 |
 |
 |
| |
Extending Peginterferon Alfa-2a Therapy in Patients With HBeAg-Positive Chronic Hepatitis B Who Did Not Achieve a Response at Week 48 Can Lead to HBeAg Seroconversion and HBsAg Clearance
|
| |
| |
Reported by Jules Levin
AASLD Nov 3 2009, Boston, MA, USA
Chen XP, Chen X-F,* Huang J, Chen W-L, Chen R, Ma X-J, Luo X-D
Guangdong General Hospital, Guangdong Academy of Medical Science, Guangzhou, China
Presented at the 60th Annual Meeting of the American Association for the Study of Liver Diseases 30 October - 3 November 2009, Boston, USA
Summary
Peginterferon alfa-2a was superior to entecavir in achieving HBeAg seroconversion and HBsAg clearance at week 72
One additional patient in the extension therapy group achieved HBeAg seroconversion between weeks 48 and 72 of therapy. The rate of response
was constant in the follow-up group
Rates of HBeAg seroconversion at week 48 and 72 were higher in patients
with elevated baseline ALT levels and with HBsAg <1500 IU/mL at either week 12 or 24 of treatment; however, even patients who had not achieved this HBsAg level could achieve HBeAg seroconversion at week 72
Conclusion
A finite 48-week course of peginterferon alfa-2a results in significantly higher rates of HBeAg seroconversion than entecavir. We demonstrate that peginterferon alfa-2a extension therapy is associated with increased HBeAg seroconversion. Although HBeAg seroconversion rates were particularly high in patients with HBsAg levels <1500 IU/mL during treatment, physicians should be careful when altering the treatment regimen of patients with higher HBsAg levels as some will still gain clinical benefit
Background
Nucleos(t)ide analogs and interferon-based therapy are the mainstays of treatment in chronic hepatitis B (CHB). Previously we have presented data comparing the nucleos(t)ide analog, entecavir, with peginterferon alfa-2a in patients with mildly elevated alanine aminotransferase (ALT) levels.1 We demonstrated that patients in the peginterferon alfa-2a group achieved significantly higher rates of HBeAg seroconversion than entecavir-treated
patients at week 48 (41% vs 12%, P=0.007)
As HBeAg seroconversion and HBsAg clearance are associated with long-term clinical benefit, including increased survival rates,2,3 in the current analysis we determine whether extension of peginterferon alfa-2a therapy to 72 weeks, in patients with HBeAg seroconversion or HBV DNA and HBsAg suppression after 48 weeks of therapy, can maintain or increase response rates
Recent studies of peginterferon alfa-2a have highlighted that quantification of HBsAg during therapy can help to predict those patients likely to respond to therapy at the end of therapy and post-treatment.4-6 Consequently we have also investigated whether HBsAg quantification can predict response to peginterferon alfa-2a in our patient population

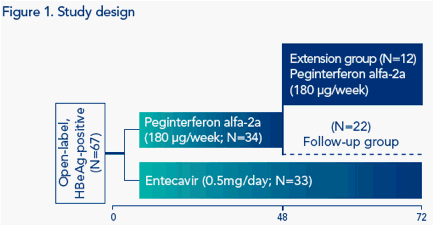
RESULTS
Response rates to peginterferon alfa-2a at week 48
After 48 weeks of peginterferon alfa-2a therapy, 14 (41%) patients had achieved
HBeAg seroconversion compared with four (12%) entecavir-treated patients
-- Three patients with HBeAg seroconversion at week 48 were included in
the extension group
At week 48, rates of HBsAg clearance were 12% (4/24) and 3% (1/33) in the
peginterferon alfa-2a and entecavir groups, respectively
-- All patients with HBsAg clearance at week 48 were included in the follow-up group
Response rates to peginterferon alfa-2a at week 72
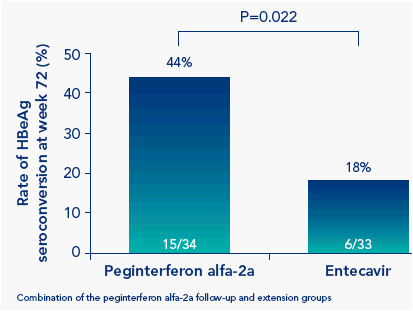
The rate of HBeAg seroconversion at week 72 was significantly higher in peginterferon alfa-2a-treated patients than in entecavir-treated patients (Figure 2)
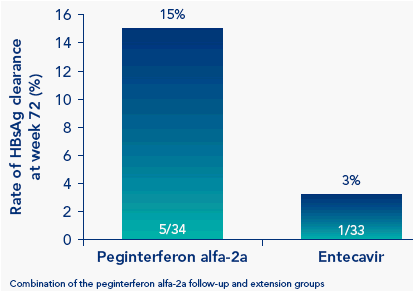
Substantially more patients receiving peginterferon alfa-2a than entecavir achieved HBsAg clearance at week 72
Figure 2. More peginterferon alfa-2a-treated patients than entecavir-treated patients achieve A) HBeAg seroconversion and B) HBsAg clearance at week 72
A
B
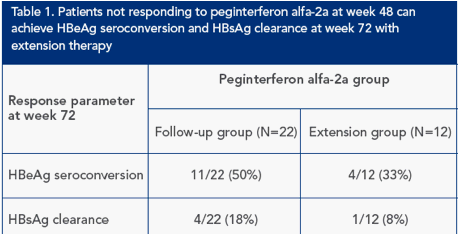
HBeAg seroconversion and HBsAg clearance at week 72 were achieved by patients in the extension and follow-up groups (Table 1)
One patient in the extension group who had not achieved HBeAg seroconversion at week 48 had achieved HBeAg seroconversion at week 72
One patient in the extension group with HBeAg seroconversion at week 48 achieved HBsAg clearance at week 72
All the patients included in the analysis had only mildly elevated ALT levels at baseline. Although the highest rates of HBeAg seroconversion at week 72 were achieved by patients with ALT> ULN at baseline (52%), even patients with normal baseline ALT levels (31%) achieved HBeAg seroconversion at week 72
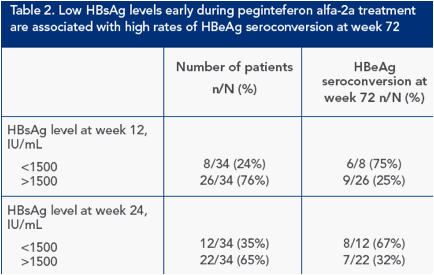
HBsAg level during treatment predicts HBeAg seroconversion at week 72 in peginterferon alfa-2a-treated patients
Response at week 72 was analyzed according to HBsAg levels at weeks 12 and 24 of peginterferon alfa-2a treatment
Rates of HBeAg seroconversion were high in patients with HBsAg <1500 IU/mL at these time points, although patients with higher HBsAg levels could also achieve HBeAg seroconversion (Table 2)
References
1. Chen X-F et al. Comparison of peginterferon alfa-2a versus entecavir in
patients with HBeAg-positive chronic hepatitis B with mildly elevated alanine
aminotransferase levels. J Hepatol 2009; 50 (suppl 1):S328
2. Fattovich G et al. Delayed clearance of serum HBsAg in compensated
cirrhosis: relation to interferon alfa therapy and disease progression. Am J Gastroenterol 1998; 93:896-900
3. Niederau C et al. Long-term follow-up of HBeAg-positive patients treated with interferon alfa for chronic hepatitis B. New Engl J Med 1996; 334:1422-1427
4. Lau GKK et al. On-treatment monitoring of HBsAg levels to predict response
to peginterferon alfa-2a in patients with HBeAg-positive chronic hepatitis B.
J Hepatol 2009; 50:S333
5. Brunetto M et al. Hepatitis B virus surface antigen levels - a guide to sustained
response to peginterferon alfa-2a in HBeAg-negative chronic hepatitis B Hepatology 2009; 49:1141-1150
6. Marcellin P et al. Increasing rates of HbsAg clearance and seroconversion
in patients with HBeAg-negative disease treated with peginterferon alfa-2a ± lamivudine: results of 5-year post-treatment follow-up. J Hepatol 2009; 50(suppl 1):S336
Disclosure information:
Editorial support for the development of this poster was provided by Elements
Communications and funded by F. Hoffmann-La Roche, Basel, Switzerland
|
| |
|
 |
 |
|
|