 |
 |
 |
| |
Effects of 4 weeks of atazanavir, lopinavir/ritonavir, or placebo on endothelial function and insulin sensitivity in healthy men
|
| |
| |
Reported by Jules Levin
Michael Dube. Indiana University School of Medicine
Indianapolis IN, USA
Effects of 4 weeks of atazanavir, lopinavir/ritonavir, or placebo in healthy men
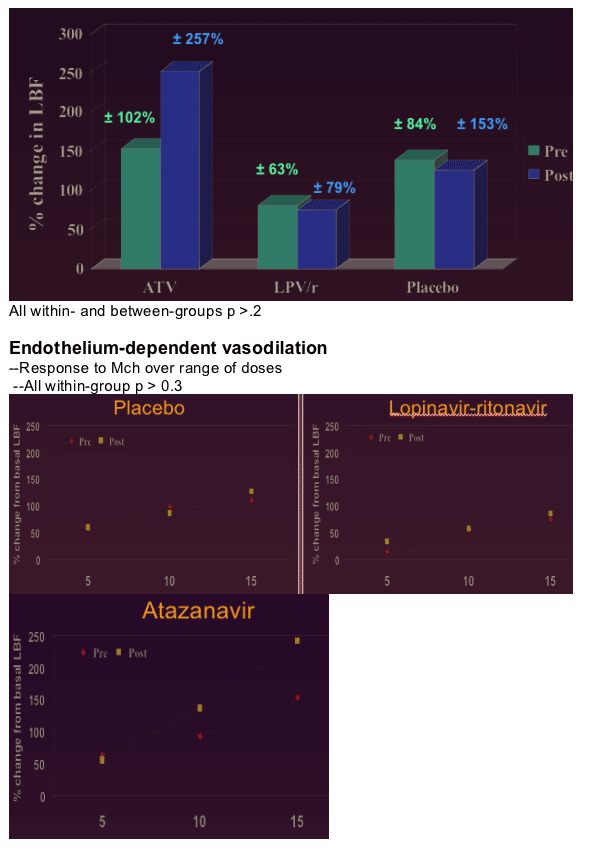
--No change in resting basal leg blood flow
--No change in response to nitroprusside
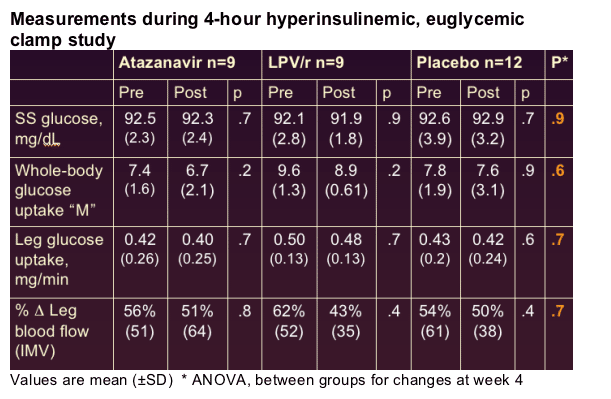
--No change in fasting glucose, adiponectin, resistin, or PAI-1 antigen levels
--Triglycerides increased with LPV/r (77 to 128 mg/dL, p=.002)
--No change in total cholesterol, LDL cholesterol, or non-HDL cholesterol with either PI
--Total bilirubin increased with ATV (0.6 to 1.2 mg/dL, p=.03) and placebo (0.5 to 0.7, p=.004)
AUTHOR CONCLUSIONS
Atazanavir and lopinavir-ritonavir did not impair endothelial function, whole-body glucose uptake, leg glucose uptake, or insulin-mediated vasodilation in lean healthy men
This lack of effects on these parameters with these PIs is strikingly different than with indinavir
Further work is needed with other PIs to determine if adverse effects on endothelial function are unique to indinavir in this drug class
BACKGROUND
Indinavir markedly impairs endothelium-dependent vasodilation by reducing NO release
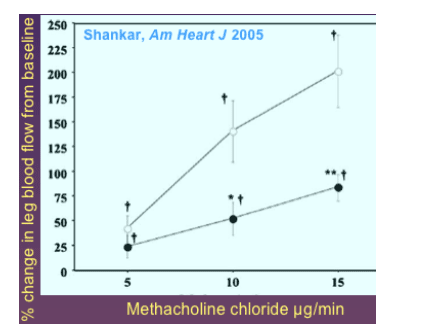
--Healthy male subjects, before and after 4 wk indinavir 800mg tid
--Response to infusion of the eNOS inhibitor L-NMMA abolished
--Marked reduction of insulin-mediated vasodilation
--Minimal effects on whole-body glucose uptake by clamp (other studies w/same design)
Is endothelial dysfunction the link between PI use and CVD?
Objective: Determine the effects of atazanavir and lopinavir-ritonavir on endothelial function and insulin sensitivity in healthy subjects
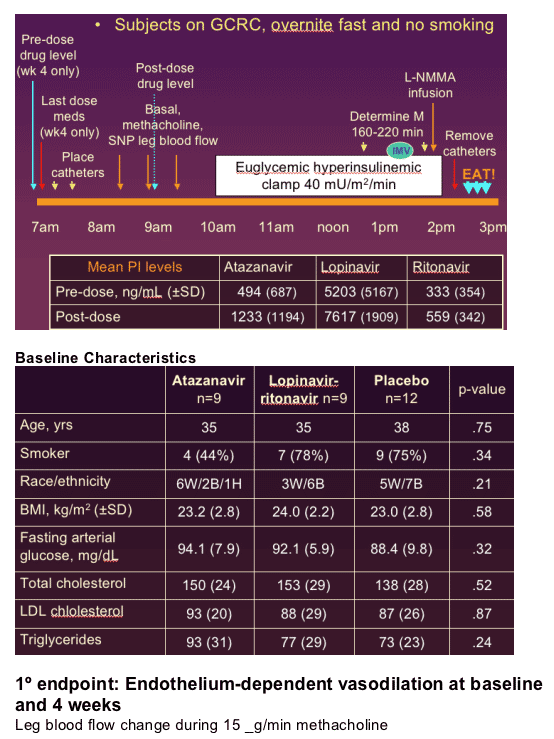
30 healthy men (median age: 36; BMI 23.4 kg/m2; DXA body fat <27%); normal lipids; no medications; no diabetes, IGT, hep B or C. Evaluated leg blood flow, glucose clamps, and metabolic labs.. Randomized to ATV 400 mg/d + LPV/r placebo (n=9), LPV 400/100 bid + ATV placebo (n=9), or Placebo + placebo (n=12).

Methods
--Randomization stratified by BMI and gender
--Femoral arterial and venous catheterization
--Thermodilution measurement of leg blood flow (LBF), accomplished in sets of 10
--Graded doses of methacholine (Mch): 5, 10, & 15 _g/min for 5 min each
--Sodium nitroprusside (SNP): 1.75, 3.5, & 7 _g/min
--Primary endpoint: % D in LBF with 15 _g/min Mch.
--10 subjects/arm 80% power to detect a 1 SD difference. Indinavir decreased the delta in LBF by 145±80%. Power to detect an 80% change within arms.
-- 4-hour hyperinsulinemic clamp: 40 mU/m2/min
--Arterial sampling, goal 90 mg/dL
--Whole-body glucose uptake (M) calculated during 160-220 min of clamp
--Leg glucose uptake calculated using LBF and measured arteriovenous glucose difference
--Insulin-mediated vasodilation (IMV): % change from basal LBF during final hour of clamp
STUDY TIMELINE
|
| |
|
 |
 |
|
|