 |
 |
 |
| |
TENOFOVIR (TDF) IS EFFECTIVE IN LAMIVUDINE (LAM)-RESISTANT CHRONIC HEPATITIS B PATIENTS WHO HARBOUR rtA194T AT BASELINE
|
| |
| |
Reported by Jules Levin
AASLD Nov 3 2009 Boston
Scott K. Fung 1, Tony Mazzulli 1, Morris Sherman 1, Vladimir Popovic 2
1. Department of Medicine, University of Toronto, Toronto, Canada 2. Gilead Sciences Canada, Mississauga, Canada
AUTHOR SUMMARY
rt A194T detected iin 12 TDF-naive HBV patients with l amivudine resistant CHB.
- Amost always in association with L180M + M204V/I
- Usually as pure viral species or mixed population
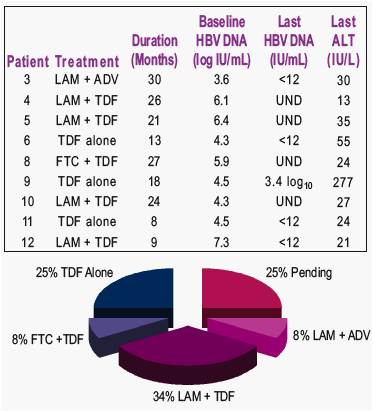
TDF alone or in combination used as salvage therapy in 10 patients
- mean treatment of 19.5 months
- HBV DNA <12 IU/mL or undetectable in 7 (88%) patients
- 1 patient underwent treatment for hepatocellular carcinoma and was admittedly non-compliant with TDF
- ALT normalized in 6 (75%) patients
AUTHOR CONCLUSIONS
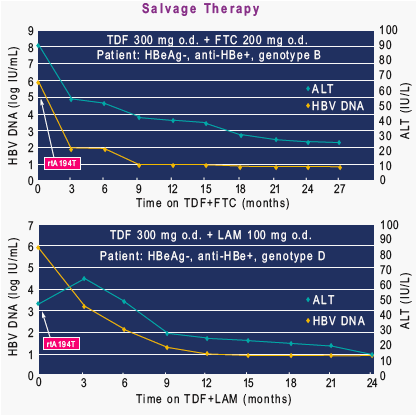
Contrary to in vitro studies, rtA194T was not associated with reduced viral suppression among LAM-resistant HBV patients salvaged with TDF alone or in combination with LAM, ADV or FTC with > 1.5 year follow-up.
These findings suggest rtA194T may represent a viral polymorphism or a LAM compensatory mutation rather than a signature TDF mutation.
Further clinical studies are required to fully characterize antiviral substitutions associated with TDF resistance.
BACKGROUND
· Tenofovir (TDF) is a potent oral nucleotide analogue of adenosine.
· TDF has demonstrated safety and efficacy in pivotal studies for the treatment of chronic hepatitis B.1
· No signature resistance mutations have been identified in patients receiving 3 years of continuous TDF therapy.2
· Antiviral-resistant mutations associated with virologic breakthrough on TDF therapy have not been fully characterized.
· 2/43 (5%) HIV-HBV coinfected patients treated with TDF plus lamivudine after 48-77 weeks were found to have rtA194T (alanine to threonine) in association with L180M + M204V.3
· An in vitro study of HBV constructs harbouring A194T+L180M+M204V showed reduced viral replication efficacy and increase in fold-resistance to tenofovir.4
· However, in vitro susceptibility of A914T examined in other studies yielded contradictory results.5
· The clinical significance of rtA194T substitution in chronic hepatitis B patients is unknown.
1 . Marcellin P. et. al. Tenofovir Disoproxil Fumarate versus Adefovir Dipivoxil for Chronic Hepatitis B. NEJM 2008; 359(23): 2442 - 2455.
2 . Heathcote E. J. et. al. Two Year TDF Treatment and ADV Switch Data in HBeAg-Positive Patients With Chronic Hepatitis B (Study 103). AASLD 2008, Abstract # 157.
3 . S h e l d o n J . e t . a l . S e l e c t i o n o f h e p a t i t i s B v i r u s p o l yme r a s e mu t a t i o n s i n H I V- c o i n f e c t e d p a t i e n t s t r e a t e d w i t h t e n o f o v i r. A n t i v i r. T h e r. 2 0 0 5 ( 1 0 ) : 7 2 7 - 7 3 4 .
4 . Amini-Bavil-Olyaee S. et. al. The rtA194T polymerase mutation impacts viral replication and susceptibility to tenofovir in hepatitis B e-antigen-positive and hepatitis B e-antigen-negative hepatitis B virus strains. Hepatology 2009 (49):1158-1165.
5 . Qi e t a l , I n t Mo l e c H B V , S e p t 2 0 0 5 , Ge rma n y.
AIM
To determine the effect of rtA194T on treatment response to TDF 300 mg daily alone or in combination with other antiviral agents in patients with lamivudine-resistant HBV.
PATIENTS & METHODS
· Adult HBV patients receiving oral antiviral therapy at University Health Network Liver Clinics (Toronto, Canada) were monitored for genotypic antiviral resistance.
· Routine bloodwork, HBV serology and HBV DNA levels were measured every 3 months on treatment.
· Resistance testing was performed on all patients who developed virologic breakthrough.
- confirmed rise in HBV DNA by ≥ 1 log IU/mL compared to nadir
- in those who failed to achieve undetectable HBV DNA 6 months after starting antiviral therapy
· Genotyping and detection of resistance mutations were performed using a line probe assay.
- InnoLiPA HBV DR v3 (InnoGenetics, Ghent, Belgium)
· HBV DNA was measured using real-time PCR (Roche, TaqMan 48, LLQ 12 IU/mL).
RESULTS

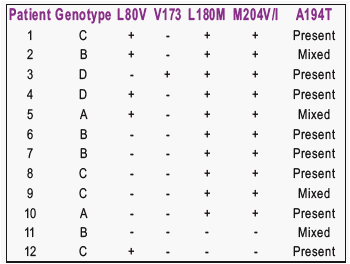
Of the 950 consecutive treatment-experienced adult patients with chronic hepatitis B tested for antiviral resistance, 12 (1.2 %) were found to harbour rtA194T.
rtA194T was found in association with rtL180M + rtM204V/I in all 10/12 (83%) patients.
After detection of LAM-resistant mutation, salvage therapy was started in 9/12 (75%) patients.

|
| |
|
 |
 |
|
|