 |
 |
 |
| |
Virological and biochemical response in patients with HBeAg-negative chronic hepatitis B treated with PEGASYS with or without lamivudine: results of 4-year follow-up: 11% HBsAg Clearance
|
| |
| |
Reported by Jules Levin
43rd EASL
April 23-27, 2008
Milan Italy
Patrick Marcellin*, Teerha Piratvisuth, Maurizia Brunetto, Ferruccio Bonino, George K.K. Lau, Patrizia Farci,
Cihan Yurdaydin, Jian Wu, Matei Popescu
*Service d'Hepatologie, Hopital Beaujon, Clichy, France
AUTHOR SUMMARY
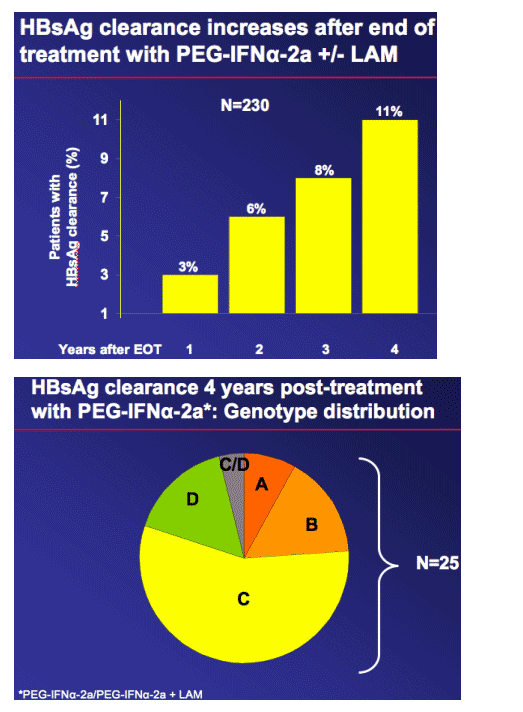
48 weeks' therapy with PEG-IFN_-2a provides continuing benefits up to 4 years after end of treatment in patients with HBeAg-negative disease
4 years after end of treatment:
--PEG-IFN_-2a continued to maintain HBV DNA suppression and normal ALT levels
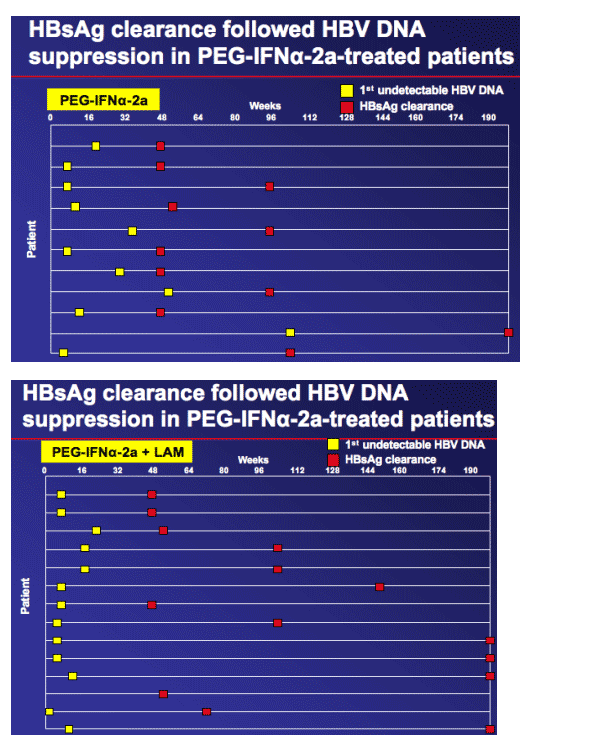
-- 11% of PEG-IFN_-2a-treated patients had cleared HBsAg.
HBsAg clearance is known to be associated with favourable clinical outcome and considered closest endpoint to a 'cure'
AUTHOR CONCUSION
PEG-IFN_-2a should be considered as first-line therapy in patients with HBeAg-negative CHB because of its:
-- Ability to achieve sustained response following a short course of treatment in a substantial proportion of patients
-- This avoids the need for long-term maintenance therapy and the associated risks of developing drug resistance in those patients who respond
Benefits of HBsAg clearance
Spontaneous HBsAg clearance may occur but at a low annual incidence:
-- 0.1Å0.8% in high endemic areas1,2
-- 1.0Å2.1% in the low endemic areas3-6
Associated with improved long-term outcome7
-- Reduced incidence of hepatic complications and HCC
Closest outcome to a 'cure'
1. Wu et al. J Hepatol 1987; 2. Liaw et al. Hepatology 1991; 3. Sampliner et al. Am J Med Sci 1979;
4. Feinman et al. Hepato-Gastro 1982; 5. McMahon et al. Ann Intern Med 2001;
6. de Franchis et al. Ann Intern Med 1993; 7. Chen et al. Gastroenterology 2002
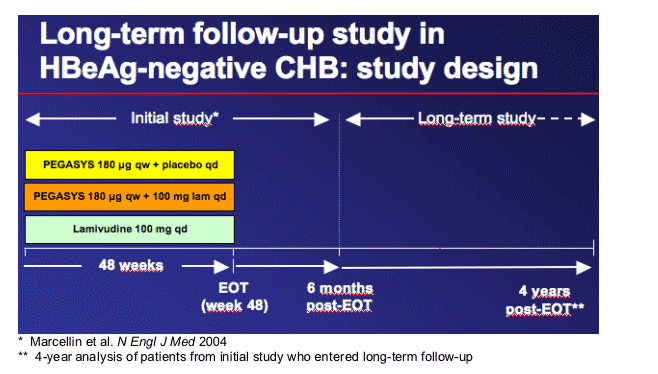
Long-term follow-up study in patients with HBeAg-negative CHB
Results of the initial part of this large international randomised study1 have documented:
-- Superior efficacy 6 months post-treatment (ALT normalisation, HBV DNA suppression) of PEG-IFN_-2a vs LAM
-- No benefit of combining PEG-IFN_-2a and LAM
All study centres were offered participation in a long-term (LT) observational follow-up study
-- Interim analyses were planned a priori for each of the yearly follow-up visits, ie at 1-yearly intervals after treatment in the initial study
4-year post-treatment data are presented here
1. Marcellin et al. N Engl J Med 2004
Methodological considerations
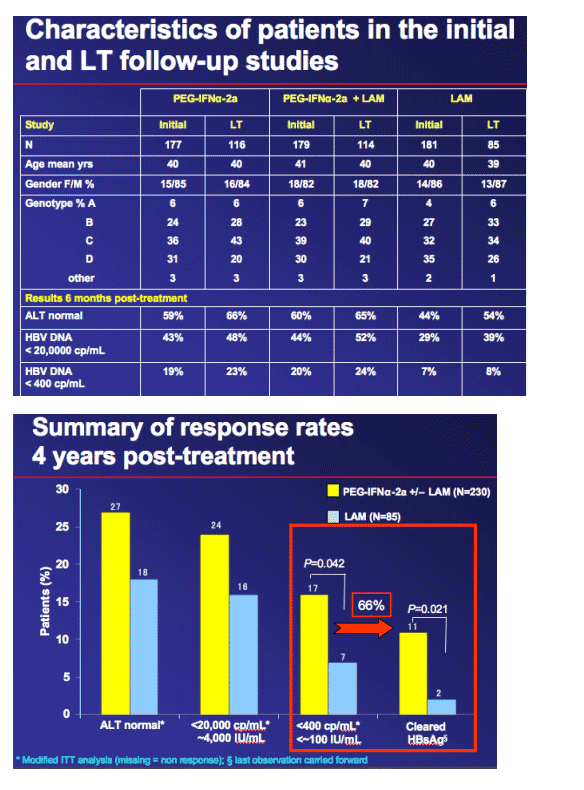
Of the 54 centres involved in the initial study,
42 participated in the LT study
-- Contributed a total of 315 (59%) of the original 537 patients
-- Significantly more patients who received PEG-IFN_-2a +/- LAM (66% and 64%, respectively) than LAM alone (47%) entered LT study (P <0.01)
-- A total of 230/356 PEG-IFN_-2a-treated and 85/181 LAM-treated patients entered the study
Participants in the LT study were more likely to have a response at the end of the initial study, especially in the LAM monotherapy arm
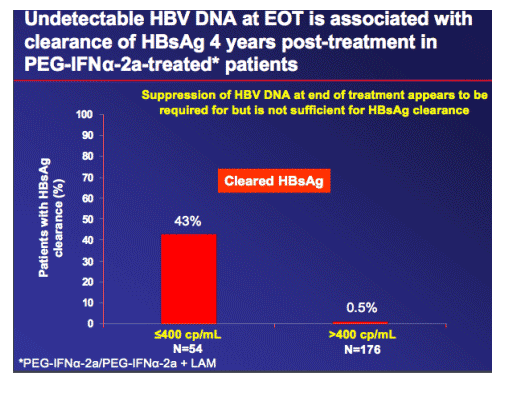
|
| |
|
 |
 |
|
|