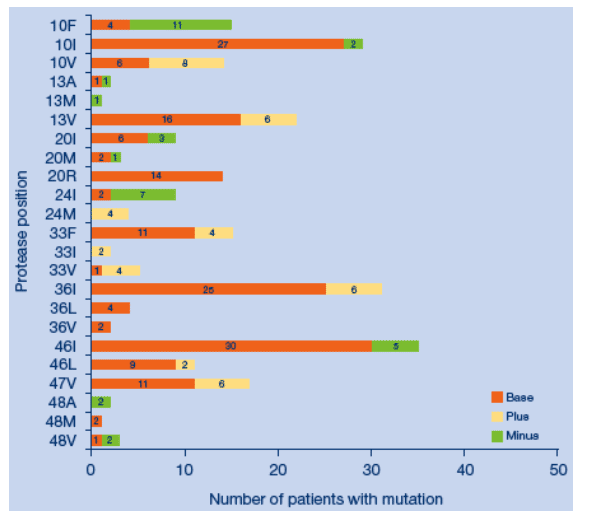
Subtraction histogram of specific amino acids at protease
positions associatedwith drug resistance: 50 tipranavir treated
RESIST patients
The two most common emergent mutations, 82LT and 84V, were not associated with any change in susceptibility to darunavir.
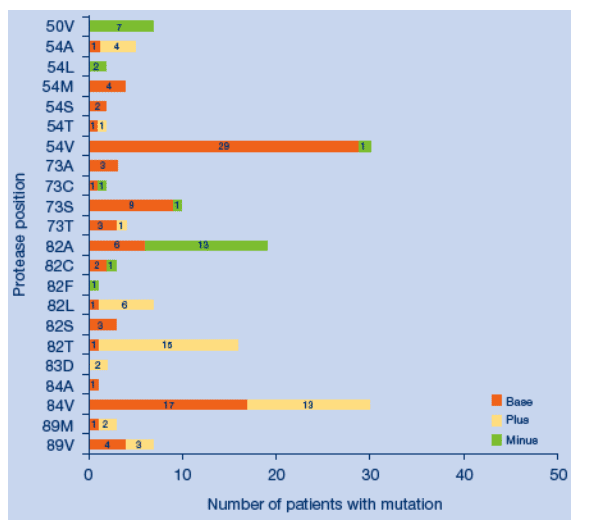
Results section 3
Mutations associated with tipranavir and darunavir resistance in tipranavir treated patients with baseline and on-treatment genotypic sequence from the tipranavir development program (all phase 2-3 trials in which resistance testing was undertaken).
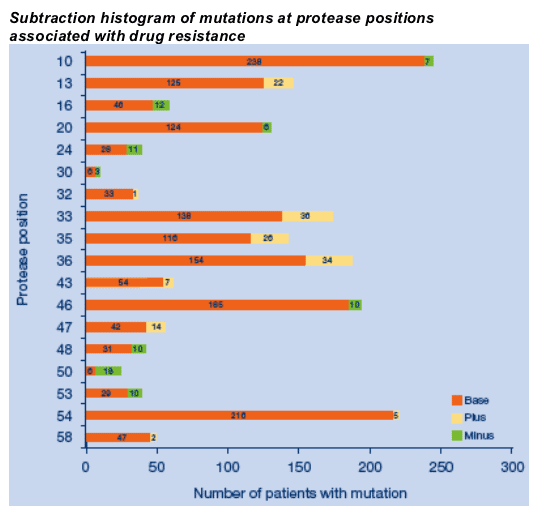
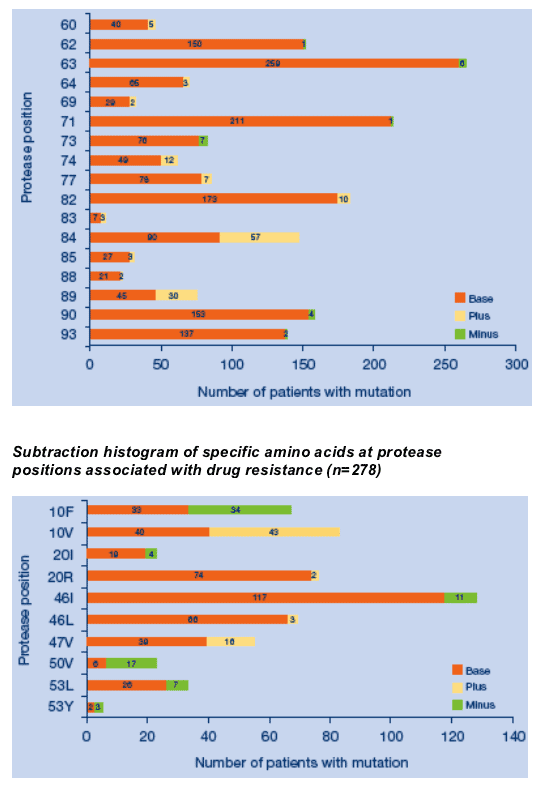
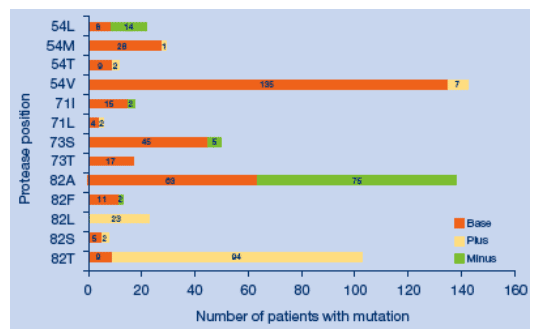
Mutations at 10F, 50V, 54L and 82A were deselected in the majority of those patients whose virus had them at baseline.
Mutations at 10V, 82L and 82T were emergent.
|
| |
|
 |
 |
|
|