| |
Chronic Hepatitis B: Preventing, Detecting, and Managing Viral Resistance
|
| |
| |
Gastroenterology and Hepatology March 2008
Emmet B. Keeffe_, Douglas T. Dieterich, Jean-Michel Pawlotsky, Yves Benhamou
_ Division of Gastroenterology and Hepatology, Department of Medicine, Stanford University School of Medicine, Stanford, California
Department of Medicine, The Mount Sinai Medical Center, New York, New York
French National Reference Center for Viral Hepatitis B, C and Delta and Virology Laboratory, Hopital Henri Mondor, Universite Paris 12, Creteil, France
INSERM U841, Creteil, France
Service d'Hepatologie Hopital Pitie-Salpetriere, Universite Paris 5, Paris, France
Licensed oral agents for antiviral therapy in patients with chronic hepatitis B virus (HBV) infection include lamivudine, adefovir, entecavir, and telbivudine. Emtricitabine, tenofovir, and the combination of tenofovir plus emtricitabine in 1 tablet, which are licensed for the treatment of human immunodeficiency virus infection, are additional off-label options for treating HBV infection. Preventing HBV antiviral drug resistance to nucleoside/nucleotide analogues and appropriate management when resistance occurs has become a major focus in the management of chronic hepatitis B. HBV antiviral drug resistance may be best prevented by using an agent or combination of agents with a high genetic barrier to resistance, and 2 potent nucleoside and nucleotide drugs with different resistance profiles may prove to be the optimal first-line treatment for chronic hepatitis B. Frequent assessment of quantitative serum HBV DNA remains the best approach to early detection of resistance, and antiviral therapy should be modified as soon as resistance is detected. Results from several clinical trials have shown that the addition or substitution of newer antiviral agents can restore suppression of viral replication, normalize alanine aminotransferase levels, and reverse histologic progression in patients with resistance to lamivudine, but little information exists regarding the long-term benefits of second-line treatment regimens. Despite the substantial advances in treatment made to date, new agents with novel viral targets will be needed for patients who ultimately may fail second- or third-line therapy.
It is estimated that 1.25 million people in the United States have chronic hepatitis B, and there were 60,000 new hepatitis B virus (HBV) infections in 2004.1 Acute HBV infection may be asymptomatic, result in self-limited or fulminant hepatitis, or progress to chronic hepatitis, which can lead to cirrhosis or hepatocellular carcinoma.2 Cirrhosis and hepatocellular carcinoma result in about 1 million deaths worldwide each year.3 In the United States, there has been a substantial decline in the incidence of acute hepatitis B over the past 15 to 20 years that has coincided with the national strategy to eliminate HBV transmission by vaccination and public health measures.4 Nevertheless, because of a high HBV prevalence rate in many developing countries and population migrations to developed countries, chronic HBV infection is an important health problem in the United States and Europe.
Antiviral therapy in patients with chronic hepatitis B is associated with improved outcomes.5, 6 Over the past 10 years, oral antiviral agents available for patients with chronic HBV infection have included lamivudine, adefovir, entecavir, and telbivudine, which are licensed to treat chronic hepatitis B, and emtricitabine, tenofovir, and the combination of tenofovir plus emtricitabine, which has been licensed for treatment of patients with human immunodeficiency virus (HIV) infection, but also is active against HBV infection.7 However, 20 years of experience with the treatment of HIV, as well as current HBV resistance data, point to the emergence of HBV antiviral drug resistance,8 as illustrated by the case of lamivudine, the nucleoside that has the longest history of use in patients with chronic HBV infection and the highest rate of resistance. The development of resistance is associated with a poorer long-term prognosis.9, 10 Clinicians need to be aware of current approaches to resistance testing and of effective treatment strategies to minimize the emergence of resistant HBV strains, and they should be cognizant of regimens that may be effective after resistance has emerged.
Viral Replication and Selection of Resistant Strains
The genome of HBV consists of partially double-stranded, 3.2-kb, covalently closed, circular DNA (ccc DNA) comprising 4 overlapping open reading frames.11 The viral genome is transcribed into 4 major subgenomic viral messenger RNAs, under the control of specific enhancers,12 and is the template for the pregenomic RNA. The virus is encapsidated after binding of the polymerase and core to the pregenomic RNA in the cytoplasm. Nucleocapsids are enveloped by budding into the endoplasmic reticulum, after which they are secreted from the cell or return to the nucleus to amplify the cccDNA reservoir.13
Viral mutations occur spontaneously during HBV replication. Viral reverse transcriptases intrinsically are error prone and lack a proofreading function, allowing for replication errors to occur. These replication errors result in the emergence of multiple HBV variant quasispecies that coexist and reach population densities in direct proportion to their relative replication fitness.11 This phenomenon is responsible for the generation of significant diversity; it has been shown that HBV genomes in one given patient displayed a rate of 1.4 to 3.2 X 10-5 nucleoside substitutions per year, a value approximately 104 times greater than DNA genomes and about 10-2 less than that for HIV.14 A chronic HBV carrier can produce up to 1013 virions per day, and, as a result, every nucleotide of the 3.2-kb HBV genome theoretically can be substituted within one patient every day.2
The dominant quasispecies is by definition the best adapted to its host environment, and, as expected, random mutations generally will impair its fitness to a degree. Thus, the emergence of successful HBV variants resistant to an antiviral drug is affected by the mutation rate and viral load and is determined eventually by the replication fitness of the mutant virus in relation to the antiviral potency of the drug and the number of mutations required to confer resistance (ie, the genetic barrier to resistance).15
Mutations Associated With Resistance to Nucleoside/Nucleotide Analogues
Defining the Consequences of Resistance
Treatment failure can be defined as primary or secondary. Primary treatment failure is the failure of a drug to reduce HBV DNA levels by 1 X log10 IU/mL or greater within 3 months of initiation of therapy, and secondary failure is defined as a rebound of HBV replication by 1 X log10 IU/mL or greater from nadir in patients in whom treatment initially produced a decrease in serum HBV DNA of 1 X log10 IU/mL or greater.16
Secondary treatment failure due to resistance to antiviral therapy can result in a decreased rate of hepatitis B e antigen (HBeAg) seroconversion17; reversion of virologic, biochemical, and histologic improvement18, 19; increased rate of disease progression5; severe exacerbations in the presence of cirrhosis20, 21; and risk for graft loss and death after liver transplantation.22
Mutations in the Polymerase Gene and Antiviral Resistance
Mutations resulting in resistance to nucleoside/nucleotide analogues mainly involve the viral polymerase gene (Figure 1).11, 23, 24, 25 This gene contains 7 functional domains (A-G), and mutations that give rise to nucleoside/nucleotide resistance are located essentially in domains A through E.23
Figure 1. Location of the major antiviral drug-resistant mutations associated with LMV, LdT, ADV, TDF, and ETV11 (Reprinted with permission). LMV, lamivudine; Ldt, telbuvidine; ADV, adefovir dipivoxil; TDF, tenofovir DF; ETV, entecavir. POL/RT, polymerase reverse-transcriptase.
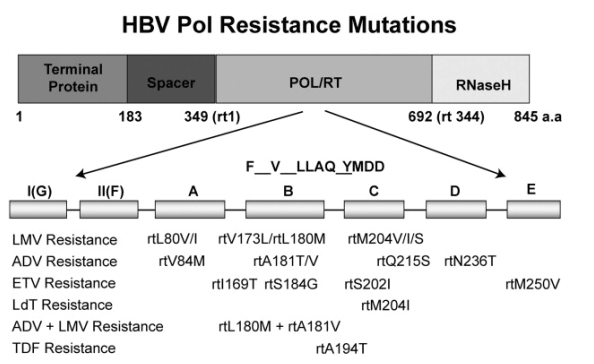
Lamivudine Resistance
Lamivudine inhibits viral reverse-transcriptase activity as a competitive inhibitor of deoxycytidine triphosphate.23 High-level lamivudine resistance results from M204V and M204I mutations in the C domain.26 HBV variants with these mutations display reduced reverse-transcriptase activity and replication capacity, but compensatory mutations in the B domain (V173L and L180M) restore the replication capacity of virus with the M204V or M204I mutation.27, 28
Results from 998 patients with HBeAg-positive compensated chronic hepatitis B who took lamivudine for up to 6 years indicated that the proportion of patients with documented lamivudine-resistant mutations increased from 23% in year 1 to 43%, 55%, 71%, and 65% in years 2 through 5, respectively. Patients with lamivudine-resistant mutations who were followed up for more than 4 years had significantly higher risk for hepatic decompensation and liver disease-related severe adverse events.9
Adefovir Resistance
Adefovir inhibits priming of reverse transcription by preventing the incorporation of deoxyadenosine triphosphate into the viral primer and inhibits viral-minus strand DNA elongation.29 Two mutations have been described that confer approximately 5- to 10-fold reduced susceptibility to adefovir in vitro: N236T in the D domain of viral polymerase and A181V in the B domain.23 Most recently, another rare mutation, I233V, has been suggested to result in primary resistance to adefovir, but that mutation has not been confirmed to be associated with a reduced susceptibility to adefovir in vitro.30 A study of adefovir in 125 patients who were followed up for up to 240 weeks indicated that the cumulative probabilities of mutations associated with a virologic breakthrough at 48, 96, 144, 192, and 240 weeks were 0%, 3%, 11%, 18%, and 29%, respectively. The respective values for mutations associated with virologic and biochemical breakthrough (alanine aminotransferase [ALT] increases) were 0%, 2%, 6%, 10%, and 11%.31, 32
Entecavir Resistance
Entecavir is a deoxyguanosine analogue that is greater than 100-fold more potent against HBV in culture than either lamivudine or adefovir, and halts HBV DNA elongation after incorporation of a few bases more than lamivudine or adefovir.33 Analysis of results from 673 patients treated with entecavir indicated that 3% showed virologic rebound by 96 weeks of treatment. Three entecavir rebounds were attributable to lamivudine-resistant virus present at baseline, and none of the others was associated with either entecavir genotypic resistance or loss of entecavir susceptibility.34 After administration of entecavir to patients who were refractory to lamivudine, entecavir resistance was detected in 1% of patients after 48 weeks and an additional 9% after 96 weeks. Entecavir resistance was associated with lamivudine-resistance-conferring mutations plus additional changes at T184, S202, and/or M250.35 Recent results have indicated that the cumulative probability of virologic breakthrough associated with entecavir resistance in nucleoside-naive patients was 0.8% over 4 years, and that for patients who received prior lamivudine therapy it was 39.5%, with 1% having resistant virus after 1 year, 10% after 2 years, 16% after 3 years, and 15% after 4 years of treatment.36
Telbivudine Resistance
Telbivudine targets the synthesis of positive-strand HBV DNA, which is thought to result in slower emergence of resistance than lamivudine, which targets negative-strand synthesis.37 Results from the GLOBE trial indicated that the 2-year cumulative rate of virologic breakthrough for HBeAg-positive patients was 21.6%, and for HBeAg-negative patients was 8.6%.38
Emtricitabine Resistance
Emtricitabine inhibits HBV polymerase in essentially the same manner as lamivudine, and the M204V mutation that confers resistance to lamivudine also results in resistance to emtricitabine.39, 40 Emtricitabine is effective as monotherapy in patients with chronic HBV infection, reducing HBV DNA levels to less than 400 copies/mL in 54% of patients over 48 weeks.39 However, at 48 weeks, 9% to 16% of patients treated with 25 to 200 mg/day of emtricitabine developed resistance-conferring mutations (M204I or M204V with or without L180M or V173L).41
Tenofovir Resistance
Tenofovir is an acyclic nucleotide analogue that retains significant activity against HBV variants with mutations (M204V plus L180M), conferring resistance to lamivudine and N236T that results in resistance to adefovir.42, 43 Tenofovir retains good activity in patients with lamivudine-resistant HBV variants, either as monotherapy or when added to continuing lamivudine treatment.44, 45 HBV mutations that confer resistance to tenofovir have not yet been identified.
Detecting, Preventing, and Managing Hepatitis B Virus Resistance
Clinical evidence, as summarized earlier, indicates that resistance to antiviral monotherapy: (1) is common, particularly in patients treated with lamivudine; (2) increases with duration of treatment; and (3) is associated with poorer clinical outcomes. Many new antiviral agents, however, are available for initial therapy and as second-line treatment for those failing lamivudine. It is important that patients with lamivudine resistance be treated with agents that are not cross-resistant, such as the nucleotide analogues adefovir or tenofovir.
The availability of genetic testing for identification of viral genotypes likely to show phenotypic resistance and the fact that different antiviral agents do not have completely overlapping resistance profiles raises 3 questions: When should genotypic resistance testing be performed in patients with HBV infection? What approach(es) to initial treatment are least likely to result in the rapid emergence of resistance? What treatment alternatives are most useful in patients who have developed resistance-conferring mutations?
Resistance Testing
Genotypic resistance precedes phenotypic resistance with virologic breakthrough, which in turn precedes biochemical and/or clinical breakthrough. Phenotypic resistance is defined as virologic breakthrough (ie, an increase in serum HBV DNA by >1 log10 above nadir after achieving virologic response during continued treatment), or virologic rebound (ie, increase in serum HBV DNA to >20,000 IU/mL or to above pretreatment level after achieving virologic response during continued treatment). Biochemical relapse is characterized as an increase of ALT to above the upper limit of normal after achieving normalization with treatment and may be associated with a hepatitis flare (Figure 2).46, 47
Development of resistance can be detected readily by monitoring serum HBV DNA levels and is indicated by a greater than 1 log10 increase from the patient's lowest level confirmed by measurement on 2 assays. Patients taking lamivudine should have HBV DNA levels evaluated with a sensitive assay every 3 to 6 months and those being treated with adefovir or entecavir should be monitored every 6 months after the first year of treatment. Patients with advanced liver disease should be monitored every 3 months.47 Guidelines published by the American Association for the Study of Liver Diseases (AASLD) recommend serum HBV DNA monitoring every 3 to 6 months for patients receiving therapy for chronic hepatitis B.46 It also has been noted that monitoring the emergence of resistance-conferring mutations may be the most sensitive way to assess patients who remain viremic on current treatments.48
Assays for Detection of Resistance-Conferring Mutations
The reference method for detection of resistance-conferring mutations is population-based sequencing (ie, a direct sequence analysis of the HBV polymerase gene). Line-probe assays using probes for individual mutations are specific and reproducible and can detect a mutant representing as little as 5% of the viral population. However, this approach is limited in that it can detect only known mutations. It requires periodic updating with new probes specific to novel mutations because HBV strains resistant to newly developed antiviral drugs are isolated and characterized.49, 50 Other powerful technologies are in development, including high-throughput systems capable of detecting polymorphisms in the entire HBV genome using gene chip technology.51 This method is limited by the fact that it, too, can detect only known polymorphisms and needs to be updated as resistance-conferring mutations are identified.50
When to test
Clinicians might consider testing at the following times: (1) before selection of initial therapy, (2) routinely during the course of therapy, and (3) at the time of virologic breakthrough, before changing the therapeutic regimen. Evaluation of viral genetic mutations has been used extensively in patients with HIV infection, and it is useful to review current recommendations for HIV genotypic resistance testing.
Testing before initiation of therapy
Current guidelines recommend HIV drug resistance testing before initiation of therapy.52 This recommendation is justified by repeated observations of transmission of viral strains resistant to one or more antiretroviral drugs53; however, it is much less clear whether resistance testing is necessary before the initiation of nucleoside/nucleotide antiviral therapy in patients with HBV infection. At this time, it is therefore reasonable to suggest that resistance testing before the initiation of nucleoside/nucleotide treatment is not warranted in patients with HBV infection unless they acquired HBV from an infected patient undergoing antiviral treatment.
Testing during treatment
Although monitoring the emergence of resistance-conferring mutations may be the most sensitive way to monitor patients taking nucleosides/nucleotides for the treatment of HBV infection,48 this approach does not seem warranted on the basis of current information. Viral rebound occurs well before a biochemical breakthrough or hepatitis flare,46 and monitoring of serum HBV DNA with a sensitive assay as recommended should permit clinicians to detect changes in viral load that signal loss of efficacy before increases in ALT levels or clinical deterioration. Notably, guidelines for management of HIV-infected patients make no recommendations for routine genotypic or phenotypic resistance testing in patients who maintain viral suppression during therapy,52 and the same can be said for HBV infection.
Resistance testing at virologic breakthrough
HIV treatment guidelines recommend resistance testing before changing treatment regimens after virologic failure,52 and recommendations for the management of patients with HBV infection also recognize the importance of resistance testing when HBV DNA testing indicates a 1 log10 or greater increase in viral load. Resistance testing should be performed, if possible, before discontinuation of the failing regimen so that the results reflect the latest detectable viral mutations. Results of resistance testing then can be used to guide selection of new treatment.46
Management of Patients With Resistant Virus
Both the AASLD guidelines and those published by Keeffe et al47 provide recommendations for changing therapy in patients with HBV antiviral drug resistance to lamivudine, adefovir, or entecavir. The AASLD guidelines also provide recommendations for new therapy in patients with resistance to telbivudine (Table 1).46, 47
Table 1. Treatment Recommendations of the AASLD and US Algorithm for Patients With Antiviral-Resistant HBV46, 47

ADV, adefovir dipivoxil; ETV, entecavir; FTC, emtricitabine; LdT, telbivudine; LMV, lamivudine; TDF, tenofovir DF
Clinical trials performed to date have focused on second-line therapy for patients who have developed resistance to lamivudine and have shown that adding on or switching to adefovir, or switching to entecavir, can resuppress viral replication, normalize ALT levels, and provide significant histologic improvement.54, 55, 56, 57 Tenofovir also has been shown to be effective in patients with HBV resistance to both lamivudine and adefovir.44 All data indicate that entecavir monotherapy should not be recommended in lamivudine-resistant patients because of the high risk of entecavir resistance and adding a second drug is always preferable to a switch.
Rapti et al58 recently showed that the addition of adefovir to lamivudine in HBeAg-negative patients with lamivudine resistance effectively suppressed serum HBV DNA in the majority of patients without the development of adefovir resistance. In this study of patients with lamivudine resistance, 14 patients were switched to adefovir, and 28 patients had adefovir added to ongoing lamivudine therapy. Serum HBV DNA levels became undetectable and ALT levels normalized in 71% and 90% of patients, with no differences between the 2 groups. However, virologic and biochemical breakthrough occurred in 3 of 14 patients (21%) who were switched to adefovir monotherapy 15 to 18 months from the start of therapy, whereas suppression of HBV replication was persistent for up to 3 years in the group receiving combination adefovir plus lamivudine.
Timing of the change in treatment also is important for patients who experience viral breakthrough while receiving nucleotide/nucleoside therapy. A prompt switch to new treatment results in more rapid viral suppression than does a delayed change in therapy. A study of HBeAg-negative patients with chronic hepatitis B and genotypic and phenotypic resistance to lamivudine indicated that adding adefovir when genotypic resistance was detected (3-6 log10 copies/mL of HBV DNA and normal ALT levels) was more effective than adding adefovir when phenotypic resistance was detected (>6 log10 copies/mL of HBV DNA and high ALT levels).59 Three months after the change in treatment, serum HBV DNA was undetectable in all patients who had adefovir added when genotypic resistance was detected, versus 46% of those who received adefovir when phenotypic resistance was observed. The 2-year rates of virologic response were 100% and 78%, respectively, and ALT levels remained normal in all patients who were given early addition of adefovir. In contrast, ALT levels were normalized in 50% of patients at 6 months, 72% at 12 months, and 93% at 24 months in the group with late addition of adefovir.59
Avoiding Resistance Development: Selection of Initial Therapy
Current treatment of HIV infection is based on creating a high genetic barrier to resistance. This refers to the number of critical drug-resistant mutations required for the virus to overcome the anti-HIV activity of the drug regimen.60 Current guidelines for initial treatment of patients with HIV infection reflect that view by recommending a combination of 3 or more agents from different classes having different mechanisms of action.52 Single-drug class monotherapy or dual therapy with nucleoside reverse-transcriptase inhibitors for the treatment of HIV has been shown to accelerate the development of resistance, cross-resistance, and virologic failure, and also limit future treatment options.
Although data are not available for all antiviral agents used to treat HBV infection, it is becoming clear that single-drug therapy may not provide a sufficiently high barrier to resistance. That is certainly the case for lamivudine9 and it also may be true for other agents. This has prompted consideration of 2-drug combinations as initial treatment in patients with chronic hepatitis B. Potential exceptions to the requirement of a 2-drug regimen to reduce or prevent resistance are entecavir and tenofovir, with the cumulative experience suggesting that HBV antiviral drug resistance occurs at very low rates of less than 1% over 4 years of treatment, in the case of entecavir.36
Several small-scale studies have assessed the efficacy of combination therapy in treatment-naive patients with HBV infection. Results from a 1-year comparison of 104 treatment-naive HBeAg-positive patients treated with telbivudine or lamivudine monotherapy or telbivudine plus lamivudine showed that telbivudine alone and in combination therapy was superior to lamivudine alone in reducing HBV DNA levels and achieving ALT normalization, and that there were no significant differences between results obtained with telbivudine versus telbivudine plus lamivudine.61 Moreover, addition of telbivudine to lamivudine, as might be expected, did not decrease the occurrence of viral breakthrough (15.8% for lamivudine alone vs 12.2% for the combination treatment) or the emergence of resistance-associated mutations.61
The concept that combination therapy should use drugs that are not cross-resistant is exemplified by a study of chronic hepatitis B patients treated with lamivudine alone or combined with adefovir for 1 year.62 Initially, both treatments resulted in equally effective viral suppression, with 4 to 5 log10 HBV DNA reductions by week 16. However, by week 52, the combination therapy group remained virally suppressed, with a median reduction from baseline of 5.4 log10, whereas HBV DNA levels increased in the lamivudine monotherapy group. That increase largely was owing to the development of lamivudine resistance-conferring mutations, which occurred in 20 of the patients who received lamivudine alone versus 2% of those treated with lamivudine plus adefovir.62 Another study of 30 treatment-naive, HBeAg-positive patients showed that 48 weeks of treatment with adefovir plus emtricitabine resulted in greater antiviral activity than adefovir monotherapy; median HBV DNA reductions at 48 weeks were -3.48 log10 versus -2.22 log10, respectively.63 No information was provided regarding the emergence of viral resistance.
The combination of lamivudine with an immune modulator, peginterferon alfa-2a, also has been shown to slow the emergence of lamivudine resistance in a 72-week study (48 weeks of active treatment and 24 weeks of additional follow-up evaluation) in which 537 treatment-naive HBeAg-negative patients with chronic hepatitis B were treated with peginterferon alfa-2a alone, lamivudine alone, or a combination of the two. At 48 weeks, mutations conferring resistance to lamivudine developed in 18% of patients treated with lamivudine alone versus 1% of those who received combination therapy.64 Results from longer-term follow-up evaluation of 36 HBeAg-negative, anti-HBe-positive, treatment-naive patients showed that treatment with lamivudine plus interferon alfa-2b for 1 year followed by lamivudine alone for 3 additional years is an approach that also substantially may slow the emergence of lamivudine resistance. Cumulative rates of breakthrough viremia at the end of 1, 2, 3, and 4 years of treatment were 0%, 14%, 32%, and 59%, respectively.65 These values compare with 23%, 46%, 55%, and 71% for the first 4 years of lamivudine monotherapy in another study.9 Further study of an induction/maintenance approach to HBV therapy with other antiviral combinations is needed.
Conclusions
The goal of therapy in patients with chronic hepatitis B is rapid viral suppression and long-lasting maintenance of undetectable levels of serum HBV DNA. Nucleoside/nucleotide analogues rapidly are becoming the treatment of choice for most patients with chronic hepatitis B. These agents generally require indefinite administration but usually are well tolerated, result in rapid viral suppression, improve Child-Pugh scores in cirrhotic patients, and improve overall survival. The major drawback of this treatment is the considerable risk of developing antiviral drug resistance, which occurs most frequently in lamivudine-treated patients, but also has been shown for other agents (eg, adefovir, emtricitabine, and telbivudine) evaluated in long-term studies.
Preventing emergence of resistance and virologic breakthrough may best be achieved by using an agent or combination of agents with a high genetic barrier to resistance. This is clearly not the case for lamivudine, which requires the selection of only a single mutation to result in high-level resistance and should no longer be used as monotherapy in patients with chronic HBV infection, except possibly in patients with compensated liver disease who become HBV DNA negative early in therapy. Yuen et al66 recently reported that serum HBV DNA measurements at week 4 and week 16 were useful in predicting lamivudine response at year 5, with the week 4 measurement the most advantageous in allowing earlier prediction of response and/or failure to therapy owing to resistance. The combination of lamivudine with other agents may increase the barrier to resistance, but large-scale, long-term studies still are needed to document this effect. We also need more information about long-term viral suppression and emergence of resistance to newer agents (entecavir, telbivudine, tenofovir, and emtricitabine) when they are used as monotherapy or in combination.
A combination of 2 potent nucleosides/nucleotides with different resistance profiles may prove to be the optimal first-line treatment for chronic hepatitis B, but it remains to be shown in clinical trials. However, these studies are extremely difficult to conduct and may not be feasible. Drugs such as entecavir and tenofovir have such low rates of resistance that any study to determine improved outcomes, such as reduction in already very low rates of resistance and/or added efficacy, would need huge numbers of patients treated for an extended period of time before any difference between combination therapy and monotherapy might be shown. Thus, the decision to use combination therapy de novo might have to be made before appropriate information becomes available from formal randomized controlled trials. Further evaluation of the combination of nucleosides/nucleotides with a course of interferon treatment also is warranted, given short-term results suggesting that this approach may slow the emergence of resistance to lamivudine.
Frequent, periodic assessment of viral load with a sensitive HBV DNA assay remains the best approach for early detection of resistance, and testing frequency must be tailored to the patient's disease severity and treatment history. The treatment regimen should be modified as soon as resistance is detected; agents should be added or discontinued on the basis of known effects of the detected mutations on the efficacy of specific drugs. Addition or substitution of newer antiviral agents can restore suppression of viral replication after emergence of lamivudine resistance. Nevertheless, new agents with viral targets that differ from those of currently available drugs will be needed for patients who have failed or ultimately may fail second or third treatment regimens.
|
|
| |
| |
|
|
|