 |
 |
 |
| |
On-Treatment Monitoring of HBsAg Levels to Predict Response to Peginterferon Alfa-2a in Patients With HBeAg-Positive Chronic Hepatitis B
|
| |
| |
Presented at the 44th Annual Meeting of the European Association for the Study of the Liver (EASL), 22-26 April 2009, Copenhagen, Denmark
Reported by Jules Levin
Lau GKK,1 Marcellin P,2 Brunetto M,3 Piratvisuth T,4 Kapprell H-P,5 Messinger D,6 Popescu M7
1Faculty of Medicine, The University of Hong Kong, Queen Mary Hospital, Hong Kong, China and Hepatitis Free Generation Liver Center, Hong Kong; 2Service d'Hepatologie and Centre de Recherches Biologiques Bichat Beaujon (INSERM CRB3), University of Paris, Clichy, France; 3UO Epatologia, Azienda Ospedaliero Universitaria Pisana, Pisa, Cisanello, Italy;
4NKC Institute of Gastroenterology and Hepatology, Department of Internal Medicine, Songklanagarind Hospital, Prince of Songkla University, Hat Yai, Thailand; 5Abbott GmbH & Co, Wiesbaden-Delkenheim, Germany; 6IST GmbH, Mannheim, Germany; 7F Hoffmann-La Roche Ltd, Basel, Switzerland
Author Summary
A finite course of peginterferon alfa-2a ± lamivudine therapy results in responses that
increase post-treatment
The HBsAg decline during therapy in patients achieving a post-treatment response to
peginterferon alfa-2a ± lamivudine is more pronounced than in patients not responding
to therapy or in those relapsing post-treatment
51% of patients with HBsAg levels ≤1500 IU/mL at week 12 achieved HBeAg 6 months
post-treatment. A similar rate of HBeAg seroconversion was achieved in patients with
HBsAg ≤1500 IU/mL at week 24, but more patients had this low level of HBsAg (32%)
Rates of HBsAg clearance in patients with HBsAg levels ≤1500 IU/mL at week 12 or week 24 (11% and 12%, respectively) were substantially higher than in patients with higher levels of HBsAg
Author Conclusions
HBsAg level ≤1500 IU/mL during treatment was associated with high rates of sustained response 6 months and 1-year post-treatment in patients with HBeAg-positive CHB.
Response could be predicted as early as week 12, but could be predicted with more confidence at week 24.
Importantly, HBsAg level during treatment was a good predictor of post-treatment HBsAg clearance - the closest we can achieve to clinical cure of CHB. Strategies are needed to achieve greater response rates in patients with the highest on-treatment levels
Background
Data from a large-scale, phase 3 study showed that treatment with peginterferon alfa-2a for 48 weeks can result in HBeAg seroconversion 6 months post-treatment in patients with HBeAg-positive chronic hepatitis B (CHB). Importantly the rate of HBeAg seroconversion in peginterferon alfa-2a-treated patients increases during longer term follow-up from 32% 6 months post-treatment to 42% 1-year post-treatment in
peginterferon alfa-2a monotherapy-treated patients1,2
Patients with HBeAg-positive CHB who achieve a sustained post-treatment response
to interferon-based therapy have an increased chance of developing HBsAg clearance
during long-term follow-up.3 These findings confirm observations on increasing
rates of HBsAg clearance seen in patients with a sustained response to conventional
interferon4
Recent advances in HBsAg measurement allow HBsAg to be quantified during therapy and it has been suggested that the change in serum HBsAg levels from baseline during treatment could be used to identify patients likely to respond to peginterferon alfa-2a post-treatment5,6
Objective
To investigate the potential association between HBsAg levels at weeks 12 and 24 during peginterferon alfa-2a ± lamivudine treatment and post-treatment response
Methods
The initial large-scale study in HBeAg-positive patients consisted of three treatment arms - peginterferon alfa-2a (180 µg/week, N=271), peginterferon alfa-2a + lamivudine (100 mg/day, N=271) and lamivudine alone (N=271). Data for the two peginterferon alfa-2a treatment arms will be presented here
Patients received treatment for 48 weeks and were then followed up for an additional 24 weeks. The endpoint of the initial study was 6 months post-treatment (72 weeks); response at 1-year posttreatment was also assessed in patients who entered a long-term observational follow-up study
Response was defined as:
· HBeAg seroconversion
· HBV DNA ≤10,000 copies/mL (1785 IU/nL)*
· HBeAg seroconversion and HBV DNA ≤10,000 copies/mL
· HBV DNA ≤400 copies/mL (71 IU/nL)*
· HBsAg clearance
Quantitative HBsAg levels in serum were measured retrospectively pretreatment, at weeks 12, 24 and 48 of treatment and 6 months post treatment using the Abbott Architect HBsAg assay The association between response 6 months and 1-year post-treatment and HBsAg level at week 12 or week 24 was determined
*HBV DNA levels were measured using the COBAS AMPLICOR HBV MONITOR, which has a conversion factor of 1 IU/mL = 5.6 copies/mL
Results
Post-treatment virologic response to peginterferon alfa-2a ± lamivudine
Response rates at 6 months and 1-year post-treatment are shown in Table 1. Rates of response were similar in the two peginterferon alfa-2a containing groups - both being associated with increasing rates of response post-treatment
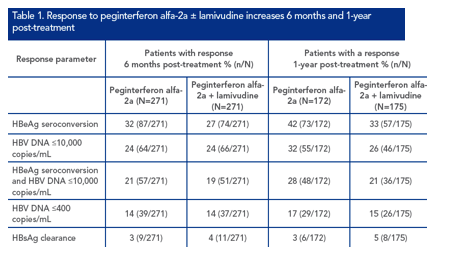
HBsAg decline in patients responding to treatment
A total of 399 peginterferon alfa-2a ± lamivudine-treated patients had HBsAg values available at baseline and at least one other timepoint (week 12, 24, 48 or 72). Of these, 137 (34.3%) - a similar proportion to the overall population - had achieved HBeAg seroconversion 6 months post-treatment and 62 (15.6%) had achieved HBV DNA ≤400 copies/mL
The rate of HBsAg decline during treatment in responders and non-responders is shown in Figure 1. Baseline HBsAg level was 3.97 ± 0.72 log10 IU/mL in patients with HBeAg seroconversion and 4.21 ± 0.63 log10 IU/mL in patients without HBeAg seroconversion.
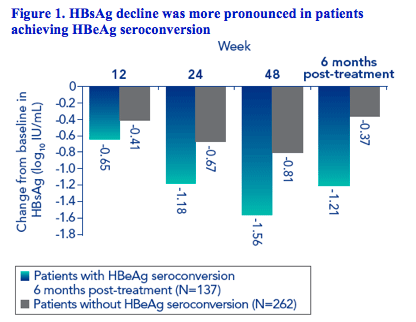
Association of HBsAg levels during therapy with HBeAg seroconversion 6 months post-treatment
Using a receiver operating curve analysis of the data at week 12 and week 24, three clusters of response were identified with low, medium, or high probability of response. The three clusters were best fitted to the HBsAg cut-offs of 1500 IU/mL and 20,000 IU/mL at weeks 12 and 24.
Consequently, the data were divided into three groups based on HBsAg level at weeks 12 or 24
The proportion of patients with HBsAg ≤1500 IU/mL increased from week 12 to week 24
· Week 12 - 21% of patients had HBsAg ≤1500 IU/mL
· Week 24 - 32% of patients had HBsAg ≤1500 IU/mL
Patients with HBsAg levels ≤1500 IU/mL at week 12 or week 24 had higher response rates than those with higher levels of HBsAg during therapy (Figures 2 and 3)
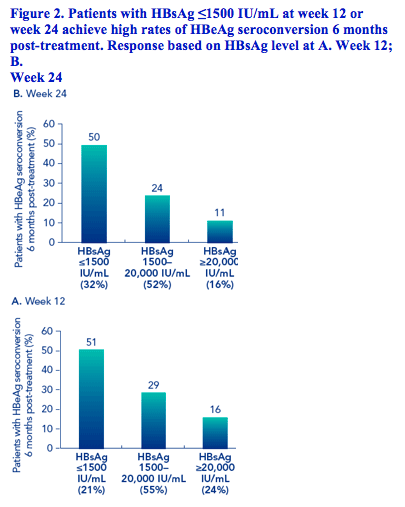
Association of HBsAg levels during therapy with HBsAg clearance 6 months post-treatment
The rate of HBsAg clearance 6 months post-treatment was higher in patients with HBsAg ≤1500 IU/mL at week 12 - 11% - compared with 2% in patients with HBsAg 1500-20,000 IU/mL and 2% in patients with HBsAg >20,000 IU/mL
Similarly using these HBsAg categories at week 24 gave HBsAg clearance rates of 12%, 1% and 0%, respectively
HBsAg decline can distinguish responders and relapsers
Some patients who achieve a response '(defined as HBV DNA ≤10,000 copies/mL) at the end of therapy (week 48) will not sustain this response post-treatment (relapsers)
The rate of decline in relapsers was less pronounced than in patients who achieved a sustained virologic response (Figure 4)
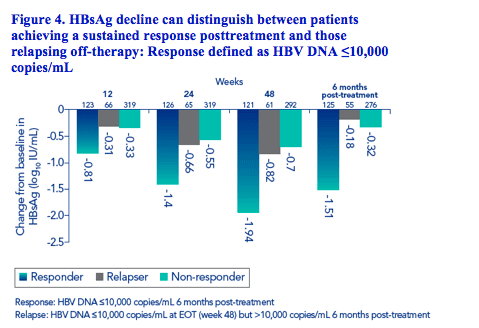
References
1. Lau GKK et al. Peginterferon alfa-2a, lamivudine and the combination for HBeAg-positive chronic hepatitis B. New Engl J Med 2005; 2682-2695
2. Lau GKK et al. Durability of response and occurrence of late response to peginterferon alfa-2a (40KD) [PEGASYS] one year post-treatment in patients with HBeAgpositive
chronic hepatitis B. EASL; April 26-30, 2006; Vienna, Austria
3. Buster EH et al. Sustained HBeAg and HBsAg loss after long-term follow-up of HBeAg-positive patients treated with peginterferon alfa-2b. Gastroenterol 2008;135:459-467
4. Moucari R et al. Early serum HBsAg drop: a strong predictor of sustained virological response to pegylated interferon alfa-2a in HBeAg-negative patients. Hepatol 2009;49:1151-1157
5. Marcellin P et al. In patients with HBeAg-negative chronic hepatitis B, HBsAg serum levels early during treatment with peginterferon alfa-2a predict HBsAg clearance 4 years post treatment. Hepatology 2008; 48 (suppl 1): 919A
6. Brunetto M et al. Hepatitis B virus surface antigen levels - a guide to sustained response to peginterferon alfa-2a in HBeAg-negative chronic hepatitis B. Hepatol 2009;49:1141-1150
Disclosure information: Editorial support for the development of this poster was funded by F. Hoffmann-La Roche, Basel, Switzerland
|
| |
|
 |
 |
|
|