| |
Long-term efficacy of tenofovir monotherapy for hepatitis B virus-monoinfected patients after failure of nucleoside/nucleotide analogues
|
| |
| |
Hepatology Jan 2010
Florian van Bommel 1 *, Robert A. de Man 2, Heiner Wedemeyer 3, Katja Deterding 3, Jorg Petersen 4, Peter Buggisch 4, Andreas Erhardt 5, Dietrich Huppe 6, Kerstin Stein 7, Jorg Trojan 8, Christoph Sarrazin 8, Wulf O. Bocher 9, Ulrich Spengler 10, Hermann E. Wasmuth 11, Jurrien G.P. Reinders 2, Bernd Moller 12, Peter Rhode 13, Heinz-Hubert Feucht 14, Bertram Wiedenmann 1, Thomas Berg 1
1Medizinische Klinik m. S. Hepatologie und Gastroenterologie Charite, Campus Virchow-Klinikum, Universitatsmedizin Berlin, Germany
2Man Erasmus MC, University Medical Center Rotterdam, Department of Gastroenterology & Hepatology, Rotterdam, The Netherlands
3Department of Gastroenterology, Hepatology and Endocrinology, Hannover Medical School, Hannover, Germany
4Department of Medicine, University of Hamburg Eppendorf, Germany
5Klinik fur Gastroenterologie, Hepatologie und Infektiologie, Universitatsklinik Dusseldorf, Germany
6Gastroenterologische Schwerpunktpraxis Herne, Germany
7Medizinische Klinik IV fur Gastroenterologie und Infektiologie, Universitatsklinikum Heidelberg, Germany
8Department of Internal Medicine I, Johann Wolfgang Goethe-University Hospital, Frankfurt, Germany
9I. Medizinische Klinik und Poliklinik, Johannes Gutenberg-Universitat Mainz, Germany
10Department of Internal Medicine, University of Bonn, Germany
11Medical Department III, University Hospital Aachen, Germany
12Hepatologische Schwerpunktpraxis, Berlin, Germany
13Abteilung fur Gastroenterologie, St. Marien Hospital, Hamm, Germany
14Laborgemeinschaft Hamburg, Germany
email: Florian van Bommel (florian.boemmel@charite.de)
Abstract
Tenofovir disoproxil fumarate (TDF) has demonstrated high antiviral efficacy in treatment-naive patients with chronic hepatitis B virus (HBV) infection but experience in nucleoside/nucleotide analogue (NA)-experienced patients is limited. In this retrospective multicenter study we therefore assessed the long-term efficacy of TDF monotherapy in patients with prior failure or resistance to different NA treatments. Criteria for inclusion were HBV DNA levels >4.0 log10 copies/mL at the start and a minimum period of TDF therapy for at least 6 months. In all, 131 patients (mean age 42 ± 12 years, 95 male, 65% hepatitis B e antigen [HBeAg]-positive) were eligible. Pretreatment consisted of either monotherapy with lamivudine (LAM; n = 18), adefovir (ADV; n = 8), and sequential LAM-ADV therapy (n = 73), or add-on combination therapy with both drugs (n = 29). Three patients had failed entecavir therapy. Resistance analysis in 113 of the 131 patients revealed genotypic LAM and ADV resistance in 62% and 19% of patients, respectively. The mean HBV DNA level at TDF baseline was 7.6 ± 1.5 log10 copies/mL. The overall cumulative proportion of patients achieving HBV DNA levels <400 copies/mL was 79% after a mean treatment duration of 23 months (range, 6-60). Although LAM resistance did not influence the antiviral efficacy of TDF, the presence of ADV resistance impaired TDF efficacy (100% versus 52% probability of HBV DNA <400 copies/mL, respectively). However, virologic breakthrough was not observed in any of the patients during the entire observation period. Loss of HBeAg occurred in 24% of patients and HBsAg loss occurred in 3%. No significant adverse events were noticed during TDF monotherapy. Conclusion: TDF monotherapy induced a potent and long-lasting antiviral response in NA-experienced patients with previous treatment failure. Our data may have implications for current add-on strategies. (HEPATOLOGY 2009.)
"Current guidelines recommend add-on strategy with ADV or TDF to ongoing lamivudine treatment as the optimal therapeutic approach for LAM resistance. However, its effectiveness depends on the level of HBV DNA at the time of treatment modification. In LAM-resistant patients with HBV DNA >106-108 copies/mL, the probability of achieving undetectable levels by adding ADV is low.[1] By contrast, no effect of viral load with respect to complete HBV DNA suppression was seen in our LAM-resistant patients on TDF monotherapy. In conclusion, TDF 300 mg per day as monotherapy is an effective and well-tolerated treatment option for patients with HBV monoinfection and NA treatment failure either due to incomplete ADV response or LAM resistance. Optimal management of genotypic ADV resistance and possible cross-resistance to TDF should be the subject of further studies."
A major limitation of nucleoside and nucleotide analogues (NA) in hepatitis B virus (HBV) therapy is the selection of HBV resistance variants, which can lead to a rebound in HBV replication and exacerbation of HBV-related disease. HBV polymerase gene variants that mediate HBV resistance are known to confer cross-resistance to other NAs. Today, increasing numbers of patients have experienced NA treatment failure, mostly to lamivudine (LAM) or adefovir dipivoxil (ADV), which poses a growing problem for antiviral treatment. Add-on combination therapy with ADV plus LAM was shown to be effective in these patients but only when initiated during the early stages of resistance development.[1]
Tenofovir disoproxil fumarate (TDF) was licensed in 2008 for the treatment of HBV infections in Europe and the United States. It has shown antiviral efficacy against a broad spectrum of viral infections, including human immunodeficiency virus (HIV-1) and HBV infections, and has a decade of use in HIV-1 patients.[2] From 2002 onwards, TDF efficacy in HBV therapy was demonstrated in small studies in which the majority of patients had HIV-1 coinfection and in some patients receiving combination therapy with LAM.[3-10] TDF is molecularly similar to ADV but can be administered at higher doses due to its more favorable safety and tolerability profile. Most recently, its strong and lasting antiviral effect was confirmed by two randomized trials in mostly treatment-naive HBV monoinfected, hepatitis B e antigen (HBeAg)-positive and HBeAg-negative patients.[11] In HIV-1 and HBV infection, TDF has a favorable safety and tolerability profile.[12]
However, the potential of TDF to treat patients with NA treatment failure has not yet been systematically studied.[5-11] In vitro, TDF exerts antiviral efficacy against variants conferring LAM resistance, but possesses some degree of cross-resistance to ADV.[13][14] This retrospective cohort study was therefore conducted to evaluate the effectiveness of TDF monotherapy in treatment-experienced, HBV monoinfected patients with prior LAM and/or ADV failure.
Results
Antiviral Efficacy of TDF.
Baseline characteristics of the 131 eligible patients are shown in Table 1. In total, 101 of these patients received TDF for at least 12 months and the median time on TDF treatment was 23 months (range, 6-60 months). TDF treatment resulted in a decrease from a mean of 7.6 ± 1.4 log10 copies/mL to undetectable HBV DNA levels in 79% of all patients during the observation period.
To assess the long-term efficacy of TDF treatment, the probability of achieving undetectable HBV DNA levels was calculated for all timepoints of TDF treatment by Kaplan-Meier analysis (Fig. 1). A Kaplan-Meier analysis, which describes a binary outcome, could be applied because during the observation period no reincrease of HBV DNA levels after suppression to undetectable levels was observed in any of the patients. In this analysis, 90% of the patients remaining in the analysis had achieved undetectable HBV DNA levels after 20 months of treatment. Host factors, such as age, sex, presence of liver cirrhosis, or the HBeAg status at baseline, had no influence on antiviral response to TDF (data not shown).
Response to TDF by Presence of Variants Associated with HBV Resistance to NA.
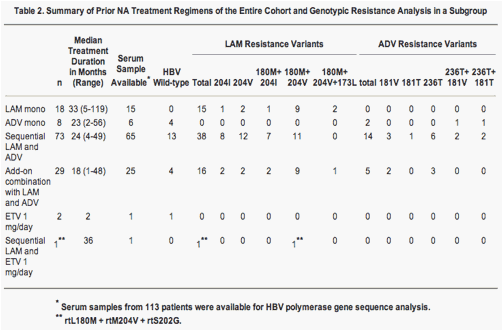
Among the 113 patients with available serum samples at baseline of TDF therapy, mutations associated with resistance against LAM or ADV were detected in 70 (62%), and in 21 (19%) patients. The remaining 22 patients (19%) had HBV wildtype sequences (Table 2). The mean baseline HBV DNA levels in these three patient groups were 8.3 ± 8.8 (range, 4.1-9.7), 8.4 ± 8.7 (range, 4.8-9.4) and 8.3 ± 8.9 (range, 4.1-9.7) log10 copies/mL (P = 0.92, log rank). In patients with HBV wildtype infection suboptimal response to previous ADV treatment was the main reason for switching to TDF (Table 2). A comparison between the patient groups with genotypic resistance either against LAM (n = 70) or against ADV (n = 21) and those with wildtype HBV (n = 22) showed that the presence of mutations associated with LAM resistance did not affect the decrease of HBV DNA during TDF treatment (Fig. 2). By contrast, patients with genotypic resistance against ADV had a significantly lower probability of achieving a complete virologic response with HBV DNA levels <400 copies/mL (P < 0.001; Fig. 2). Thus, after 12 months of TDF treatment, 33% of the patients with initial ADV genotypic resistance and 90% of the patients without initial ADV resistance had reached HBV DNA levels below the limit of detection. The mean HBV DNA levels in the three patient groups with LAM resistance, ADV resistance, and HBV wildtype after 12 months of TDF treatment were 3 ± 3.4 (range, 2.6-4.2), 5.6 ± 6.2 (range, 2.6-6.8), and 2.9 ± 3.3 (range, 2.6-4.1) log10 copies/mL (P = 0.001). Overall, during the complete observation period the probability of achieving HBV DNA levels below 400 copies/mL was 52% for patients with ADV-resistant variants and 100% for those without.
Within the subgroup of ADV-resistant patients the level of HBV DNA at the beginning of TDF treatment was the only factor that significantly influenced the probability of complete virologic response (Fig. 3). In contrast, no other factors like ALT levels, age, gender, treatment history with ADV per se (without genotypic resistance) either as monotherapy or add-on combination therapy with LAM as well as pretreatment duration with either ADV or LAM influenced TDF response (Fig. 3). Additionally, the different HBV polymerase gene mutation patterns within the groups of patients with either LAM or ADV resistance had no influence on subsequent responsiveness to TDF (Fig. 4).
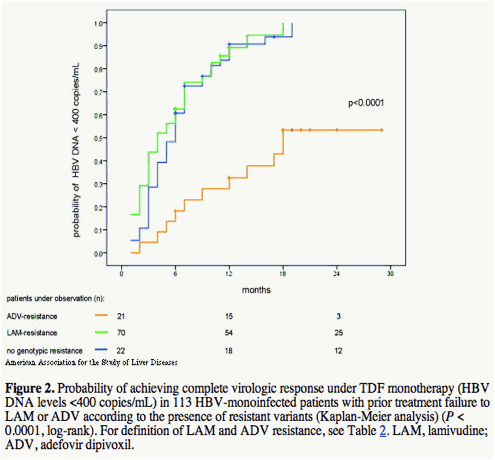
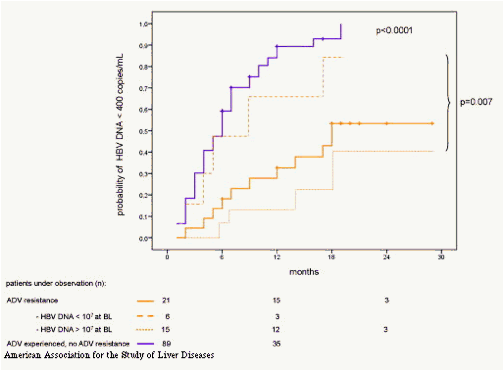
Figure 3. Probability of achieving complete virologic response under TDF monotherapy (HBV DNA levels <400 copies/mL) in 110 HBV-monoinfected patients with prior ADV treatment failure according to the presence (orange line) or absence (purple line) of genotypic ADV resistance. In ADV-resistant patients, response to TDF was additionally calculated for those with low (<107 copies/mL; dotted line), and high HBV DNA levels (=107 copies/mL; broken line) at baseline. ADV, adefovir dipivoxil.
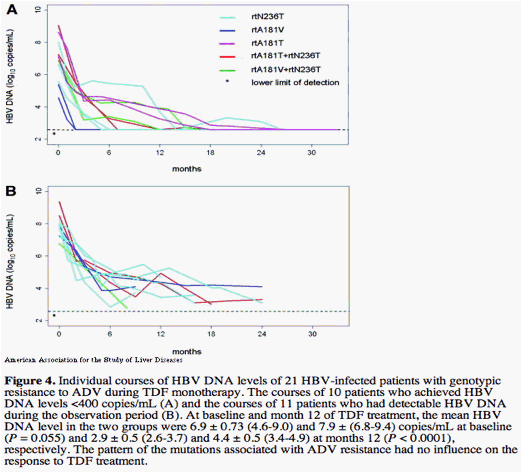
One patient who previously had ongoing HBV replication during 36 months of entecavir treatment was found to have the entecavir-resistant HBV variants rtL180M, rtM204V, and rtS202G. Nevertheless, this patient showed an immediate response to TDF, and HBV DNA levels were reduced to <400 copies/mL after 12 weeks.
HBeAg and HBsAg Response.
After a median duration of 11 months (range, 2-34 months) of TDF treatment, HBeAg seroconversion occurred in 20 of the 85 HBeAg-positive patients (24%). HBsAg loss was observed in four patients (3%) after 9, 17, 23, and 25 months of TDF treatment, respectively; all of them were HBV genotype A.
ALT levels normalized in 64 of 96 patients (65%) who had elevated ALT levels at the start of the observation period. At the end of observation ALT levels were within normal ranges in 84% of all patients.
Safety and Tolerability.
TDF treatment was well tolerated and no clinically significant side effects were reported. No significant increase in creatinine was observed (mean creatinine levels were 72 mol/L [range, 27-105] at baseline and 74 mol/L [range, 28-97] at the end of the observation period) apart from a mild elevation of creatinine level from 91 mol/L to 112 mol/L which was observed in one patient at month 12. However, for this patient creatinine levels subsequently decreased to within the normal range without modification of TDF treatment. No ALT flares (>5 x upper limit of normal) were observed during TDF treatment.
Patients and Methods
This retrospective cohort study included 16 centers in Germany and one center in the Netherlands. Patients were collected from the Departments of Hepatology and had received TDF monotherapy between 2002 and 2007. Criteria for entering this study were HBV DNA levels >/=4 log10 copies/mL at the initiation of TDF treatment and a minimum period of the TDF therapy of 6 months after failure to a previous NA therapy. Failure of a preceding NA treatment was defined as either null response (<1 log decline in HBV DNA within the first 3 months), as incomplete response (persistent and measurable HBV DNA levels during NA treatment >/=6 months), or as resistance to therapy (reincrease of HBV DNA >1 log after initial decrease). Adherence to previous therapy was a further prerequisite for study inclusion as assessed by the treating physician. All patients were directly switched from their preceding therapy to TDF (300 mg orally, once daily). Patients had to be at least 18 years of age and could be either HBeAg-positive or HBeAg-negative.
In total, 168 HBV monoinfected patients who were treated with TDF monotherapy were identified. Informed consent was obtained from all patients before the start of TDF treatment. Thirty-seven out of the 168 patients did not fulfill the entry criteria and were excluded from this analysis: nine patients had an HBV DNA level <4 log10 copies/mL at baseline, 14 patients had been treated with TDF for less than 6 months, in six patients nonadherence to medication was reported by the treating physician, and eight patients had no preceding exposure to other NA treatments and received TDF as first-line therapy. The remaining 131 patients fulfilled all entry criteria and were further followed in this analysis (Table 1).
Pretreatment Prior to TDF Monotherapy.
Of the 131 eligible patients, 121 patients (93%) were LAM-experienced and 110 (85%) were ADV-experienced. Most patients had previously received sequential therapy with LAM and ADV (56%) or combination therapy with LAM and ADV (22%) after HBV DNA breakthrough during monotherapy. Three patients showed no response to entecavir treatment (Table 2).
Study Objectives and Endpoints.
The primary study objective was to evaluate the long-term efficacy and safety of TDF monotherapy in HBV treatment-experienced patients with chronic HBV monoinfection. The secondary study objective was to determine the frequency of viral breakthrough due to HBV resistance, defined as an increase of HBV DNA >1 log copies/mL during TDF treatment.
The primary endpoint was defined as HBV DNA level <400 copies/mL (lower limit of detection) at the end of follow-up. Secondary endpoints were serum HBeAg and hepatitis B surface antigen (HBsAg) loss or seroconversion, alanine aminotransferase (ALT) normalization, genotypic resistance development, and safety and tolerability.
Clinical and Laboratory Assessments.
Serum HBV DNA levels and ALT and creatinine levels were routinely assessed by the treating physician every consecutive 3-6 months after starting TDF treatment. Serum HBV DNA was measured using either Roche Amplicor or Roche TaqMan (lower limit of detection 400 copies/mL; Roche Diagnostic Systems, Branchburg, NJ; all results are expressed in copies/mL). All data were collected from patient records and retrospectively analyzed.
Adherence to TDF therapy was assessed according to the judgment of the treating physician. Safety and tolerability were assessed by evaluation of documented side effects and laboratory abnormalities.
In all, 113 out of the 131 patients were screened for HBV resistance-associated mutations within the HBV polymerase gene by direct sequencing, as described,[15] using stored frozen serum samples taken at TDF baseline. No baseline serum samples were available for sequencing in the remaining 18 patients. The recently published nomenclature for amino acid positions in the HBV polymerase gene was used.[16] With nested polymerase chain reaction (PCR) using the primers 252 (5-AGACTCGTGGTGGACTTCTCT-3) and 1309 (5-AGAATGTTTGCTCCAGACC-3) as external primers and 377 (5-GGATGTGTCTGCGGCGTTT-3) and 998 (5ACGTTGACAGACTTTCCAATC-3) as internal primers, a PCR product bridging region from codon rt88 to codon rt282 was amplified. The PCR products were separated on 2% agarose gel (NuSieve 3:1; FMC, Rockland, ME), eluted with Gene-Clean (Bio 101, Vista, CA), and directly sequenced using a BigDye Terminator Cycle Sequencing Ready Reaction Kit (Applied Biosystems, Foster City, CA) with an automated sequencer (ABI-Prism; Applied Biosystems). The derived sequences of both strands of the amplification products were investigated for all mutations described to be associated with resistance against LAM and telbivudine as rtV173L, rtL180M, and rtM204I/V/S, against entecavir as rtT184G, rtS202I, and rtM250V, and against ADV as rtA181V/T and rtN236T. HBV genotypes were determined by sequence alignment of the overlapping HBsAg with HBV sequences derived from GenBank.[17]
Statistical Analysis.
For statistical data analysis, SPSS software for Windows, release 11.0 (Chicago, IL), was used. HBV DNA initially measured in copies/mL was converted to the base 10 logarithmic scale, with a lower limit of detection of 400 copies/mL, corresponding to 2.6 log10. For the primary endpoint, the time until the HBV DNA level reached <400 copies/mL was estimated using Kaplan-Meier methodology, and time to event subgroup comparisons were performed using the Log-Rank test. Categorical data were analyzed with the two-sided Fisher exact test. To determine the influence of baseline HBV DNA levels, the cohort was divided into patients with high and low HBV DNA levels, using a threshold of 107 copies/mL.
|
|
| |
| |
|
|
|