 |
 |
 |
| |
Tenofovir Disoproxil Fumarate (TDF) versus Adefovir Dipivoxil (ADV) in Asians with HBeAg-Positive and HBeAg-Negative Chronic Hepatitis B Participating in Studies 102 and 103
|
| |
| |
Reported by Jules Levin
AASLD Nov 4 2008, San Francisco, CA
S Lee1, E J Heathcote2, W Sievert3, H Trinh4, K Kaita5, Z Younossi6, J George7, M Shiffman8, P Marcellin9, J Sorbel10,
J Anderson10, E Mondou10, J Quinn10, and F Rousseau10
1University of Calgary, Calgary, AB, Canada; 2Toronto Western Hospital,
University of Toronto, Toronto, ON, Canada; 3Monash University, Melbourne, VIC, Australia; 4San Jose Gastroenterology,
San Jose, CA, USA; 5University of Manitoba Health Science Center, Winnipeg, MB, Canada; 6Inova Fairfax Hospital, Falls Church, VA, USA; 7Westmead Hospital and University of Sydney, Westmead,
NSW, Australia; 8Virginia Commonwealth University Medical Center, Richmond VA, USA; 9Hopital Beaujon, Clichy, France; 10Gilead Sciences, Durham, NC, USA
AUTHOR CONCLUSIONS
TDF demonstrated superior HBV DNA suppression relative to ADV in Asian patients following 48 weeks of randomized treatment
Efficacy, safety and resistance analyses were consistent with the
results of the overall studies following 48 weeks of randomized treatment
INTRODUCTION
Tenofovir DF has shown superior effi cacy to adefovir dipivoxil in treatment-naïve patients with chronic hepatitis B in 2 pivotal studies
-- After 48 weeks of tenofovir DF treatment 76% of HBeAg-positive patients and 93% of HBeAg-negative patients had HBV DNA <400 copies/mL (c/mL)
Asian patients comprised a substantial subset of the participants in these pivotal studies
Evaluation of effi cacy and safety in Asian patients was considered important given the prevalence of HBV infection in this population
OBJECTIVE
To evaluate the efficacy and safety of tenofovir DF among Asian patients with chronic hepatitis B participating in tenofovir DF pivotal studies GS-174-0102 (HBeAg-) and GS-174-0103 (HBeAg+)
METHODS
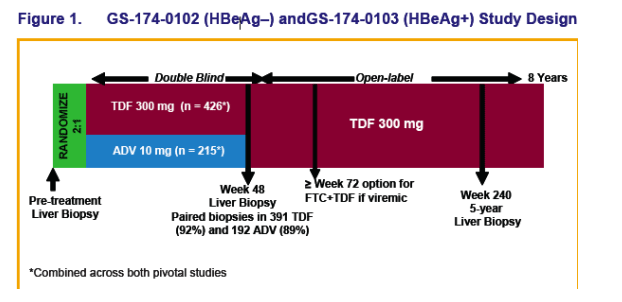
Patients were randomized 2:1 to double-blind tenofovir DF (TDF) 300 mg or adefovir dipivoxil (ADV) 10 mg once daily for 48 weeks in studies GS-174 0102 (HBeAg-) and GS-174-0103 (HBeAg+)
Virologic (HBV DNA < 400 c/mL [69 IU/mL]) and histologic response (≥ 2 point decrease in Knodell necroinfl ammation without worsening fibrosis) were prospectively evaluated
HBV DNA and safety laboratory parameters were performed every 4 weeks through Week 48 with annual resistance surveillance
Asian ethnicity was determined by self-report as recorded on the case report form
Eligibility criteria required elevated ALT*, Knodell necroinfl amatory score ≥ 3, and viremia with HBV DNA > 105 copies/mL with the Roche COBAS TaqMan assay (LLOQ=169 copies/mL [29 IU/mL])
(*Upper normal limit [ULN] 34 U/L for women; 43 U/L for men)
Figure 2. Asian Patients Participating in Pivotal Studies
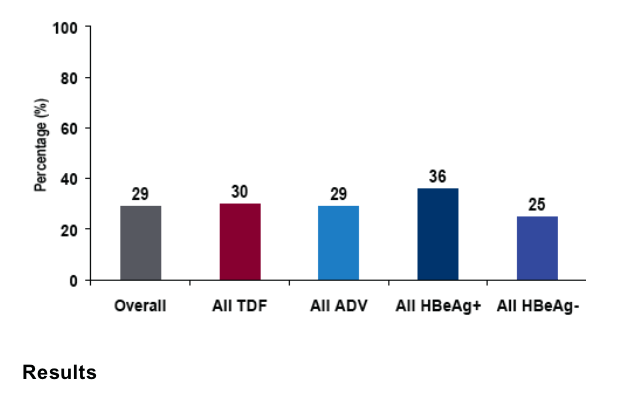
189 Asians were enrolled across the 2 studies
Asians comprised about 30% of all patients
-- 127/426 (30%) on TDF
-- 62/215 (29%) on ADV
Combined study results are presented to maximize sample size
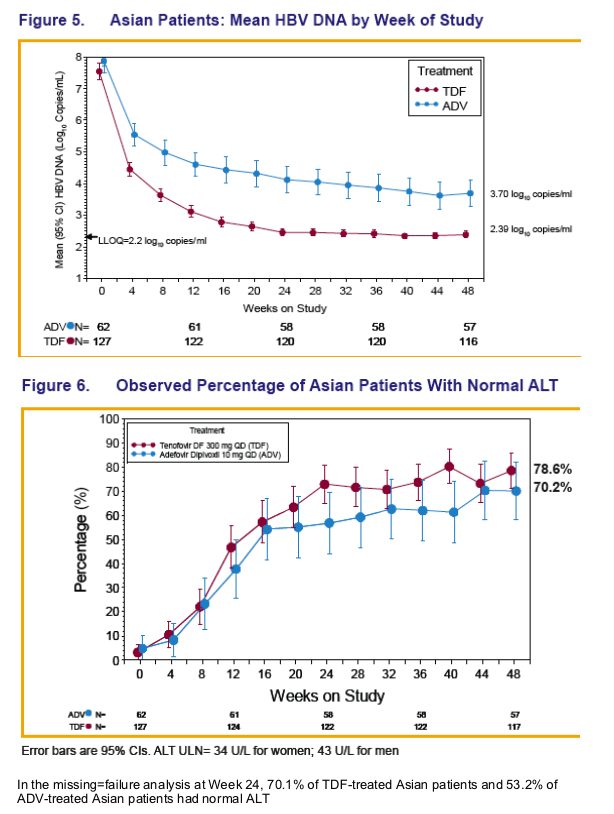
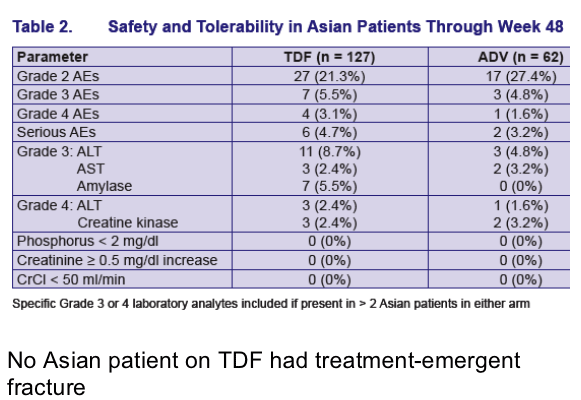
TDF Resistance Surveillance:
No HBV polymerase/reverse transcriptase amino acid substitutions associated with resistance to tenofovir were detected at Week 48 in any patient (see Poster 977 for details to Week 96)
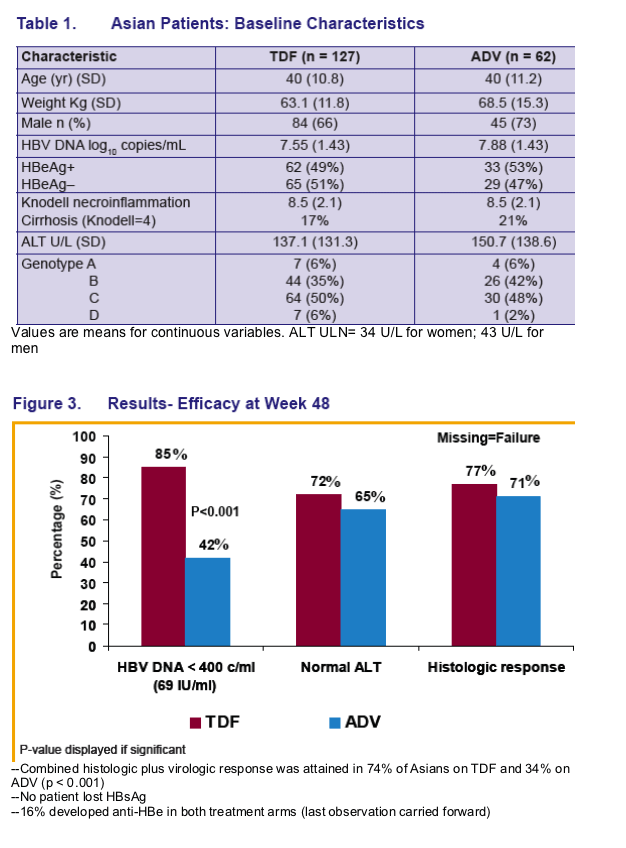
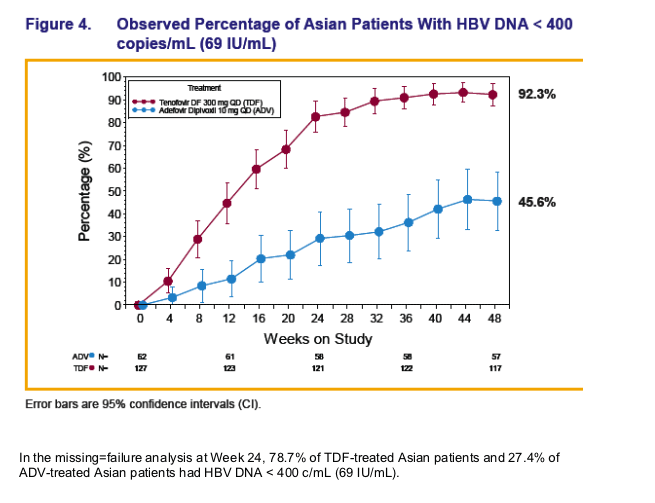
Across both pivotal studies 10 Asian patients had HBV DNA ≥ 400 copies/mL (≥ 69 IU/mL) at Week 48, which included 4 Asians with a virologic breakthrough* 3 of whom were non-adherent
|
| |
|
 |
 |
|
|